Abstract
Plant virus-like particles (VLPs) structurally resemble their progenitor viruses, but are noninfectious due to absence of viral nucleic acids. Since the 1980s, VLPs have been actively studied with the aim of constructing different nanomaterials, including immunologically active carriers for peptides and whole proteins and proteinaceous shells for the packaging of different ligands.
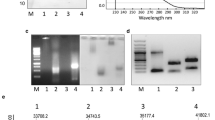
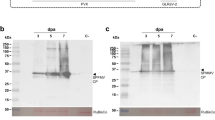
The technological developments using VLPs require large amounts of purified particles. Here, we describe the laboratory process for isolation and purification of two unmodified plant VLPs, derived from two sobemoviruses, cocksfoot mottle virus (CfMV) and rice yellow mottle virus (RYMV), which is based on cultivation of recombinant Escherichia coli cells, VLP precipitation from bacterial extracts and ultracentrifugation. The suggested purification scheme allows the production of 4–45 mg of purified sobemoviral VLPs from a 1 l bacterial culture, depending on the required purity level. Additionally, we provide short protocols for VLP characterization using SDS-PAGE, agarose gel electrophoresis, ultraviolet and mass spectrometry, dynamic light scattering, and electron microscopy.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Zeltins A (2016) Viral nanoparticles: principles of construction and characterization. In: Khudyakov YE, Pumpens P (eds) Viral nanotechnology. CRC Press, Boca Raton, pp 93–119
Haynes JR, Cunningham J, von Seefried A et al (1986) Development of a genetically-engineered, candidate polio vaccine employing the self-assembling properties of the tobacco mosaic virus coat protein. Biotechnology 4:637–641
Zeltins A (2013) Construction and characterization of virus-like particles: a review. Mol Biotechnol 53:92–107
Clinical Trials Identifier: NCT02188810 (2014) Safety and reactogenicity of a PAL combined with seasonal flu vaccine in healthy adults. https://clinicaltrials.gov/ct2/show/NCT02188810. Accessed 3 Apr 2017
Clinical Trials Identifier: NCT02013687 (2013) Safety and immunogenicity of plant-derived Pfs25 VLP-FhCMB malaria transmission blocking vaccine in healthy adults. https://clinicaltrials.gov/ct2/show/NCT02013687. Accessed 3 Apr 2017
Studier FW, Rosenberg AH, Dunn JJ, Dubendorff JW (1990) Use of T7 RNA polymerase to direct expression of cloned genes. Methods Enzymol 185:60–89
Kalnciema I, Balke I, Skrastina D et al (2015) Potato virus M-like nanoparticles: construction and characterization. Mol Biotechnol 57:982–992
ExPASy bioinformatics resource portal. Peptide mass. http://web.expasy.org/peptide_mass/. Accessed 3 Apr 2017
Qubit® 2.0 Fluorometer https://tools.thermofisher.com/content/sfs/manuals/mp32866.pdf Accessed 3 Apr 2017
Kalnciema I, Skrastina D, Ose V et al (2012) Potato virus Y-like particles as a new carrier for the presentation of foreign protein stretches. Mol Biotechnol 52:129–139
Tars K, Zeltins A, Liljas L (2003) The three-dimensional structure of cocksfoot mottle virus at 2.7 A resolution. Virology 310:287–297
Hesketh EL, Meshcheriakova Y, Dent KC et al (2015) Mechanisms of assembly and genome packaging in an RNA virus revealed by high-resolution cryo-EM. Nat Commun 6:10113
Cheng A, Speir JA, Yuan YA et al (2009) Preliminary X-ray data analysis of crystalline hibiscus chlorotic ringspot virus. Acta Crystallogr Sect F Struct Biol Cryst Commun 65:589–593
Hirel PH, Schmitter MJ, Dessen P et al (1989) Extent of N-terminal methionine excision from Escherichia coli proteins is governed by the side-chain length of the penultimate amino acid. Proc Natl Acad Sci U S A 86:8247–8251
Porterfield JZ, Zlotnick A (2010) A simple and general method for determining the protein and nucleic acid content of viruses by UV absorbance. Virology 407:281–288
Nikitin N, Trifonova E, Evtushenko E et al (2015) Comparative study of non-enveloped icosahedral viruses size. PLoS One 10(11):e0142415
Acknowledgment
We wish to thank Dr. V. Ose for help in the preparation of EM pictures and J. Bogans for DLS experiments. G. Grinberga and V. Zeltina are acknowledged for their technical assistance. The chapter was written with the help of Grant No. SP 672/2014 provided by the Latvian Science Council.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Balke, I., Reseviča, G., Zeltins, A. (2018). Isolation and Characterization of Two Distinct Types of Unmodified Spherical Plant Sobemovirus-Like Particles for Diagnostic and Technical Uses. In: Wege, C., Lomonossoff, G. (eds) Virus-Derived Nanoparticles for Advanced Technologies. Methods in Molecular Biology, vol 1776. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7808-3_2
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7808-3_2
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7806-9
Online ISBN: 978-1-4939-7808-3
eBook Packages: Springer Protocols