Abstract
Based on tanaidacean material collected primarily during the IceAGE cruises in the North Atlantic, three new species from the family Akanthophoreidae are described; one is classified in the genus Parakanthophoreus, and the other two represent a new genus—Brixia n. gen. The main characters that distinguish Brixia n. gen. from other akanthophoreids is the lack of fully developed pleopods in adult females and a seven-articled antenna. The diagnosis of the family Akanthophoreidae is amended. Cheliped ornamentation of Parakanthophoreus catharina n. sp. is illustrated using SEM pictures.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sieg (1986a) proposed the tanaidacean subfamily Akanthophoreinae (Anarthruridae) to accommodate paratanaoids characterized by, e.g., a four-articled antennule, six-articled antenna, mandibular molar usually pointed, at least the carpus of pereopod-1 with spines, pleopods well-developed, reduced or absent, and uropods with one or two rami. The broad diagnosis resulted in a prolific composition of this subfamily and in a short time it appeared to be a non-monophyletic taxon. Most of the genera originally included in Akanthophoreinae were moved later to other families (Larsen and Wilson 2002), e.g., Araphura Bird & Holdich, 1984 and Araphuroides Sieg, 1986 to Tanaellidae, and Filitanais Kudinova-Pasternak, 1973 to Colletteidae; other genera were placed in the family incertae sedis, e.g., Andrognathia Sieg, 1983, Leptognathioides Bird & Holdich, 1984, and Robustochelia Kudinova-Pasternak, 1983.
The Akanthophoreinae was rediagnosed and elevated to family level by Błażewicz-Paszkowycz and Bamber (2011) with only the genus Akanthophoreus Sieg, 1986 included. Later, Błażewicz-Paszkowycz et al. (2013) added Chauliopleona Dojiri & Sieg, 1997 to this family, but without giving a justification for this reclassification. The most recent revision of this family was by Larsen and Araújo-Silva (2014), who, with a phylogenetic approach, accommodated within Akanthophoreidae a few other genera: Mimicarhaphura Sieg, 1986; Paraleptognathia Kudinova-Pasternak, 1981; Stenotanais Bird & Holdich, 1984; Tumidochelia Knight, Larsen & Heard, 2003; and the newly described Parakanthophoreus Larsen & Araújo-Silva, 2014. Most recently, Bird (2015) added Saurotipleona to this list of akanthophoreids. One of the new taxa described below, a Parakanthophoreus species, has a strong resemblance to the genus Paraleptognathia Kudinova-Pasternak, 1981, which has been systematically confusing since its first diagnosis (Bird 2007; Błażewicz-Paszkowycz and Bamber 2011; Larsen and Araújo-Silva 2014).
Larsen (2005) pointed out that in deep-sea tanaidomorphan taxa females may lack pleopods. It was previously recorded for, e.g., all genera in the family Agathotanaidae; Caudalonga Larsen, 2005; Cetiopyge Larsen & Heard, 2002 and Collettea Lang, 1973 from the Colletteidae, and Anarthrura Sars, 1882 from the Anarthruridae. Until now, the Akanthophoreidae was represented exclusively by taxa with well-developed pleopods but a new genus described below is with reduced pleopods or completely without pleopods.
In this paper, we deal with a series of akanthophoreids collected in the waters off Iceland during the IceAGE (Icelandic Animals Genetic and Ecology; Brix et al. 2014) initiative, which allows us to erect a new genus with two new species and describe a Paraleptognathia-like species of Parakanthophoreus.
Material and methods
Samples were collected during two IceAGE cruises. The first took place in August/September of 2011 on board the R/V Meteor and the second in August 2013 on board the R/V Poseidon. During those initiatives, various sampling gears were deployed, e.g., Van Veen grab, Shipek grab, epibenthic sledge, or box corer (Brix et al. 2014). Reference is also made to material collected during the BIOICE, BIOFAR, and AFEN projects from the same, and adjacent, areas (e.g., Bird 2001, 2015) (Fig. 1). For environmental data, please refer to Bird (2015), Brix et al. (2011), and Table 1 herein.
Specimens were dissected using chemically sharpened tungsten needles, mounted in glycerin on slides and sealed with nail varnish. Drawings were initially prepared using a microscope combined with a camera lucida and redrawn on a digital tablet as proposed by Coleman (2003).
The body length to width ratio was calculated using measurements from the tip of the carapace to the end of the pleotelson, and of the widest part of carapace. The length and width of articles were measured along their central axes.
Dehydration for SE microscopy was carried out sequentially in the tissue processor (Leica EMTP) with 95% and absolute ethanol. Specimens were then transferred to a critical point dryer (Leica EMCPD 300) with parameters set as follows: mixing 50%, CO 2 in–slow with 2 min delay, exchange speed–5 with 20 cycles, gas out heat and speed–fast. Dried specimens were transferred on coverslips and coated with 6-nm-thick layer of gold (Leica EM ACE200). SEM pictures were made on Phenom ProX.
The morphological terminology follows that proposed by Błażewicz-Paszkowycz et al. (2013). The term “palm” is used for the proximal part of the cheliped propodus, i.e., without the fixed finger. It is delimited by the line running through dactylus insertion and reaching the propodus ventral margin at a right angle.
The material is deposited in the Zoological Museum, Hamburg and the National Museums of Scotland (NMS), Glasgow.
Systematics
Suborder Tanaidomorpha Sieg, 1980
Superfamily Paratanaoidea Lang, 1949
Family Akanthophoreidae Sieg, 1986
Diagnosis see Larsen and Araújo-Silva (2014) but with additions: all pereopod meri and carpi with slender spines. Pleopods well-developed, reduced or absent.
Remarks. The diagnosis of the family is modified primarily regarding the pleopods, where they may be absent or rudimentary in the new genus described below.
Among the original members of the family, formerly the Tribe Akanthophoreinae within the family Anarthruridae Lang, 1971, Paraleptognathia was distinguished by a combination of: a five-articled antennule, with two aesthetascs on the terminal article, a maxillule with eight spines, an epignath tipped with a seta, the cheliped with a strong carpal shield (and all cheliped articles covered with setules), and a pleopod exopod of two articles. The unique five-articled antennules in the type species of the genus, Paraleptognathia typica Kudinova-Pasternak, 1981 (Kudinova-Pasternak 1981) were later discussed by Guerrero-Kommritz (2004) and were treated as a deviation of the standard state or a unique character of the species, especially since that P. bacescui Kudinova-Pasternak, 1985 has standard four-articled antennules (Kudinova-Pasternak 1985). This character is known to be a feature of immature or “preparatory” males in several paratanaoid groups (Bird 2007, Błażewicz-Paszkowycz et al. 2011). For over a decade, Paraleptognathia was known only from two species, but Dojiri and Sieg (1997) described P. bisetulosa and moved Akanthophoreus gracilis (Krøyer, 1842) to Paraleptognathia.
Guerrero-Kommritz (2004) synonymized Akanthophoreus with Paraleptognathia, resulting in a total of 17 species classified within this genus. This was not accepted by Bird (2007) and it was again split into Akanthophoreus, Parakanthophoreus, and Paraleptognathia by Larsen and Araújo-Silva (2014). Although the morphology of one of the new akanthophoreid species found in the Northeast Atlantic during IceAGE cruises corresponds with the main characters defining Paraleptognathia suggested by Bird (2007) and Larsen and Araújo-Silva (2014), namely a large carpal shield and cheliped articles with “ornamentation,” it is considered that Paraleptognathia is too ill-defined and poorly supported by type material to warrant the addition of new species.
Brixia n. gen.
Registered in ZooBank under: urn:lsid:zoobank.org:act:094408C2-6DD8-4161-9ACCEE0F743E505A
Diagnosis. Pleonite-5 without ventral spur. Pleotelson without lateral spurs, as long as broad or longer. Antenna with seven distinct articles. Cheliped with distinct carpal shield; carpus and propodus without ornamentation. Pleopods rudimentary or absent. Uropod basis and endopod segment without spur.
Type species. Brixia aurora n. sp., by original designation.
Etymology. The genus is dedicated to Saskia Brix, the leader of the IceAGE project.
Gender. Feminine.
Species included. B. aurora n. sp., B. borealis n. sp.
Remarks. Most of the characters in the amended diagnosis of Akanthophoreidae by Larsen and Araújo-Silva (2014) are observable in Brixia n. gen. Nevertheless, seven distinct articles in the antenna, and rudimentary or absent pleopods may be unique characters for the family; the pleotelson is also longer than in most, if not all, Akanthophoreus, Chauliopleona, Parakanthophoreus, and Paraleptognathia species, where it is shorter than broad. The antennal character derives from two articles (4 and 5) with complete division, homologous to article-4 in those antennae exhibiting partial or indistinct articulation (“fusion line” sensu Larsen et al. 2006) within this element as expressed in other akanthophoreids. The pleopod character (lack or rudimentary) is somewhat compromised by the stated absence of these in Parakanthophoreus fastuosus (Guerrero-Kommritz, 2004: 56), although this is not remarked on or included in the generic diagnosis (op.cit.: 8–10). Moreover, the results of molecular phylogeny (Błażewicz and Jennings unpublished data) unequivocally support Brixia having an affinity to, but separateness from, other akanthophoreids such as Parakanthophoreus or Chauliopleona.
Brixia aurora n. sp.
Registered in ZooBank under: urn:lsid:zoobank.org:act:42A284B1-1D31-4375-A208-6F1D8E17057C
Material examined: holotype non-ovigerous female (3.5 mm long) dissected on slides (ZMH K-52920), st. 853/989-1, 61° 42.26′ N 19° 32.21′ W-61° 42.17′ N 19° 32.03′ W, depth 1919.9–1925.6 m, EBS, 31 August 2011.
Etymology. From the Latin aurora, meaning sunrise. The name refers to the Aurora Borealis that delighted and entertained the IceAGE crew and participants during the collection of the material.
Diagnosis. Cephalothorax 1.7 times longer than wide. Pleotelson elongate, 1.4 times longer than wide, as long as last three pleonites. Antennule article-1 3.7 times longer than wide, longer than distal three articles combined length. Ischium of pereopods 1–3 and 6 with one seta (pereopods 4–5 missing). Uropod basis four times longer than wide and as long as endopod segment-1; exopod 0.6 times as long as endopod segment-1.
Description of female. Body (Fig. 2A, B) slender, 10.5 times longer than wide, 3.3 mm long. Cephalothorax subrectangular, tapering anteriorly, 1.7 times longer than wide, with slight triangular rostrum; carapace with single seta at each anterolateral corner; eyelobes poorly marked. Pereonites parallel-sided or weakly convex; pereonite-1 rectangular, shortest, 0.4 times as long as cephalothorax, 1.5 times longer than wide; pereonites 2, 4, and 5 subequal in length, 1.5 times longer than pereonite-1, as long as wide, with parallel lateral margins; pereonite-6 just shorter, 1.4 times longer than pereonite-1, as wide as long. Pleon as long as combined length of three last pereonites; pleonites subequal in length; each pleonite 2.4 times wider than long, without distinct lateral setae. Pleotelson elongate (relative to most akanthophoreids), 1.4 times longer than wide, as long as last three pleonites, slightly tapering distally, bearing two setae mid-posteriorly.
Antennule (Fig. 2A, C) about 0.75 times length of cephalothorax; article-1 3.7 times longer than wide, longer than distal three articles together, with two outer distal groups of penicillate setae and one simple seta that reaches distal margin of article-2; article-2 1.5 times longer than wide, 0.4 times as long as article-1, with outer pair of penicillate setae and single simple distal seta; article-3 1.3 times longer than wide, 0.7 times as long as article-2, with inner distal one simple seta and one penicillate seta; article-4 tapering, little longer than article-2, with five distal setae and one aesthetasc.
Antenna (Fig. 2B, D) about 0.75 times as long as antennule; article-1 very short, naked; article-2 with two distal setae; article-3 as long as wide, with dorsodistal finely denticulate seta; article-4 0.8 times as long as article-5 and as long as article-6, with ventrodistal penicillate seta; article-5 1.2 times longer than article-6, with two long ventrodistal, finely denticulate setae; article-6 more slender than article-5, with one distal seta; article-7 minute, with four distal setae.
Labrum not recovered (but visible in whole view, Fig. 2B), conical. Left mandible (Fig. 2E) with six rounded teeth on pars incisiva; lacinia mobilis narrow, crenulate; pars molaris slender, distally bent downward, with rosette of numerous distal spinules. Right mandible (Fig. 2F) pars incisiva with three blunt teeth; pars molaris as in left mandible. Maxillule (Fig. 2G, G′) with nine terminal spines (three with fine denticulation, one plumose); palp with two plumose setae. Maxilla (Fig. 2H) larger than half of maxilliped plate, subtriangular. Labium (Fig. 2I) lobes narrow, distally setulose, outer lobe with microtrichia distally.
Maxilliped (Fig. 2J) basis 1.8 times longer than wide, with single long seta reaching over distal margin of endite; endite distally with two small setae and two round tubercles; palp article-1 naked; article-2 with one outer distal finely denticulate and one simple and two inner distal finely denticulate setae; article-3 with two longer finely denticulate and one smaller simple inner distal setae; article-4 with subdistal simple seta and five distal finely denticulate setae. Epignath not recovered.
Cheliped (Fig. 3A) attached via sclerite, with elongate basis 2.2 times longer than wide, posterior lobe as long as anterior mass, latter with one dorsal seta; merus subtriangular, with single ventral seta; carpus stout, 1.5 times longer than wide, with prominent carpal shield, well-developed rounded dorsoproximal extension, two mid-ventral setae and single dorsoproximal and dorsodistal setae; propodus large, just longer but narrower than carpus, palm about as long as wide, dorsal and ventral margins smooth, with comb of three fine spines on inner surface near dactylus insertion; fixed finger with one seta near dactylus insertion, two setae ventrally and four teeth and three setae on cutting edge; dactylus with slightly undulate dorsal margin and two minute spines on cutting margin.
Pereopod-1 (Fig. 3B) coxa with simple seta; basis 3.4 times longer than wide, narrow in most proximal part, naked; ischium with one ventral seta; merus one-third as long as basis, with one minute seta and robust ventrodistal finely denticulate spine that reaches beyond distal margin of carpus; carpus 1.2 times longer than merus, with numerous microtrichia and fine distal setulation, with single dorsodistal robust finely denticulate spine and two unequal (small and long) ventrodistal finely denticulate spines; propodus 1.4 times longer than carpus, with microtrichia on ventral margin, with single ventrodistal finely denticulate spine and dorsodistal fine seta; dactylus just shorter than unguis, together 0.9 times as long as propodus.
Pereopods 2 and 3 (Fig. 3C, D) similar to pereopod-1, but pereopod-3 dactylus with accessory setae (possibly unobserved in pereopods 1–2).
Pereopods 4 and 5 missing from dissected specimen.
Pereopod-6 (Fig. 3E) basis twice as long as wide, narrow distally, naked; ischium with one ventral seta; merus one third as long as basis, with two ventrodistal finely denticulate spines (one broken), that reach midway along carpus, and ventral microtrichia; carpus 1.7 times longer than merus, with microtrichia on ventral margin, and with three distal finely denticulate spines (one broken) reaching one third along propodus and fine dorsodistal seta; propodus little shorter than carpus, with microtrichia on ventral margin, with two ventrodistal finely denticulate spines and two short dorsodistal setae; dactylus denticulate on ventral margin, 1.4 times longer than unguis, together 1.2 times longer than propodus.
Pleopods absent.
Uropod (Fig. 3F) basis naked, as long as endopod segment-1; exopod of two segments, 0.6 times as long as endopod segment-1; segment-2 with at least one short distal seta; endopod of two segments; endopod segment-1 1.2 times as long as segment-2, that is armed with one seta distally (setation incomplete on specimen).
Remarks. Brixia aurora n. sp. differs from its congener (see below) by having a relatively long pleotelson—1.4 times longer than wide, a more slender antennule article-1, slight crenulation on the dorsal margin of the cheliped dactylus, only one seta on the ischium of pereopod-6 (and possibly on pereopods 4–5), no pleopods, and elongate uropod endopod segment-2, that is 0.9 times as long as segment-1.
Distribution: Single record from the Iceland Basin, 1919.9–1925.6 m; see Fig. 1.
Brixia borealis n. sp.
Registered in ZooBank under: urn:lsid:zoobank.org:act:0139C2E7-E1F7-4F7F-9FE7-8B556EE082F4
Material examined: holotype non-ovigerous female (3.3 mm long) dissected on slides (ZMH K-52921), st. 1149, 67° 50.57' N 23° 41.85' W, depth 1246 m, AGT, 15 September 2011.
Etymology. From the Latin borealis, northern or North.
Diagnosis. Cephalothorax 1.5 times longer than wide. Pleotelson just longer than broad, shorter than last two pleonites. Antennule article-1 2.7 times longer than wide, clearly shorter than distal three articles combined length. Pereopod-6 ischium with two setae. Pleopods rudimentary. Uropod basis 1.5 times longer than wide and 0.7 times as long as endopod segment-1; exopod less than half as long as endopod segment-1.
Description of female. Body (Fig. 4A, B) slender, 11.5 times longer than wide, 3.3 mm long. Cephalothorax subrectangular, tapering towards anterior, 1.5 times longer than wide, with slight triangular rostrum, carapace with fine seta at each anterolateral corner, eyelobes poorly marked. Pereonites sub-hexagonal, wider over pereopod insertions; pereonite-1 tapering distally, shortest, 0.5 times as long as cephalothorax, 1.3 times longer than wide; pereonites 2, 3 and 4 subequal in length, 1.7 times longer than pereonite-1, 1.2 times longer than wide; pereonite-5 1.2 times longer than pereonite-6; pereonite-6 just longer than pereonite-1, as wide as long. Pleon as long as combined length of pereonites 5, 6 and two-thirds of pereonite-4; pleonites subequal in length, each pleonite 1.6 times wider than long, without lateral setae. Pleotelson 1.1 times as long as wide, and shorter than last two pleonites, slightly tapering distally, with pair of distal setae.
Antennule (Fig. 4A, C) about 0.9 times as long as cephalothorax, article-1 2.7 times longer than wide, shorter than distal three articles together, with outer distal group of penicillate setae and one simple distal seta shorter than article-2; article-2 2.3 times longer than wide, 0.7 times as long as article-1, with outer single simple distal seta; article-3 0.7 times as long as wide, and 0.7 times as long as article-4, with two small distal setae; article-4 tapering, 1.5 times longer than article-3, with four distal setae and one aesthetasc.
Antenna (Fig. 4D) about 0.75 times as long as antennule; article-1 very short, naked; article-2 1.4 times longer than article-3, with one dorsodistal seta; article-3 as long as wide, with dorsodistal seta; article-4 0.7 times as long as article-5 and 0.8 times as long as article-6, with penicillate seta distally; article-5 almost as long as article-6, with two simple and two penicillate distal setae; article-6 more slender than article-5, with one distal seta; article-7 minute, with four distal setae.
Labrum (Fig. 4E) hood shaped, relatively elongate, finely setose. Left mandible (Fig. 4F) pars incisiva with three teeth; lacinia mobilis narrow, weakly crenulate dorsally; pars molaris slender, distally bent downwards, with rosette of numerous short and two long distal spinules. Right mandible (Fig. 4G) pars incisiva with several pointed teeth. Maxillule (Fig. 4H) with nine terminal spines (three with fine denticulation, one plumose); palp (Fig. 4H′) with two plumose setae. Maxilla (Fig. 5A) larger than half of maxilliped plate, subtriangular, distally setulose. Labium (Fig. 4I) lobes narrow, distally setulose, inner lobes with two distinct spines; outer “lobe” with microtrichia.
Maxilliped (Fig. 5B, B′) basis 1.8 times longer than wide, with single, long seta reaching over distal margin of endite; endite distally with two rounded tubercles and one, fine seta; palp article-1 naked; article-2 with one outer distal and three inner distal setae (at least one of them finely denticulate); article-3 with two short simple and two finely denticulate inner setae; article-4 with subdistal seta and five finely denticulate distal setae. Epignath not recovered.
Cheliped (Fig. 5C) attached via elongate sclerite reaching to posterior of cephalothorax inserted to dorsum of basis; basis elongate, 2.3 times longer than wide, posterior lobe as long as anterior mass; merus subtriangular, with fine ventral seta; carpus stout, almost as long as wide, with well-developed rounded dorsoproximal extension and prominent carpal shield, with two fine mid-ventral setae and single dorsoproximal and dorsodistal setae (one broken); propodus large, longer than wide but shorter than carpus, palm about as long as wide, dorsal and ventral margins smooth, with comb of three fine spines on inner surface near dactylus insertion; fixed finger with one seta near dactylus insertion, two setae ventrally and three teeth and three setae on cutting edge; dactylus dorsal margin smooth, with two minute spines on cutting margin.
Pereopod-1 (Fig. 5D) coxa with simple seta (not shown on figure); basis 2.6 times longer than wide, narrow in most distal part, with long penicillate, dorsal seta; ischium with one ventral seta; merus one-third as long as basis, with one robust ventrodistal finely denticulate spine that reaches beyond distal margin of carpus; carpus 1.2 times longer than merus, with numerous microtrichia, with single dorsodistal and ventrodistal finely denticulate robust spines; propodus 1.4 times longer than carpus, with microtrichia on ventral margin and with single ventrodistal finely denticulate spine; dactylus 0.8 times as long as unguis, ventrally denticulate, together 0.9 times as long as propodus.
Pereopod-2 (Fig. 5E) similar to pereopod-1, but carpus with additional ventrodistal spine.
Pereopod-3 (not figured) as pereopod-2.
Pereopod-4 (Fig. 5F) basis twice as long as wide, narrow in most distal part, apparently with two dorsal penicillate setae; ischium with two ventral setae; merus one third as long as basis, with two ventrodistal finely denticulate spines, which reach beyond midway on carpus; carpus 1.6 times longer than merus, with microtrichia on ventral margin, and with three distal finely denticulate spines, the longest spine about 0.6 times as long as propodus and dorsodistal seta; propodus as long as carpus, with microtrichia on ventral margin, with two ventrodistal finely denticulate spines and one short dorsodistal seta; dactylus as long as propodus, denticulate on ventral margin; unguis about as long as half of dactylus, with ventral denticulation.
Pereopod-5 (not figured) as pereopod-4.
Pereopod-6 (Fig. 5G) similar to pereopod-4, but propodus with two dorsodistal setae.
Pleopods (Fig. 4B) biramous, rudimentary, without setae.
Uropod (Fig. 5H) basis 0.7 times as long as endopod segment-1, naked; exopod 0.4 times as long as endopod segment-1; segment-1 with distal seta; segment-2 with one long and one short distal setae; endopod segment-1, five times as long as wide, twice as long as segment-2, with one penicillate seta distally; segment-2 with one subdistal and two distal setae.
Remarks. In contrast to B. aurora, in B. borealis n. sp., the pleotelson is shorter, (but still as long as or just longer than broad), the cheliped dactylus is apparently smooth, the ischium of pereopods 4–6 has two setae, and the uropod endopod segment-2 is about half as long as segment-1.
Distribution: Single record from the Denmark Strait, 1246 m; see Fig. 1.
Parakanthophoreus Larsen & Araújo-Silva, 2014
Parakanthophoreus catharina n. sp.
Registered in ZooBank under: urn:lsid:zoobank.org:act:62B32816-9167-4CB9-82A9-C69AD931AE52
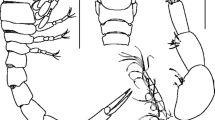
Parakanthophoreus catharina n. sp. female (ZMH K-52922) A antennule; B antenna; C labrum; D left mandible; E right mandible; F maxillule; G maxilla; H labium; I maxilliped; J epignath. A–D, F–H and J female paratype; E and I preparatory male allotype (NMS.Z.2018.26.3), DTI2000 Stn 55374#1. Scale bars 0.1 mm for B–D, F, H and J and 0.25 for A, E and I
Akanthophoreus inermis (Hansen, 1913): Bird (2001): 32–33.
Material examined: holotype non-ovigerous female (2.8 mm long) (NMS.Z.2018.26.1), DTI2000 st. 55283#1, 61° 35.06′ N 1° 58.43′ W, depth 1262 m, Megacore, 5 August 2000.
Allotype preparatory male (2.5 mm long) (NMS.Z.2018.26.2), partly dissected on microslide(NMS.Z.2018.26.3), DTI2000 st. 55374#1, 62° 18.48′ N 0° 10.51′ E, depth 907 m, Megacore, 22 August 2000.
Paratype female dissected on slides (ZMH K-52922), st. 853/1132-1, 67° 38.39′ N 26° 45.46' W-67° 38.28′ N 26° 45.72′ W, depth 316.6–316.8 m, EBS, 14 September 2011. Other material taken during IceAGE project: one non-ovigerous female (2.4 mm long), one manca (1.3 mm long), st. 853/1116-1, 67° 12.82′ N 26° 16.31′ W, depth 683.1 m, GKG, 14 September 2011; one non-ovigerous female (1.6 mm long), one manca (1.3 mm), st. 853/1129-1, 67° 38.77′ N 26° 44.78′ W, depth 320.6 m, GKG, 14 September 2011; four non-ovigerous females (1.4, 1.6, 2.3, and 2.3 mm long), one manca (1.2 mm long), st. 853/1141-1, 67° 50.22′ N 23° 42.11′ W, depth 1241.6 m, GKG, 15 September 2011; one female with marsupium, one non-ovigerous female (1.7 mm long), one manca (0.9 mm long), st. 873-4, 61° 46.52′ N 03° 52.51′ W, depth 833.8 m, Van Veen grab, 28 July 2013.
Supplementary material and records examined—see Table 1 for station data: one specimen, AFEN 1996 53819#3; one specimen, AFEN 1996 53828#1; one specimen, AFEN 1996 53833#1; one specimen, AFEN 1998 54509#2; one specimen, AFEN 1998 54521#1,3; one specimen, AFEN 1998 54522#1,2; two specimens, AFEN 1998 54524#2; one specimen, BIOICE 2010; 11 specimens, BIOICE 2011; four non-ovigerous females, BIOICE 2030; one non-ovigerous female, one ovigerous female, BIOICE 2077; two non-ovigerous females, BIOICE 2088; two manca-3, four non-ovigerous females, BIOICE 2315; six non-ovigerous females, two ovigerous females, one post-ovigerous female, BIOICE 2317; one manca-3, 12 non-ovigerous females, four ovigerous females, one preparatory male (uncertain?), BIOICE 2318; one ovigerous female, BIOICE 2323; one non-ovigerous female, one ovigerous female, two post-ovigerous females, BIOICE 2364; one non-ovigerous female, one ovigerous female, BIOICE 2638; one preparatory male (?), BIOICE 2743; one non-ovigerous female, BIOICE 2749; three non-ovigerous females, one non-ovigerous female, BIOICE 2762; one specimen, BIOICE 2765; one specimen, BIOICE 3023; one non-ovigerous female, BIOICE 3108; four specimens, two non-ovigerous females, two preparatory males (?), BIOICE 3115; two specimens, BIOICE 3121; one specimen, BIOICE 3198; one specimen, BIOICE 3225; three non-ovigerous females, four specimens, BIOFAR 167; six specimens, BIOFAR 168; one specimen, BIOFAR 169; two specimens, BIOFAR 170; seven specimens, BIOFAR 172; seven specimens, BIOFAR 478; six specimens, BIOFAR 769; four non-ovigerous females, DTI2000 55283#1; three non-ovigerous females, DTI2000 55322#1; three non-ovigerous females, DTI2000 55324#; one non-ovigerous female, DTI2000 55336#1; three non-ovigerous females, DTI2000 55344#1; one preparatory male (?), DTI2000 55344#2; one non-ovigerous female, DTI 2000 55347#1; one preparatory male (?), DTI2000 55351#1; two non-ovigerous females, DTI2000 55352#1; one non-ovigerous female, DTI2000 55354#1; two non-ovigerous females, DTI2000 55355#1; one non-ovigerous female, DTI2000 55374#1; two non-ovigerous females, DTI2000 55375#3; one non-ovigerous female, DTI2000 55376#1; one non-ovigerous female, DTI2000 55378#1; one non-ovigerous female, DTI2000 55379#1; one non-ovigerous female, DTI2000 55395#1; two non-ovigerous females, SEAS4 57003#1&2; two non-ovigerous females, two post-ovigerous females, SEAS4 57008#1-3; one specimen, SEAS4 57080#1; one non-ovigerous female, SEAS4 57088#1.
Etymology. Catharina (Lat.) (= Katarzyna in Polish): the species is dedicated to the senior author’s wife, Katarzyna Pawicka-Jóźwiak.
Diagnosis. Body about ten times longer than wide. Pereonite-1 as long as wide. Maxilliped basis with setae. Maxilliped endite with pair of distal setae. Cheliped carpus, propodus and dactylus without crenulation but carpus with deep shield; cheliped carpus with single ventral seta; fixed finger ventral setae unequal in thickness. Pereopods 1–3 carpus with dorsodistal seta. Pereopods 4–6 ischium with pair of setae and dactylus with denticulate ventral margins. Pereopod-6 propodus with three dorsodistal finely denticulate spines. Uropod exopod 0.7 times as long as endopod segment-1.
Description of non-ovigerous female. Body (Fig. 6A) slender, 10.1 times longer than wide, 2.8 mm long (range 1.24–2.98 mm, n = 70 G. Bird material). Cephalothorax 15% of total body length, about 1.5 times longer than wide, carapace with fine seta at each anterolateral corner, eyelobes poorly marked. Pereonites parallel-sided, length/width ratio: 1, 1.1, 1.4, 1.4, 1.1, and 0.9. Pleon 18% of total body length, pleonites subequal in length, epimera with single seta. Pleotelson longer than pleonites 4–5, with rounded apex. Cephalothorax and tergites of pereonites and pleonites with dense pitting.
Antennule (Figs 6A, 7A) about 0.75 times as long as cephalothorax; article-1, twice as long as wide, with one simple and two penicillate setae on outer distal margin; article-2 about half as long as article-1, with one short inner distal seta and one long outer distal seta; article-3 half as long as article-2, with one short inner distal seta, one longer outer distal seta and some distal spinules; article-4 1.5 times longer than article-3, with two simple setae distally; article-5 minute, with four simple setae distally. Articles with dense pitting.
Antenna (Figs 7B, 9A) article-1 very short, naked; article-2 just longer than wide, with two dorsodistal and distolateral setae; article-3 0.7 times as long as wide, with one simple distal seta; article-4 3.8 times longer than wide, with fusion line, one penicillate seta near fusion line, one penicillate seta subdistally (partly broken), one minute and four simple setae distally; article-5 just longer than half of article-4, with two simple setae distally; article-6 minute, with five simple setae distally. Articles with dense pitting.
Labrum (Fig. 7C) trapezoidal in lateral view, distally setose. Left mandible (Fig. 7D) pars incisiva with five blunt denticles; lacinia mobilis weakly crenulate dorsally; pars molaris narrowing distally, with rosette of numerous distal spinules. Right mandible (Fig. 7E) pars incisiva with three denticles distally; pars molaris as in left mandible. Maxillule (Fig. 7F) endite with some inner microtrichia, subdistally with setae, with eight terminal spines (at least three finely denticulate); palp not observed. Maxilla (Fig. 7G) triangular, inner and distal margin setose. Labium (Fig. 7H) simple, slender, distally with spinules.
Maxilliped (Fig. 7I) basis with one distal seta, half as long as endite; endite with distal tubercle, pair of distal setae and outer distal setules; palp article-1 naked; article-2 with one simple outer seta and one plumose and two simple setae on inner distal margin; article-3 with two simple short, and two long setae on inner margin; article-4 with one subdistal seta and five distal setae. Epignath (Fig. 7J) slender, tapering distally to the pointed tip.
Cheliped (Figs 8A, 9B, 10A–D) attached via large posterodorsal sclerite; posterior lobe and anterior mass subequal in size, naked; merus with one simple ventral seta; carpus with well-developed rounded dorsoproximal extension and deep shield, one simple midlength seta ventrally and one simple seta dorsodistally; propodus about half as long as wide, palm about as long as wide, dorsal and ventral margins smooth, with simple seta near dactylus insertion, and one longer and three short finely denticulate spines on inner margin, inner and outer margins with microtrichia, outer margin additionally with protrusions; fixed finger with two denticles and three simple setae on cutting edge, and two setae unequal in thickness ventrally; dactylus as long as fixed finger, naked. Articles with dense pitting (Figs 9B, 10).
Pereopod-1 (Figs 8B, 9C) coxa with simple seta (not shown on figure); basis 1.9 times longer than wide, naked; ischium with one seta; merus about as long as carpus, with ventral microtrichia, distal seta, and ventrodistal finely denticulate spine; carpus with one complex-tip seta and finely denticulate spine dorsodistally, one finely denticulate spine ventrodistally and numerous microtrichia along article; propodus 1.4 times longer than carpus, with ventrodistal spine and ventral microtrichia; dactylus and unguis combined 0.6 times as long as propodus; unguis 1.7 times longer than dactylus. Articles of all pereopods with dense pitting.
Pereopod-2 (Fig. 8C) similar to pereopod-1, but basis with long penicillate seta dorsally and carpus with additional ventrodistal finely denticulate spine.
Pereopod-3 (Fig. 8D) similar to pereopod-2, but basis without penicillate seta (possibly lost) and dactylus with proximal seta.
Pereopod-4 (Fig. 8E) basis 2.4 times longer than wide, narrower than those of pereopods 1–3, with one penicillate midlength seta ventrally; ischium with two setae; merus one third of basis length, with two finely denticulate spines and microtrichia ventrodistally; carpus 1.3 times longer than merus, with one complex-tip seta and one finely denticulate spine dorsodistally, two finely denticulate spines ventrodistally and some ventral microtrichia; propodus just longer than carpus, with one finely denticulate spine dorsodistally, two finely denticulate spines ventrodistally and ventral microtrichia; dactylus and unguis combined 1.3 times longer than propodus, both denticulate ventrally; unguis about half as long as dactylus.
Pereopod-5 (Fig. 8F) as pereopod-4.
Pereopod-6 (Fig. 8G) as pereopod-4, but propodus with three dorsodistal finely denticulate setae.
Pleopod (Fig. 8H, H') basis shorter than ramus, with ventrodistal seta; endopod with 12 setae along ventral and distal margins; exopod with eight distal setae. All setae plumose.
Uropod (Figs 8I, 9D) basis 0.8 times as long as exopod, naked; exopod of two segments, 0.7 times as long as endopod segment-1, segments subequal; segment-1 with short seta; segment-2 with one short and one long setae; endopod of two equal segments; segment-1 with one simple and three penicillate setae distally; segment-2 with one long midlength seta and one penicillate and four simple setae distally.
Manca-3. Length 1.03–1.05 mm (n = 3).
Ovigerous female. As female (described above) but more dorsoventrally compressed, with oostegites; length 1.93–2.51 mm (n = 10).
Post-ovigerous female. As female (described above) but dorsoventrally compressed, lacking oostegites, but with scars or remnants thereof; length 2.15–2.35 mm (n = 5).
Preparatory male. As female (described above) but with proportionately larger antennules, with stouter articles 1–2; length 1.79–3.00 mm (n = 6). Body (Fig. 6B) slender, 10.3 times longer than wide, 2.8 mm long. Cephalothorax 14% of total body length, about 1.5 times longer than wide, carapace with fine seta at each anterolateral corner, eyelobes poorly marked. Pereonites parallel-sided, length/width ratio: 0.8, 1.1, 1.1, 1.0, 0.9, and 0.6. Pleon 18% of total body length, pleonites subequal in length, epimera with single seta. Pleotelson as long as pleonites 3–5 and half of pleonite-2, with rounded apex, terminated with two pairs of setae.
Antennule (Fig. 6B, C) about 0.75 times as long as cephalothorax; article-1, twice as long as wide, with one simple and five penicillate setae on outer distal margin; article-2 0.6 times as long as article-1, with one long and three penicillate outer distal setae; article-3 0.7 times as long as article-2, with one short and one longer inner distal setae; article-4 just shorter than article-2, with four simple setae distally and one aesthetasc.
Remarks. Parakanthophoreus catharina n. sp. differs from other members of the genus by a combination of: elongated body (about ten times longer than wide), maxilliped basis with seta, maxilliped endites with a pair of distal setae, cheliped carpus, propodus, and dactylus without crenulation, but carpus with deep shield, pereopods 4–6 ischium with pair of setae and dactylus with denticulate ventral margins, uropod exopod distinctly longer than half of endopod segment-1. Another useful characteristic distinguishing P. catharina from its congeners, but also from other akanthophoreids, is the setation of the cheliped fixed finger, where the two ventral setae are of unequal thickness.
Of the described species of Parakanthophoreus known from the general area of the IceAGE project—P. alba (Hansen, 1913), P. brachiatus (Hansen, 1913), P. inermis (Hansen, 1913), P. longiremis (Lilljeborg, 1864), P. multiserratus (Hansen, 1913), and P. vikingra (Błażewicz-Paszkowycz & Bamber, 2011)—the most likely species to be confused with P. catharina is P. inermis, since it can be sympatric, and most of the non-IceAGE records of P. catharina were originally assigned, provisionally, to that species. A review and comparison of these species (except P. vikingra) was given by Guerrero-Kommritz (2004), while Hansen (1913) is still a good reference for species comparisons.
Parakanthophoreus inermis was characterized by Hansen (1913) by having a relatively short antennule article-1 compared to other similar species, as well as a deep cheliped carpal shield and a short claw on pereopods 1–3, all of which are shared by P. catharina but not by the other Parakanthophoreus species mentioned above. Redescriptions of P. inermis by Guerrero-Kommritz (2004) and Sieg (1986b) were unhelpful in clarifying the nature of this species because of several factors such as unclear attribution of material, poor illustrations, or use of non-topotypical specimens. Indeed, the taxon dealt with by Sieg may be conspecific with, or at least clearly related to, P. catharina rather than P. inermis and it is possible that some of Hansen’s material may too be of both species. A revision of P. inermis is in progress (Bird, in prep.).
Examination of near-topotypical specimens of Parakanthophoreus inermis (Fig. 11) indicates that it can be separated from P. catharina by, inter alia, its shorter cephalothorax, shorter pereonites (especially pereonite-1, Fig. 11A)–none longer than wide, weaker (but still relatively deep) cheliped carpal shield (Fig. 11B), lack of a distodorsal seta on the carpus of pereopods 1–3 (Fig. 11C; very conspicuous in P. catharina), and only two dorsodistal spines on the propodus of pereopod-6 (Fig. 11D; three in P. catharina); the cuticular ornamentation is also less pronounced, without the pitting seen in P. catharina. Other akanthophoreids in the Iceland-Faroe region with large cheliped carpal shields include Chauliopleona hastata (Hansen, 1913) and Saurotipleona julii Bird, 2015 but these can be distinguished, at least, by their ventral spur on pleonite-5.
Distribution: BIOICE records; 23, from the Greenland-Iceland Rise, Iceland Plateau, Iceland-Faroes Rise (all but one from northern flank), Kolbeinsey Ridge, and Norwegian Basin, 326–1994 m, in temperatures < 1 °C. Other records, 53 records from the Faroe-Shetland Channel, Faroe-Norway Rise, North Shetland Slope, and Norwegian Basin, 434–1513 m (Table 1; Bird 2001 and ined.) (see Fig. 1).
References
Bird GJ (2001) Tanaidacea of the Atlantic Margin: the AFEN surveys of 1996 and 1998. Unpublished report to the Atlantic Frontier Environmental Network and UK Offshore Oil Association, February 2001
Bird GJ (2007) Family incertae cedis [sic]. In: Larsen K, Shimomura M (eds) Tanaidacea (Crustacea: Peracarida) from Japan III. The deep trenches: the Kurile-Kamchatka Trench and Japan Trench. Magnolia Press, Auckland, pp 121–149
Bird GJ (2015) Tanaidacea (Crustacea: Peracarida) of the Northeast Atlantic: Chauliopleona Dojiri and Sieg, 1997 and Saurotipleona n. gen. from the ‘Atlantic Margin’. J Nat Hist 49:1507–1547. https://doi.org/10.1080/00222933.2015.1005715
Błażewicz-Paszkowycz M, Bamber R (2011) Tanaidomorph Tanaidacea (Crustacea: Peracarida) from mud-volcano and seep sites on the Norwegian margin. Zootaxa 3061:1–35
Błażewicz-Paszkowycz M, Bamber RN, Cunha MR (2011) New tanaidomorph Tanaidacea (Crustacea: Peracarida) from submarine mud-volcanoes in the Gulf of Cadiz (North-East Atlantic). Zootaxa 2769:1–53
Błażewicz-Paszkowycz M, Bamber RN, Jóźwiak P (2013) Tanaidaceans (Crustacea: Peracarida) from the SoJaBio joint expedition in slope and deeper waters in the Sea of Japan. Deep-Sea Res II 111:325–332. https://doi.org/10.1016/j.dsr2.2014.08.021
Brix S, Bauernfeind W, Brenke N, Blazewicz M, Borges V, Buldt K, Cannon J, Díaz Agras G, Fiege D, Fiorentino D, Haraldsdóttir S, Hoffmann S, Holst S, Hüttmann F, Jeskulke K, Jennings R, Kocot K, Khodami S, Lucas Rodriguez Y, Martinez Arbizu P, Meißner K, Mikkelsen N, Miller M, Murray A, Neumann H, Ostmann A, Riehl T, Schnurr S, Savavarsson J, Yasuhara M (2011) Cruise Report M85/3 IceAGE1 2011 with RV Meteor. Reykjavik, Cuxhaven
Brix S, Meißner K, Stransky B, Halanych KM, Jennings RM, Kocot KM, Svavarsson J (2014) The IceAGE project a follow up of BIOICE. Pol Polar Res 35:141–150. https://doi.org/10.2478/popore-2014-0010
Coleman CO (2003) Digital inking: how to make perfect line drawings on computers. Org Divers Evol 14:1–14
Dojiri M, Sieg J (1997) The Tanaidacea. In: Blake JA, Scott PH (eds) Taxonomic atlas of the benthic fauna of the Santa Maria Basin and Western Santa Barbara Channel. Volume 11. The Crustacea Part 2. The Isopoda, Cumacea and Tanaidacea. Santa Barbara Museum of Natural History, Santa Barbara, CA, pp 181–278
Guerrero-Kommritz J (2004) A revision of the genus Paraleptognathia Kudinova-Pasternak, 1981 (Crustacea: Tanaidacea) and description of four new species. Zootaxa 481:1–63
Hansen HJ (1913) Crustacea, Malacostraca. II. IV. The order Tanaidacea. Danish Ingolf Expedit 3:1–145
Kudinova-Pasternak RK (1981) Tanaidacea. In: Kusnecov AP, Mironov AN (eds) Bentos podvodnych gor Markus-Nekker i smeznych rajonov tichogo okeana [Benthos of the Submarine Mountains Markus Necker and adjacent Pacific regions]. Institut Okeanologii, Moscow, pp 94–112
Kudinova-Pasternak RK (1985) Tanaidacea (Crustacea, Malacostraca) collected on the summit and at foot of Great-Meteor Seamount. T I Ocean 120:52–64
Larsen K (2005) Deep-sea Tanaidacea (Peracarida) from the Gulf of Mexico. Brill, Leiden
Larsen K, Araújo-Silva CL (2014) The ANDEEP Tanaidacea (Crustacea: Peracarida) revisited III: the family Akanthophoreidae. Zootaxa 3796:237–264. https://doi.org/10.11646/zootaxa.3796.2.2
Larsen K, Błażewicz-Paszkowycz M, Cunha MR (2006) Tanaidacean (Crustacea: Peracarida) fauna from chemically reduced habitats—the lucky strike hydrothermal vent system, mid-Atlantic [sic] ridge. Zootaxa 1187:1–36
Larsen K, Wilson GDF (2002) Tanaidacean phylogeny, the first step: the superfamily Paratanaidoidea. J Zool Syst Evol Res 40:205–222
Sieg J (1986a) Tanaidacea (Crustacea) von der Antarktis und Subantarktis. II. Tanaidacea gesammelt von Dr. JW. Wägele wahrend der Deutschen Antarktis Expedition 1983. Zoologischen Museum der Universität Kiel 2:1–80
Sieg J (1986b) Crustacea Tanaidacea of the Antarctic and the Subantarctic. 1. On material collected at Tierra del Fuego, Isla de los Estados, and the west coast of the Antarctic Peninsula. In: Korniker LS (ed.) Biology of the Antarctic Seas 18. Volume 45 in the Antarctic Research Series. American Geophysical Union, Washington, D.C., pp 1–180
Acknowledgements
Authors would like to thank Saskia Brix for making the material available for study and the crew of R/V Meteor and R/V Poseidon as well as the scientists who participated in IceAGE Program. We are also grateful for two anonymous reviewers for their valuable comments. Studies were financed by Polish National Science Centre grant UMO-2014/13/B/NZ8/04702.
Funding
This study was funded by the Polish National Science Centre grant (UMO-2014/13/B/NZ8/04702).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed by the authors.
Field study
Permits and approval of field or observational studies are not applicable for authors.
Additional information
Communicated by S. Brix
This article is registered in ZooBank under: urn:lsid:zoobank.org:pub:6D7D4138-0170-44BD-BCD9-48B9983B88E1
This article is part of the Topical Collection on Biodiversity of Icelandic Waters by Karin Meißner, Saskia Brix, Ken M. Halanych, and Anna Jazdzewska.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Jóźwiak, P., Drumm, D.T., Bird, G.J. et al. A new genus of family Akanthophoreidae and new species of genus Parakanthophoreus Larsen & Araújo-Silva, 2014 (Crustacea: Tanaidacea: Tanaidomorpha) from the North Atlantic. Mar Biodiv 48, 897–914 (2018). https://doi.org/10.1007/s12526-018-0866-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12526-018-0866-9