Abstract
Proteases play an important role in health and disease of the lung. In the normal lungs, proteases maintain their homeostatic functions that regulate processes like its regeneration and repair. Dysregulation of proteases–antiproteases balance is crucial in the manifestation of different types of lung diseases. Chronic inflammatory lung pathologies are associated with a marked increase in protease activities. Thus, in addition to protease activities, inhibition of anti-proteolytic control mechanisms are also important for effective microbial infection and inflammation in the lung. Herein, we briefly summarize the role of different proteases and to some extent antiproteases in regulating a variety of lung diseases.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The lung possesses a large number of anti-inflammatory components [1], which fight against microbial infections.
Serine, cysteine, aspartic, and metalloproteases are the principal classes of protease present in the human lung. A good number of evidence suggest that neutrophil serine proteases (NSPs) such as elastase, proteinase 3 (PR3), cathepsin G (CatG), and matrix metalloproteases (MMPs) are major pathogenic determinants of chronic inflammatory lung disorders [1]. The lung proteases act in concert with the proteases of invading microbes, inactivate antiproteases, and antimicrobial compounds and thereby play a pivotal role in different types of lung diseases including chronic obstructive pulmonary disease (COPD), asthma, acute respiratory distress syndrome (ARDS), influenza, and cancer [1].
The lung proteases can either intracellularly or extracellularly regulate processes such as tissue remodeling, mucin production, neutrophil chemotaxis, and microbial destruction. Additionally, they regulate infection and inflammation in the lung, for example neutrophil elastase (NE), a serine protease, which plays critical role in the progression of a variety of lung diseases. It can regulate activities of CatB and MMP-2 in alveolar macrophages [2] and also activates proMMP-2, MMP-7, and MMP-9 [3,4,5], indicating that NE may act as a proinflammatory mediator. In some cases, NE regulates important signaling pathways that modulate innate immunity [6, 7]. NE’s multiple roles characterize it as a decisive factor controlling many aspects of infection and inflammation in the lung.
2 Pulmonary Hypertension
Pulmonary hypertension (PAH) occurs due to elevation of pulmonary artery pressure and if it is prolonged, then right ventricular failure may occur with subsequent fatality [8]. PAH often leads to secondary complications of many pulmonary disorders such as COPD, asthma and chronic bronchitis bronchopulmonary displasia, cystic fibrosis, chronic bronchitis, and emphysema [9, 10].
2.1 Serine Protease and Pulmonary Hypertension
The role of oxidants such as hydrogen peroxide, hydroperoxides, superoxide, and peroxynitrite in producing PAH is now well established [11,12,13,14,15]. Administration of the oxidant, tert-butylhydroperoxide (tert-buOOH) to the perfusate of isolated rabbit lungs causes pulmonary vasospasm [16]. Oxidant-induced pulmonary vasoconstriction can be blocked by the cyclooxygenase or thromboxane synthase inhibitor, indomethacin and is closely correlated with the thromboxane level in the effluent perfusate [13, 16,17,18,19], suggesting a critical role of thromboxane in pulmonary vasoconstriction. On the other hand, TMB-8, an intracellular Ca2+ ([Ca2+] i ) antagonists, has been shown to prevent oxidant-mediated pulmonary vasoconstriction [13]. Thus, oxidant-mediated PAH is triggered by an increase in [Ca2+] i . In many occasions PAH occur due to an increase in [Ca2+] i caused by stimulants such as thromboxane A2 and endothelin-1 that generates oxidants. Oxidant and Ca2+ ionophore-mediated pulmonary hypertension has been observed to be inhibited by serine protease inhibitors, for example, aprotinin [19,20,21].
The mechanism by which oxidants stimulate production of the arachidonic acid (AA) metabolites has gained considerable interest. A report by Chakraborti et al. [22] indicated that oxidants, e.g., tert-buOOH stimulation of pulmonary artery endothelial and smooth muscle cells caused a marked increase in phospholipase A2 (PLA2) activity with subsequent generation of AA. Mepacrine, an inhibitor of PLA2 inhibits tert-buOOH-induced increase in PLA2 activity, thromboxane B2 production, and PAH [19, 22]. Some investigators, considering the analogy of activation of pancreatic phospholipase A2 [23], suggested that a serine protease might be involved in regulating PLA2 activity [24]. Chakraborti et al. [25, 26] demonstrated that oxidant-mediated activation of PLA2 activity in pulmonary endothelial and smooth muscle cells occur with the involvement of proteolytically activated protein kinase Cα (PKCα). They have also demonstrated that oxidants caused increase in [Ca2+] i in pulmonary endothelial and smooth muscle cells can activate an aprotinin sensitive protease having mol mass of ~43 kDa [26]. The protease then proteolytically activates PKCα resulting in stimulation of cPLA2 (Fig. 1), which generates thromboxane and that has been observed to be important in producing PAH [25, 26]. Oxidants elicit an increase in [Ca2+] i due to proteolytic activation of PKC-δ by MMP-2 resulting in phosphorylation of a pertussis toxin sensitive protein (Gi) leading to inhibition of Na+ dependent Ca2+ uptake (Na+/Ca2+ exchanger) in the endoplasmic reticulum (ER) [27,28,29,30,31] (Fig. 2), whereas the role of cell membrane for an increase in [Ca2+] i has been observed to be due to phosphorylation of G i via proteolytically activated PKCα by an aprotinin sensitive serine protease leading to inhibition of Na+ dependent Ca2+ efflux (Na+/Ca2+ exchanger) (Fig. 1) in pulmonary vascular cells [24,25,26].
Schematic representation of the underlying mechanism associated with oxidant (reactive oxygen species: ROS)-mediated cPLA2 activation in pulmonary vascular endothelial and smooth muscle cells. A calcium channels; B membrane bound Ca2+ stores; C anion channels; D diffusion; E inhibition of Na+ dependent Ca2+ uptake
Schematic representation of the underlying mechanism of oxidant (reactive oxygen species: ROS) triggered inhibition of Na+ dependent Ca2+ uptake in bovine pulmonary smooth muscle ER resulting in an increase in [Ca2+] i . A Anion channel; B diffusion; MMP-2 matrix metalloprotease-2; TIMP-2 tissue inhibitor of metalloprotease-2; PKCδ protein kinase C delta; RACK-1 receptor for activated C kinase-1; Giα inhibitory G protein α subunit; GiαP phosphorylated Giα; C Na+-K+-ATPase/Na+-H+ exchanger. (Taken from Chakraborti et al. (2005) Mol Cell Biochem. 280: 107–117 with permission)
In many systems, Ca2+-ATPase represents only about 1% of the total proteins [32]. In heart sarcolemmal vesicles, Ca2+ uptake via NCX produces maximum transport velocity and that has been demonstrated to be about 30-fold up than that elicited by the sarcoplasmic reticulum {S(ER)} Ca2+ pump system [33]. In addition to the Ca2+ pump, Na+ dependent Ca2+ uptake system is an important mechanism to sequester Ca2+ in the ER of pulmonary vascular cells [29,30,31,32,33]. A decrease in Ca2+ sequestration by proteolytic inhibition of Na+ dependent Ca2+ uptake has been observed to measure duration of free [Ca2+] i transient, which eventually produces vasoconstriction [34, 35]. In different systems, Na+/Ca2+ exchanger controls the contractility of smooth muscle cells [34, 35]. For example, contractile dysregulation in the myocardium could be related with activation of proteases [36]. Thus, the role of cell membrane associated aprotinin sensitive protease and ER MMP-2 on Na+/Ca2+ exchange in pulmonary artery endothelial and smooth muscle cells under oxidant triggered condition is an important mechanism for the pathological manifestation of pulmonary vasoconstriction [24,25,26, 29,30,31].
2.2 MMPs and Pulmonary Hypertension
PAH is characterized by persistent vasoconstriction and remodeling of pulmonary vasculature associated with activation of proteases, for instance, MMPs [34]. Remodeling of pulmonary artery is associated with an alteration of extracellular matrix (ECM) turnover with concomitant change in ECM proteins level. In PAH, dysregulation of ECM turnover has been suggested to play an important role in the pathological remodeling process [35, 36]. ECM degradation occurs by different proteases of which matrix metalloproteases (MMPs) has been shown to play the crucial role [37, 38]. Of the MMPs, MMP-2, and MMP-9 are able to cleave basement membrane associated type IV collagen, which increase remodeling of the pulmonary vasculature in PAH [39]. Given the potency of MMPs, its activity is tightly regulated at the transcriptional and post-translational level, where the tissue inhibitors of MMP (TIMPs) play a pivotal role [40, 41].
IL-1, a potent endogenously generated inducer of PAH, elicits its effect via an increase in the level of TGF and TNF in pulmonary smooth muscle cells. TGF causes an increase in the expression of the 92 kDa proMMP-9 and 72 kDa proMMP-2 mRNAs, while TNF triggers activation of proMMP-9 and proMMP-2 [42].
MMP-2 is produced upon activation of proMMP-2 by a variety of stimuli under different pathophysiological conditions. It has been observed that the activation of proMMP-2 occurs at the cell membrane. Interaction between MT1-MMP and TIMP-2 is an important phenomenon in the activation of proMMP-2. The MT1MMP-TIMP2 associates with proMMP-2 and forms a trimolecular complex, which triggers the activation of proMMP-2 and subsequently generates MMP-2 [41, 42]. The activation of proMMP-2 in pulmonary artery smooth muscle cells occur with the involvement of protein kinase C-α dependent and NF-кB-MT1MMP-mediated signaling mechanism. TNF-α augments mRNA and protein expression of MT1MMP, while the expression level of TIMP-2 diminishes. The increase in TNF-α leads to IKK activation, IB phosphorylation and degradation, and subsequently activation of NF-кB. Upon activation, NF-кB binds to the MT1-MMP promoter, thereby enhancing its expression and subsequently increases proMMP-2 level in association with TIMP-2 that is modulated by protein kinase C-α at the cell membrane [41, 42] (Fig. 3). This indicates therapeutic potentiality of PKC inhibitors in ameliorating the PAH, where activation of proMMP-2 is an important phenomenon.
Schematic representation of TNFα-induced proMMP-2 activation in the SMCs. TNFα binds to cell surface receptor TNFR1. Upon binding, TNFα induces PKCα activation, which subsequently activates IKK by phosphorylation. Activated IKK then phosphorylates IkB-α, which upon phosphorylation is ubiquitinated and degraded in the cytosol. The free NF-кB then translocates to the nucleus and increases the expression of MT1-MMP, which then accumulates on the cell surface. PKC-α, on the other hand, also down regulates TIMP-2 expression by mechanism that is currently unknown. (Taken from Roy et al. (2013) J Biochem 153:289–302 with permission)
ProMMP-9 activation by TNF-α has been observed to occur with the involvement of an aprotinin sensitive serine protease [42]. TNF-α was shown to inhibit aprotinin and TIMP-1 mRNA and protein expression, which trigger activation of proMMP-2 resulting in the stimulation of MMP-2 (Fig. 4). Under IL-1β stimulation, the aprotinin sensitive protease was not activated, although a discernible inhibition of TIMP-2 mRNA and protein expression were triggered by TNF-α [42]. Thus, IL-1-induced stimulation of the two progelatinases occurs via different mechanisms.
Schematic representation of proMMP-9 activation by an aprotinin sensitive protease during IL-1β stimulation of pulmonary artery smooth muscle cells. IL-1β treatment to the cells stimulates TGF-β and TNF-α. TGF-β stimulates expression of proMMP-9, while TNF-α activates proMMP-9 via an increase in a ~43 kDa aprotinin sensitive protease concomitant with the down regulation of aprotinin and TIMP-1 expression
3 Influenza
Influenza viruses are highly infectious and trigger acute respiratory diseases with significant morbidity and mortality in humans and other animals [43,44,45,46].
Influenza viruses can be classified as A, B, or C. Influenza virus A, found in humans and other mammals and birds, played a nefarious role in causing the three twentieth century major influenza outbreak and also the influenza outbreak of swine origin that occurred in the recent past [47]. Many of the influenza A-related mortalities are attributable to secondary bacterial pneumonia [48, 49].
Haemagglutinin (HA) protein contributes critically to influenza virus-mediated pathogenicity. HA of influenza virus binds to sialic acid containing cell surface receptors. HA upon cleavage by a good number of host of protease(s) forms HA1 and HA2 subunits and that has been fused with host cell membrane, which subsequently initiates the infection process [49,50,51,52]. In most cases, the cleavage site of HA of avian and mammalian influenza viruses is a single arginine, albeit a single lysine amino acid has also been observed at the cleavage site in some cases. Cleavage can occur extracellularly by trypsin [53, 54] and proteases such as plasmin [55,56,57], tryptase of bronchiolar epithelial and mast cells [58], and also by bacterial proteases [59,60,61].
Several other proteases expressed in the lung are also able to facilitate influenza virus spread. Böttcher et al. [62] demonstrated that TMPRSS2 and TMPRSS11D, transmembrane serine proteases, (a.k.a. human airway trypsin-like protease: HAT) activate the influenza viruses H1N1 (A/Memphis/14/96), H2N9 (A/Mallard/Alberta/205/98), and H3N2 (A/Texas/6/96) upon cleavage of haemagglutinin (HA) and contribute to the high pathogenicity of these influenza viruses in the lung [63]. In addition to HAT, TMPRSS-2 and -4, the granzymes (Gzm) such as GzmA, GzmB, and GzmE are known to play a key role in the process of cleavage of 1918 H1N1 HA as a part of the progression of the influenza disease [62, 63].
The sites of virus replication in the microenvironments of respiratory tract represent complex extracellular proteases (such as trypsin and tryptase), which activate a family of receptors called protease activated receptors (PARs) [64, 65] and that play an important role in both virus replication and innate immune response [58, 66]. Four PARs (PAR1-4) are known to be activated by different proteases. After cleavage of the receptor(s) by proteases, the newly released amino terminal sequence binds and internally activates the receptor [67]. In the airways of IAV-infected mice, an increase in the level PAR2 upon IFNγ-mediated modulation has been shown to play a crucial role in influenza pathology [68, 69].
Multiple serine protease activities are implicated in mediating influenza virus infection. Inhibition of influenza A virus infection in cultured lung epithelial cells by serine protease inhibitors, for example, aprotinin markedly protects mice from infection [70]. Another serine protease inhibitor, camostat has also been shown to possess anti-influenza (Taiwan/1/86) virus pathology [71].
4 Chronic Obstructive Pulmonary Disease
Chronic obstructive pulmonary disease (COPD) is associated with the pathological manifestations of emphysema and chronic bronchitis. Emphysema is characterized by a marked destruction of the alveolar septa with concomitant decrease in lung plasticity and that results in gas trapping leading to a marked decrease in pulmonary oxygenation in the lung. Chronic bronchitis usually occurs with inflamed and thickened airways along with an increase in mucus production by the cells in the airways, which leads to a marked increase in cough and difficulty in breathing. Noxious particles present in cigarette smoke and automobile exhaustion have been observed to be an important causative agent of COPD [72, 73].
In the early 1960s, proteases have been shown to produce lung lesions in experimental animals similar to human emphysema. Initial studies in this scenario included metalloproteinases, papain and subsequently serine proteases, for example, porcine pancreatic elastase [74].
4.1 Serine Proteases and COPD
The identification of α1-PI (an endogenous serine protease inhibitor) and subsequent research confirmed that a strong association exists between the development of emphysema and inherited deficiency of the inhibitor [75]. A critical role of serine proteases have been established in the pathobiology of emphysema. Preliminary studies in this genetic condition of inherited α1-PI deficiency indicated that chronic bronchitis is associated with the early onset of the disease [76, 77].
The lung epithelium of normal individuals is protected from the detrimental effects of neutrophil serine proteases (NSPs) by a battery of antiproteases. Large quantities of NSPs (~20-fold w.r.t. normal subjects) released by neutrophils in acute and chronic inflammatory conditions overpowered antiprotease activities, leading to uncontrolled proteolysis and subsequently lung damage [74,75,76].
Alpha-1-PI deficiency increases activities of different enzymes secreted by activated neutrophils such as NE, cathepsin G (CatG), and proteinase 3 (PR3), all of which are capable of damaging different components of the ECM such as collagen, laminin, fibrillin, and elastin. However, several evidences suggest that it is the destruction of lung elastin that is important in causing emphysema, which generates COPD pathophysiology in animal model systems [75, 76].
Human neutrophil elastase (HNE), by destroying elastin, plays a critical role in the development of pulmonary emphysema [76, 77]. HNE has many other biological activities. For example, it stimulates mucin production [78], activates MMPs [5], inactivates TIMPs [79], and generates neutrophil chemotactic elastin-derived fragments [80, 81]. PR3 and CatG, two elastase homologues secreted in massive quantities from neutrophils at inflammatory sites, have also been shown to have proinflammatory functions acting through various mechanisms [80,81,82]. The most abundant is the α1-PI, which targets preferentially HNE. Secretory leukoprotease inhibitor (SLPI), an inhibitor of HNE and CatG, but not of PR3, has been shown to control excess proteolysis in the upper airways [83]. Elafin, derived from trappin-2 (pre-elafin) [84], is an NSP inhibitor that controls the activities of HNE and PR3 [85]. Other NSP inhibitors including 1-antichymotrypsin and monocyte/NE inhibitor (MNEI) were found to play relatively minor role as protease inhibitors [86, 87]. NSPs, therefore, could prove useful as therapeutic target for a number of inflammatory lung diseases.
There are differences in pathological manifestations among smoking and non-smoking α1-PI deficient COPD patients. The smokers with COPD frequently show pulmonary emphysema and bronchitis, while patients of the latter category often show emphysema without bronchitis. Although the chronic smokers usually suffer from the antiprotease inactivation in both the airways and respiratory units as a result of oxidant-mediated inactivation of α1-PI, the non-smoking α1-PI deficient individuals possess diminished antiprotease content primarily in respiring units, which are free of mucus glands and depends upon α1-PI for antiprotease defence [88].
Oxidative stress induced by cigarette smoke in COPD patients may promote the inflammatory state by recruiting additional neutrophils and upregulating the inflammatory transcription factor, NF-кB and neutralizing TIMPs in addition to α1-PI and SLPI [89].
4.2 Urokinase Plasminogen Activator and COPD
In COPD patients, an increase in urokinase plasminogen activator (uPA) level in the airway epithelial and alveolar cells, and lung macrophages cause destruction of small airways and alveolus of the lung [90]. Urokinase plasminogen activator receptor (uPAR), in addition to functioning as a protease receptor, mediates intracellular signaling [91]. An increase in uPAR level in the macrophages has been observed in patients with COPD, which suggests the critical role of uPAR in inflammation and tissue remodeling including parenchymal destruction and fibrosis of small airways [92].
4.3 MMPs and COPD
MMPs are known to induce morphological changes in the lung that are prevalent in COPD. Several MMPs are known to play important roles in the pathogenesis of COPD [93]. Lung parenchyma and inflammatory cells such as neutrophils and macrophages are the major sources of MMPs in patients with COPD [94].
MMP-12 (a.k.a. macrophage elastase) is known to play an important role in COPD pathogenesis. A marked increase in MMP-12 expression in alveolar macrophages is associated with smoking associated emphysema [95]. In mouse, deletion of MMP-12 gene prevents cigarette smoke-induced inflammation, neutrophil influx, and emphysema in the lung [96, 97]. Genetic analysis of human COPD patients demonstrated that the common serine (codon 357) of the MMP-12 gene plays a crucial role in the pathological manifestations of matrix degradation, which has been observed to be related with the severity of the disease [98, 99].
Analysis of COPD lung tissue indicated an increase in the activity of MMP-1 and MMP-8, but not MMP-13 [100, 101]. An increase in MMP-1 activity was found in type II pneumocytes in patients with emphysema, but not in normal control subjects [101]. Neutrophil-derived MMP-8 levels were markedly increased in patients with COPD in comparison to the normal subjects [101]. Prominent increase in MMP-2 and MMP-9 expression has been observed in the lung of COPD patients [102]. During interleukin-10 (IL-10)-mediated airflow obstruction, an imbalance between MMP-9 and TIMP-1 results in an increase in MMP-9 activity was found in an animal model system [103].
Acrolein, a component of cigarette smoke‚ has been shown to initiate cleavage of proMMP-9, thereby producing active MMP-9. However, MMP-9 knockout mice do not completely inhibit cigarette smoke-induced emphysema, suggesting that other MMPs also play role in COPD pathogenesis [103]. Importantly, MMP-14, the membrane-type MMP (MT1-MMP), has been observed to be induced by acrolein which upon increase in mucin production leads to COPD [104] (Fig. 5).
5 Lung Fibrosis
Lung fibrosis (a.k.a. interstitial lung diseases) is a chronic disorder, exemplified by a marked increase in matrix degradation and intra-alveolar fibrosis leading to dyspenea, impaired oxygen transfer and alveolar collapse [105, 106]. Lung fibrosis occurs in the alveolar space and interstitium and is characterized by a widespread accumulation of differentiated fibroblasts (i.e., myofibroblasts) and ECM components.
Fibrotic disorders in the lung are associated with dysregulation of proteolytic activities. A considerable number of reports have suggested the involvement of cathepsins in this scenario. Enhanced proteolytic processing of CatB was observed in the lungs during an increase in TGF-β, suggesting that CatB may participate in fibrogenesis [107].
Microarray studies have revealed that in addition to NE, MMP-7 (a.k.a. matrilysin) is an important COPD marker. MMP-7 degrades decorin, the extracellular proteoglycan, which subsequently releases decorin-bound transforming growth factor-β (TGF-β) [108] and that subsequently contributes to TGF-β activation, which is known as a critical marker of COPD [109].
6 Silicosis
An increase in the activity of MMP-2, MMP-9, and stromelysin has been demonstrated in alveolar macrophages from silica-treated rats, which contribute to extracellular matrix (ECM) and basement membrane (BM) degradation [110]. Administration of silica particles to mice causes upregulation of cathepsin K (CatK) expression and activity in silicotic lung homogenates compared to control lungs. Lung fibroblasts and macrophages were known as the main CatK-producing cells. Expression of CatK is inversely correlated to the level of TGF-β1, suggesting a protective role of CatK during silicotic process [111]. Mature active CatB, -H, -K, -L, and -S were identified in the broncho alveolar lavage fluids (BALFs) of patients suffering from silicosis. Among them, CatH has been observed to be the most abundant aminopeptidase, while CatB and CatL were mostly found thiol dependant endoproteases. Importantly, an increase in cathepsins/inhibitors ratio has been shown to favor uncontrolled proteolysis during silicosis [110, 111].
7 Cystic Fibrosis
In cystic fibrosis (CF), a marked increase in the activities of proteases could damage the airway architecture and that contributes to progressive bronchiectasis, a condition where the bronchial tubes of lungs are permanently damaged and enlarged due to infection in the bronchi [112, 113]. CF is an autosomal recessive genetic disorder caused by loss of expression or functional mutations to the cystic fibrosis transmembrane conductance regulator (CFTR) [114, 115]. CF affects multiple organs, albeit the pathology associated with CF appears to be due to its effect on the respiratory system. Non-functional CFTR channels in CF patients prevent the regulation of chloride and sodium ions across epithelial membranes leading to an increase in dehydrated mucus secretions in the lungs [112,113,114,115,116].
The key immune cell mediators seen in CF patients are polymorphonuclear neutrophils [115]. Upon recruitment, activated neutrophils release a wide variety of proteases, which induce inflammatory response and subsequently tissue damage [115, 116].
The impairment of mucociliary clearance mainly revolves around the interactions between NE and mucins. Mucins are a family of highly glycosylated proteins produced by epithelial cells and are the main components of the mucus found clogging of the airways in CF patients [117, 118]. NE has been shown to regulate the mucins via activation of TNF—converting enzyme, which upregulates their expression via epidermal growth factor receptor (EGFR) pathway [118,119,120]. CF patients cannot efficiently clear mucus due to damage by proteases to the cilia structures in the lungs and, therefore, are highly susceptible to chronic bacterial infections [121,122,123].
MMPs were found to play a crucial role in CF pathogenesis [124]. MMP levels are increased in the BAL of CF individuals [125]. MMPs produce proline-glycine-proline (PGP), a neutrophil chemoattractant derived from extracellular matrix, which regulates the immune response during CF [126].
A marked increase in NE has been shown in CF lung, which causes airway remodeling by degrading ECM proteins such as elastin and fibronectin [127]. The resulting alteration of airway epithelial cell membrane by NE induces neutrophil-mediated inflammation upon increase in the expression of proinflammatory cytokines, for example, IL-8, which results in neutrophil-mediated inflammation by upregulating the proinflammatory MMPs, CatG, and PR3 leading to tissue damage in the CF lung [6].
8 Asthma and Allergy
8.1 Asthma
Asthma is a chronic inflammatory disease of the airways and its occurence and propagation is on the rise. The number of patients with asthma is estimated to attain a staggering figure of 100 million globally by 2025 [128]. Generally, asthma is triggered by the activation of adaptive immune response that upon inducing the lung triggers mucus production, increased IgE level, airway remodeling, and airway hyperactivity [129].
Manifestation of Asthma is characterized by acute inflammatory response and airway obstruction [130]. In both acute and chronic asthma, proinflammatory cells, including neutrophils, eosinophils, mast cells and macrophages enters into the lung tissue [131, 132]. These proinflammatory cells secrete a variety of extracellular proteases of which serine proteases and MMPs are important as these enzymes play prominent role in asthma pathogenesis [133,134,135,136,137,138,139].
Plasminogen can be converted to the active enzyme plasmin by tissue type plasminogen activator (PA) or urokinase-type PA (u-PA). Tissue PA and u-PA are associated with the dissolution of fibrin and also in the degradation of ECM components [140]. Plasminogen activator inhibitor-1 (PAI-1) is a major inhibitor of tissue type plasminogen activator and u-PA, and thereby contributes to matrix formation by preventing matrix degradation. Mast cells (MCs) in the airways of patients with asthma are crucial in initiating allergic inflammation [141]. MCs and bronchial epithelial cells (BECs) are the major source of plasminogen activator inhibitor (PAI-1). The interactions between the BECs and the MCs are important in maintaining persistent inflammation and structural changes in asthma [142].
IgE-mediated inflammation is well known for the pathogenesis of asthma. MCs-derived TGF-β upon cross-linking with IgE receptor enhances PAI-1 production in BECs. This increase in the production of PAI-1 has been suggested to play a critical role in the development of fibrosis that occurs adjacent to the epithelium [143]. Conceivably, drugs that inhibit activation of MCs, for example, by anti-IgE may prove useful in preventing airway remodeling in asthma.
8.2 Serine Protease and MMP in Asthma Pathophysiology
Proteolytic enzymes including NE and MMP-9 play important roles in tissue remodeling and repair in the airways [144]. The proteolytic enzymes levels are increased in asthma, which occurs due to an imbalance in the protease–antiprotease system.
In neutrophilic asthma, high levels of active NE and proMMP-9 were observed, whereas only a small amount of MMP-9 has been observed to be active. However, eosinophilic asthma was characterized with high level of active MMP-9 without free elastase. Thus, a differential profile of protease activity has been observed in asthma. A deficiency of antiproteases may explain the differential enzyme activity observed in eosinophilic and neutrophilic asthma. An increase in the level of MMP-9 bound to TIMP-1 in subjects with neutrophilic asthma in comparison to the eosinophilic asthma has been observed. This could explain about the presence of low level of active MMP-9 in neutrophilic asthma. This relative deficiency of TIMP-1 in subjects with eosinophilic asthma in comparison to neutrophilic asthma may explain about the high level of active MMP-9 that exists in the sputum of subjects in this group [144,145,146].
Alpha1-PI level has been observed to be increased in neutrophilic asthma, but its function was impaired leading to a marked increase in free elastase (NE) activity. Proteolytic inactivation of α1PI may lead to a form, which acts as an activator of neutrophils and that could result in superoxide (O2 . −) production [146]. However, role of proteolytic enzymes in specific inflammatory phenotypes of asthma is not clearly known. In contrast, IL-8 (a potent chemoattractant and an activator of neutrophils) plays an important role in eosinophilic asthma [6]. NE can induce production of IL-8 and its potency has been observed to be elevated upon proteolytic processing by MMP-9 [147].
Patients with persistent inflammatory asthma elicit more non-eosinophilic asthma progression than that of the eosinophilic asthma. These exacerbations are not prevented by corticosteroid treatment [148]. COPD and neutrophilic asthmatic patients generally show chronic airway inflammation associated with a marked airway neutrophilia, which are not discernibly responsive to inhaled corticosteroids [148, 149].
8.3 Cytokines and Asthma
Inflammation in asthma has been observed to be mediated by a specific subclass of T-lymphocytes referred primarily to Th2 lymphocytes, which causes inflammation and remodeling via secretion of specific cytokines [150].
Cytokines, for example, IL-11 play primary role in mediating asthma pathophysiology through its receptor (IL-11R). ADAM-10, a matrix metalloprotease, can release the IL-11R ectodomain upon cleavage of IL-11 receptor. Serine proteases such as NE and PR3 can also cleave the IL-11R. The resulting truncated soluble IL-11R (sIL-11R) activates the inflammatory cells. Thus, IL-11 signaling pathology proceeds upon proteolytic cleavage of its receptor [151].
An increase in the numbers of apoptotic airway epithelial cells in COPD has been observed to be associated with secondary necrosis [152, 153]. In severe asthma, conditions associated with increased airway neutrophilia, tissue damage and an increase in apoptosis of airway epithelial and smooth muscle cells have also been demonstrated [154]. Granzymes, a family of serine protease, have a repute to initiate immune-mediated cell death. Cytotoxic T cells and natural killer (NK) via granzyme-mediated pathway induces apoptosis of target cells, e.g., bronchial epithelial cells. Granzymes play critical roles in a variety of age-related chronic inflammatory diseases. There are five human granzymes identified so far in the lungs. These are granzyme A (GzmA-tryptase), granzyme B (GzmB-aspase), granzyme H (GzmH-chymase), granzyme K (GzmK-tryptase), and granzyme M (GzmM-metase). Granzymes, especially granzyme B and perforin, are stored in secretory granules of cytotoxic cells, and are released into the intercellular space following adhesion to the target cells. In presence of Ca2+, perforin pores in the cell membrane enable entry of granzyme B and subsequently induces caspase-dependent apoptosis [155], which may be an important mechanism of lung injury in asthma.
8.4 Allergy
Many aeroallergens like house dust mites and fungal allergens associated proteases play important role in asthma pathophysiology. Epidemiological studies suggested that sensitivity to fungal allergens could be an important cause of allergic asthma [156].
8.5 Alternaria Alternate and Asthma Severity
The fungus Alternaria alternate has been observed to cause asthma under certain circumstances [157, 158]. The allergen of the fungus possesses intrinsic proteolytic activities and that upon activating protease activated receptors (PARs) play a prominent role in mediating allergic airway diseases. Interleukin-33 (IL-33) has been observed to be associated with the development of allergic asthma [159]. IL-33 expression in the lung was found to be elevated in the asthmatic subjects and the asthma severity could be positively correlated with the IL-33 expression in the airways [160, 161].
Alternaria driven release of IL-33 occurs with the involvement of a serine protease specific to this aeroallergen. The Alternaria serine proteases cause marked inflammation because of the capacity of the serine protease to drive IL-33 release, which in turn induces rapid onset of asthma exacerbations. Thus, targeting the protease—IL-33 signaling axis could prove useful as a therapeutic measure in this kind of asthma pathogenesis [162].
8.6 Aeroallergenicity of Acanthamoeba
The free living amoeba, Acanthamoeba trophozoite, is found in human airway cavities and possesses high protease activities, which can elicit allergic airway inflammation [163]. Intranasal inoculation of A. trophozoite or its excretory secretory (ES) proteins in mice have been shown to elicit allergic airway inflammation. ES proteins with strong protease activities stimulate dendritic cells and also able to enhance the differentiation of early T cells into mature IL-4 secreting T cells. Treatment of ES proteins in the protease activated receptor (PAR-2) knockout mouse showed inhibition of lung airway inflammation and Th2 immune responses with lower IgE level compared with the normal mouse. This suggests a role of PAR-2 in the aeroallergenicity of Acanthamoeba allergens [163].
8.7 Seasonal Rhinitis and Asthma
Allergic diseases like seasonal rhinitis and asthma are generally result from exposure to airborne pollens. Asthmatic patients allergic to pollen have been observed to develop a chronic inflammation of the airways leading to bronchial obstruction and hyper responsiveness. Upon inhalation, pollen grains release a wide variety of allergens with protease activities, which may act as inflammatory mediators and subsequently pathogenesis of respiratory allergies. The proteases were known to inactivate lung regulatory neuropeptides, for example, substance P and vasoactive intestinal peptide (VIP) [164] leading to dysregulation of the contraction-relaxation rhythm of the respiratory airways. The inhaled allergens were processed by dendritic cells (DCs) present at the subepithelial regions, and then present allergen peptides to native T-lymphocytes for stimulation of IgE production. Some pollen allergens exhibit proteases such as aminopeptidase and trypsin-like serine protease activities, which can cleave proteins from junctional complexes between epithelial cells [165]. A 98 kDa aminopeptidase of Parietaria judaica, for instance, has been shown to cause detachment of A549 human alveolar epithelial cells by degrading intercellular adhesion proteins from tight junctions and adherens junctions [165, 166]. Pollen proteases can induce degradation of cell junction proteins such as occluding, claudin-1 and E-cadherin, thereby help allergens to cross the epithelial barrier and contact with DCs for intensifying immune response [164,165,166].
8.8 Organic Dust Allergy
Organic dusts made for agriculture may lead to airway inflammation, which may cause sinusitis and chronic bronchitis to workers in agricultural industries [167,168,169,170]. Workers in livestock industries working in concentrated animal feeding operations (CAFOs) are susceptible to chronic airway diseases [171]. Extracts of dust collected from CAFOs are potent stimulators of lung inflammatory responses. Hog dust extract (HDE) contains active proteases, which play a critical role in lung inflammatory processes [172, 173].
Epidermal growth factor receptor (EGFR) signaling has been shown to play an important role in the proinflammatory response of bronchial epithelial cells (BECs) to HDE [172]. The proinflammatory effect of HDE has been suggested to be due to the proteolytic activation of PARs, especially PAR-2 [173]. In lung epithelial cells, actions of fungal, cockroach and dust mite allergen proteases are mediated by the cleavage and activation of protease activated receptor-2 (PAR-2) [174,175,176]. However, PAR-1 is unable to mediate the effects of these proteases. PARS play an important role in bronchial fibroblast proliferation; epithelial cell wound healing and hypersecretion of mucus [177, 178]. By inhibiting HDE proteases or abrogating activation of epithelial cells, PAR-2 could inhibit HDE-induced inflammatory indexes in bronchial epithelial cells (BECs) and subsequently inhibition of the allergic response [179]. Thus, targeting the protease activity of organic dusts made for agricultural usage and other air borne dusts may prove useful as a strategy for preventing airway inflammation in agricultural workers, who are generally exposed to dusty agricultural environments.
8.9 Cockroach Allergy
Bernton and Brown [180] first observed skin rashes upon exposure of cockroach over the skin of allergic patients. Subsequently, a considerable number of evidence have confirmed that exposure to cockroach can induce allergy. Proteases associated with cockroach can produce bleb formation to the skin, which could play a critical role in the development of allergic disease [181,182,183].
Cockroach allergens such as saliva, feces, cast skins, and dead bodies contain serine protease activities, however, feces (frass) was found to be the prominent source of allergens, which contains serine protease activity [184, 185]. Sensitization of wild type mice to German cockroach frass (GC frass) has been shown to increase allergic hyperactivity (AHR) due to a marked increase in serum IgE and also production of cytokines such as IL-13, IL-4, IL-5, and IL-17 [186]. Sensitization of PAR-2 deficient mice with GC frass, however, did not show a discernible increase in allergic airway inflammation indicating a role of PAR-2 in mediating allergic airway inflammation [185].
9 Lung Cancer
Lung cancer is one the most prevalent and lethal diseases worldwide. Despite recent advances in chemotherapy, the molecular basis of its progression to a metastatic disease remains unclear.
Exposure of bronchial epithelial cells of smokers produce oxidants and reactive oxygen species (ROS) with consequent cellular responses to activate NF-кB and other transcription factors that regulate inflammation-related genes and initiates several signaling pathways depending on both genetic and epigenetic factors and manifest COPD and lung cancer [187, 188].
9.1 MMPs and Lung Cancer
Lung cancers are of two major types: (i) small cell lung carcinoma (LC); and (ii) non-small cell lung carcinoma (NSCLC). ECM and basement membrane components are proteolytically cleaved by MMPs. These components play a pivotal role in cancer progression. Lung cancers express high levels of MMPs. MMP-7 and MMP-9 expressions were found to be markedly high in NSCLC in comparison to the normal tissue [189]. MMP-1 and MMP-3 promoter polymorphisms are associated in modifying susceptibility to NSCLC and also to an increase in the risk of lymphatic metastasis of these tumors [190]. MMP-2 and MMP-9 activities have been found to be associated with an increase in tumor spread [191].
Treatment of mice with CH1104I (dual inhibitor of MMP-2 and -9) has been shown to markedly inhibit metastasis of lung carcinoma, which suggests that inhibition of MMP-2 and -9 could significantly inhibit tumor invasion and metastasis [192]. MMP-2 and -9 expressions may have prognostic implications in patients with NSCLC [193]. Over expression of MMP-1 has been observed to induce the formation of lung metastases [194, 195].
9.2 High Temperature Requirement A (HtrA) Serine Protease and Lung Cancer
HtrA (a.k.a. DegP) is a heat shock-induced serine protease that is active in the periplasm of Escherichia coli. Homologues of HtrA were found in a wide range of bacteria and in eukaryotes. Till date, four human homologues of the bacterial serine protease HtrA have been described, which are named as HtrA-1, -2, -3, and -4 [196, 197]. They have a variety of functions including cancer [198].
HtrA1 has been observed to be downregulated in lung cancers [198]. In human cancer cells, HtrA1 over expression prevents cancer cell growth and proliferation suggesting HtrA1 as a tumor suppressor. A modest expression of HtrA1 has been observed in primary tumors and lymph node metastases. However, the exact functions of HtrA1 in cancer are mostly unknown. A previous report suggested that HtrA1 elicits its function by inhibiting TGF-β pathway [199]. The role of TGF-β in cancer progression is well documented [200]. Accordingly, the TGF-β signaling pathway has been considered as both a tumor suppressor and promoter pathway of tumor progression and invasion. It, therefore, seems probable that activation of TGF-β signaling pathway occurs when HtrA1 is downregulated, thereby contributing to the cancer progression. Alternatively, over expression of HtrA1 has been shown to induce apoptosis [201]. Thus, loss of HtrA1 expression alters regulation of apoptosis and could lead to cancer progression [201].
HtrA1 degrades tubulin by disrupting microtubules (MTs), which suggest that HtrA1 could play an important role in regulating MT and tubulin stability and MT-associated cellular functions [245]. HtrA1 also regulates cell migration and offers a potential role in regulating MT organization associated with cell migration. However, the exact mechanism(s) by which HtrA1 regulates cell migration is currently unclear. Active HtrA1 upon removal of N-terminal Kazal-type trypsin inhibitory domains contributes to cell death through caspase dependent, as well caspase-independent mechanisms [202].
The role of HtrA2 protease in stress responses and apoptosis in lung cells has been established and a previous report suggested its involvement in cisplatin-induced death of renal cells [203]. Over expression of HtrA2 by cisplatin has been observed to follow the release of HtrA2 from mitochondria to the cytosol and it degrades anti-apoptotic proteins. This mechanism seems to be obligatory to trigger mitochondrial permeabilization for HtrA2 to participate in cell death [203, 204]. Therefore, HtrA2 plays a vital role in programmed cell death upon eliminating the caspase inhibitory activity of apoptosis [205, 206]. However, the detail mechanism(s) by which HtrA2 regulates lung cancer is currently unknown.
Smoking is a critical factor for lung cancers and the HtrA3 has been shown to be associated with smoking-induced lung cancer. HtrA3 expression has been found to be markedly downregulated in lung cancer cell lines and also primary lung tumors isolated from heavy smokers [207]. HtrA3, in contrast to the steady HtrA1 and HtrA2 expression, has been identified as a probable target for cigarette smoke-induced changes in normal human bronchial epithelial cells [207]. It has also been suggested that cigarette smoke-induced methylation of HtrA3 may play an important role in the etiology of smoking-linked lung cancer [207].
Studies on HtrA3 exon in lung cancer cell lines indicates that it possesses core xenobiotic response element (XRE) consensus sequence, 5-TNGCGTG-3 and is a target for methylation at CpG. XRE is located in the promoter region of the genes involved in metabolizing xenobiotic carcinogens, mainly aryl hydrocarbons from cigarette smoke and environmental pollutants, for instance, automobile exhaust [208, 209]. These compounds upregulate the gene products of XRE via aryl hydrocarbon receptors and multitude of other transcription factors [210, 211]. The degree of methylation of HtAr3 is similar when studied in A549 and H157 lung epithelial cells, when treated with NNK (Nicotine-derived nitrosamine ketone), a.k.a 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone, an important tobacco-specific nitrosamines, which play key role in carcinogenesis [208]. NNK suppressed the expression of HtAr3, whereas 5-azo-dc has been shown to induce it [208]. Differential expressivity of HtAr3 may be controlled by other cross-talking mechanisms such as histone deacetylation, micro RNA activation, loss of heterozygosity and genetic mutation. Along with that HTAr3, HtAr1, and HtAr2 are also believed to be upregulated by xenobiotic stress [208, 212]. However, more research is needed to ascertain them as therapeutic targets pertaining to lung cancer for epigenetic therapies as well as prognosis to forecast tumor response.
10 Tuberculosis
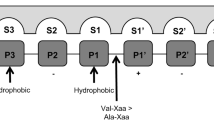
Upregulation of CatG, but not NE, has been shown to induce cell death of activated Mycobacterium tuberculosis infected macrophages. The substrate specificity of CatG and NE is distinct. CatG cleaves the C-terminus of aromatic or positively charged amino acid residues, while NE cleaves the C-terminus of small hydrophobic amino acid residues [213, 214]. Enhanced necrosis in infected macrophages by CatG may result from proteolysis of specific target sequences, which are currently unknown. It has been shown that serpinb3a inhibition of CatG is necessary to prevent necrosis induced by IFN-γ in M. tuberculosis infected macrophages [214].
Role of matrix metalloproteinases (MMPs) as an important mediator of tissue destructive response in TB has now been clearly known [215, 216]. MMPs can cleave ECM components [217]. In humans, MMP-1 cleaves fibrillar components (types I and III collagen) of ECM. The level of MMP-3, the activator of MMP-1 is high in respiratory secretions of TB patients than control subjects [216,217,218]. In rabbits, MMP-1, -3, -7, -12, and -13 expressions are elevated in granulomatous and cavitary pathologies in human respiratory secretions observed in vivo model systems of TB [218,219,220]. In a human monocyte infection model, a marked reduction in TIMP-3 has been observed to be correlated with TB pathogenesis [221]. In a mice model, TIMP-3 has been observed to be associated with cavity formation and subsequently ECM degradation, which are important for TB pathogenesis [221, 222].
11 Acute Lung Injury
Severe acute respiratory distress syndrome (SARS) was identified in 2003, which triggered death of thousands of people worldwide [223, 224]. A new type of coronavirus has been identified and found responsible for the SARS, which produces pneumonia and associated high fever and severe dyspnea and subsequently acute respiratory distress syndrome (ARDS) followed by death due to acute lung injury (ALI) [224, 225]. ARDS is characterized by accumulation of inflammatory cells and severe hypoxia that leads to pulmonary edema [226].
Renin-angiotensin system (RAS) has been observed to play a critical role in SARS. In animal studies, a prominent role of angiotensin converting enzyme (ACE) in the pathogenesis of ARDS has been suggested [227, 228]. ACE2, a homologue of ACE, was shown to be a key regulator for coronovirus infection that produces SARS. ACE2 has been shown to be expressed in the lungs of both healthy and diseased humans, and it protects against SARS-induced ALI [229,230,231]. Therefore, it seems conceivable that ACE2 might prove a novel therapeutic target for SARS-coronovirus-induced ARDS that develops in emerging lung infectious diseases including influenza [231].
12 Elafin and ALI
Elafin, a serine protease inhibitor having mol wt of 6 kDa, found in lung secretions. Elafin is formed by proteolytic cleavage of its precursor protein, trappin-2 [232]. The antiprotease activity of elafin is located in the C-terminal domain having specificity for NE and proteinase 3. The N-terminus transglutaminase substrate binding motif (GQDPVK) of elafin cross-links with extracellular matrix proteins [233, 234].
In ALI, the protease–antiprotease balance alters in favor of proteases leading to an increase in protease activity, and this protease burden can produce pulmonary edema [235]. A decrease in plasma elafin level has been observed to be correlated with altered elafin gene expression and seems a critical component for an increase in acute respiratory distress syndrome (ARDS) [236,237,238].
In ALI patients, the 20S proteasome was observed to be markedly higher compared with normal subjects [239], but elafin level was shown to be decreased with consequent proteolytic degradation of antiproteases by the 20S proteasome in lung patients with ALI. This decrease may contribute to an increase in NE activity in ALI regulation and expression; however, its biological role in the lung is currently unknown. The cleavage of elafin by 20S proteosome suggests that the increment of antiprotease levels in ALI patients could prove clinically beneficial in attenuating uncontrolled activity of NE. Elafin’s multifunctional properties could prove useful as the therapeutic target for ALI [239,240,241].
13 Elane and ALI
Elane has a potential catalytic activity to hydrolyze elastin. Under physiological conditions, lungs are protected from this enzyme by endogenous inhibitors such as α1-PI, α2-macroglobulin, and SLPI. However, in the course of ALI, the balance between elane and its endogenous inhibitors is disregulated in favor of the enzyme [242,243,244] leading to massive infiltration of neutrophils into the lungs and subsequently tissue injury. Therefore, peptidic and non-peptidic elane inhibitors may prove useful for treating ALI associated with systemic inflammation [245, 246].
14 Age-Related Pulmonary Diseases
Granzymes, especially granzyme A and granzyme B, are the most abundant granzymes involving the membrane perforating molecule, perforin, which induce cell death [247]. Perforin facilitates granzymes entry into the target cell and that subsequently induces cell death [248]. Granzyme A has originally been thought to induce caspase-independent cell death; however, recent findings suggest that granzyme A may be involved in immune regulation of age-related lung disorders [249, 250]. In contrast, granzyme B induces apoptosis through caspase-dependent and -independent pathways [251]. An increase in granzyme A and B activities is known to promote generation of proinflammatory cytokines. ECM degradation and formation of autoantigens may exacerbate the inflammatory response [251]. Chronic inflammation is a hallmark of age-related cardiovascular and lung diseases. Thus, granzyme A and B may serve as important agent in promoting a positive feedback cycle that may be common to many persistent age-related disorders [251, 252]. However, role of other granzymes such as GzmH, GzmK, and GzmM in age-related ARDS are currently unknown.
15 Aspergillosis
Invasive pulmonary aspergillosis (IPA) elicited by the filamentous members of the genus Aspergillus can have devastating role in immune compromised individuals [253, 254]. Aspergillus fumigatus is responsible for the majority of IPA infections. Normal individuals are at little risk because of the effectiveness of their lung defences. However, inhalation of conidia becomes life threatening to subjects having weak immunity, which could allow the conidia to germinate into invasive hyphae in the lung [255].
The populations at greatest risk for IPA are patients with cancer, solid organ transplants, bone marrow transplants and those with advanced AIDS [256]. A. fumigatus secretes proteases like an alkaline serine protease (ALP), a metalloprotease (Mep), an aspartic protease (Pep) and a prolyl endopeptidase in the lungs [257,258,259,260]. A. fumigates secreted proteases are expressed in the lung during infection [261, 262].
A. fumigates binds to ECM proteins with the involvement of polysaccharides and glycoproteins of the conidial cell wall [263]. Several agents secreted from fungus such as proteases and toxins have been shown to influence its infection in host lung tissue [263].
16 Systemic Sclerosis
Systemic sclerosis (SSc) is an autoimmune disease, which could occur due to vascular injuries and fibrosis in skin and certain internal organs [264]. Some cytokines and growth factors like transforming growth factor-β (TGF-β) have been observed to stimulate fibroblast proliferation [264]. The disintegrin and metalloprotease-12 (ADAM-12) possess the extracellular cell binding functions. ADAM-12 is expressed in two alternative forms: (i) membrane-anchored form (ADAM12-L); and (ii) a short secreted form (ADAM12-S). An increase in the level of serum ADAM12-S level plays an important role in the pathological events of diffuse cutaneous systemic sclerosis (dcSSc) [264, 265].
ADAM-12 plays a critical role in fibrotic process. ADAM12-S has the ability to degrade physiological substrates such as the ECM substrates: fibronectin, type IV collagen [265] and also insulin-like growth factor binding protein (IGFBPs) [266, 267]. Degradation of IGFBPs augments the association between insulin-like growth factors, for example, IGF-I and its receptors. IGF-I down regulates collagenase activity with consequent increase in collagen production [268], which indicates that IGF-I could be an important mediator in the progression of fibrosis. Additionally, ADAM-12 has been observed to be upregulated in chronic wound suggesting that ADAM-12 could be related to the fibrotic process [269].
17 Bronchopulmonary Dysplasia
Bronchopulmonary dysplasia (BPD) usually occurs in prematurely born infants. Due to deficiency of lung development, BPD patients require prolonged medical ventilation for oxygen. BPD causes an increase in morbidity and mortality in preterm infants. BPD is characterized by chronic inflammation, alveolar hypoplasia and respiratory infections [270, 271]. In an animal model of BPD, a significant high elastase activity along with excessive proteolytic degradation of the elastic fibers due to a marked decrease in the levels of endogenous protease inhibitors are usually observed in the lung secretion [272]. Additionally, a discernible increase in mRNA and protein expression and also activities of cathepsins-B, -H, -K, -L, and -S have been observed in new borne BPD tracheal aspirates [273]. BAL fluid of newborn preterm infants with BPD showed elevation of MMP-9 and a decrease in free TIMP-1 level [274, 275].
18 Lymphangioleiomyomatosis
Lymphangioleiomyomatosis, a rare and progressive lung disease, usually affects women of pre-menopausal age. This disease is characterized by the infiltration of smooth muscle cells that express contractile proteins, for example, desmin. Immunohistochemical studies have demonstrated a strong expression of CatK restricted to lymphangioleiomyomatosis cells. CatK has been suggested as a marker for diagnosis of lymphangioleiomyomatosis [276].
19 Bronchiolitis Obliterans Syndrome
Bronchiolitis obliterans syndrome (BOS) is a complication, which usually occurs during chronic rejection of lung transplant patients. Elevated levels of MMP-8 and MMP-9 were observed in obliterative bronchiolitis patients after a few years of lung transplantation [277]. A marked elevation in gelatinase activity was found in BAL fluid from BOS patients that could be due to MMP-9 secretion by local neutrophils [278]. In lung transplant model, inhibition of matrix metalloproteases in the donor and recipient, respectively, before lung harvest and after lung transplantation have been shown to improve oxygenation and markedly decreased PMN leukocyte influx into the isograft [279]. Both MMP-8 and MMP-9 deficient mice were observed to be protected from BOS as evidenced by a marked decrease in neutrophil influx and collagen deposition [280, 281].
MMPs and TIMPs were suggested to play a crucial role important role during lung allograft rejection. While TIMP-1 and TIMP-2 over expression have not been observed to elicit consistent effect on the level of cytokines or rejection pathology, MMP inhibition via systemic administration of MMP inhibitors were shown to reduce lung allograft rejection [282].
20 Lung Surfactant Proteins and Proteases
Lung surfactants are a mixture of lipids and proteins complex, which form a thin film in the lung alveoli and that plays a vital role in respiratory function especially gas exchange [283]. Additionally, the surfactant also shows the first line of innate immune defence in the lung. Its mode of action appears to lie in the inhibition of microbial infectivity and attenuation of inflammatory responses [284]. SP-A, SP-B, SP-C, and SP-D are the major surfactant proteins, which elicit important roles to trigger immune response in the lung [285,286,287,288,289]. SP-A has been demonstrated to be an important surfactant component having relevant functional immune response during Staphylococcus aureus infection [290].
Surfactant protein D (SP-D) is an important target of numerous proteases present in the CF lung. Host defence appears to be impaired due to proteolysis of SP-D and may contribute to the supportive lung disease in CF. SP-D, a glycoprotein of the collecting family, is produced and secreted by alveolar type II cells and non-ciliated bronchial epithelial cells [291].
SP-D has been observed to be protective against a wide variety of pathogens such as Pseudomonas aeruginosa, Haemophilus influenza, and A. fumigates [291,292,293]. Upon binding with SP-D, these pathogens trigger their agglutination, enhanced killing and clearance [294, 295]. SP-D has been shown to cause a number of secretions, which present larger protection in the body. An important characteristic of CF is chronic neutrophil-mediated inflammation in the airways mainly with an increase in the levels of HLE and PR3 [296,297,298]. Proteolytic degradation of some proteins such as SP-A and SP-D was found in BALF of CF patients [299].
Proteases have been observed to modulate surfactant activity in addition to its action on mucus proteins. Secretory proteases of P. aeruginosa can degrade SP-A and SP-B from lipid–protein complexes [300]. Purified elastase or secretory protease IV of P. aeruginosa supernatants have also been shown to degrade SP-A and SP-D [301,302,303]. The protease IV-mediated degradation of SP-A and SP-D may cause a discernible loss of bacterial aggregation or increase bacterial phagocytosis by alveolar macrophages [304]. NSPs like NE, PR3 and CatG can cleave the surfactant proteins [305]. These proteases may also cleave SP-D within the conserved sub-region of the C-terminal lectin domain with the generation of a ~35-kDa fragment, which decreases bacterial aggregation and mannan binding of SP-D [306]. P. aeruginosa elastase digestion of SP-D has also been shown to produce the 35-kDa fragment that retain the N-terminal collagen tail, albeit devoid of functional C-terminal globular lectin domain, which consequently elicit loss of innate immune functions [300, 303, 304].
In CF, COPD and asthma, like chronic lung airway diseases, an increase in the epidermal growth factor receptor (EGFR) could be the mechanism for mucus production [307, 308]. EGFR phosphorylation has been observed to activate mitogen activated protein kinases (MAPKs)-dependent signaling pathways, which in turn stimulates MMPs, for example, ADAM-17 and also NSPs leading to the production of mucins [309,310,311,312].
Human airway trypsin-like protease (HAT) has been observed to enhance the synthesis of mucus glycoconjugates in airway epithelial cells [313]. HAT is a natural ligand for PAR-2 present in bronchial cells [314] and HAT-dependent upregulation of mucin genes have also been shown to occur via PAR-EGFR signaling pathway [313].
21 Particulate Matter
Particulate matter (PM) having diameter of about 10 nm (PM10) is a complex mixture of metals, polycyclic aromatic hydrocarbons, nitrates, sulfates and other chemicals [315], where traffic and industrial activities have an important impact on that composition. Adverse effects of PM10, especially on alveolar epithelia were related to inflammation triggered by phagocytic cells upon PM10 internalization [316]. An immediate response is to augment generation of cytokines and chemokines such as IL-1, IL-6, and IL-8, TNF-α [317, 318].
Airborne PM10 is a risk factor for the development of a variety of lung diseases including cancer [315, 316]. In vitro, treatment of PM10 induces an increase in MMP-2 and MMP-9 activities, which causes ECM degradation during acute lung injury [319]. PM10 was found to be responsible for lung diseases such as tuberculosis [320], emphysema [321] and COPD [322]. In A549 lung epithelial cells, PM10 causes a marked decrease in E-cadherin/β-catenin expression and subsequently induces potentially invasive characteristics and thereby could contribute to cancer development [323].
22 Conclusion and Future Perspective
In order to fight against infections, the lung is orchestrated with different antiproteases and anti-inflammatory components. In pulmonary diseases like COPD and asthma, the balance between host proteases and their secreted endogenous inhibitors shift toward the proteases. Proteases such as NSPs, MMPs, and cathepsins are known to act along with the bacterial proteases and play important role in the manifestation of a variety of pulmonary diseases. Thus, agents that restore lung protease–antiprotease balance by upregulating endogenous protease inhibitors and/or down regulating host protease activities, appear important to control excessive inflammatory responses in the lung.
Inflammatory cells, which are rich in oxidants and proteases cause proteolytic inactivation of protein inhibitors of proteases. SLPI and trappin-2/elafin are relatively stable; even though they may be cleaved and inactivated by proteases, for example, by cathepsins at their N-terminal end, albeit it does not affect their inhibitory potency [324]. In contrast, cleavage of elafin by P. aeruginosa proteases may inactivate its antiprotease activity [325]. To overcome the unwanted proteolysis of antiproteases, encapsulations of protease inhibitors within liposomes have been suggested [326]. Aerosol delivery of liposomes entrapped antiproteases may prove useful since it has several features such as sustained release and relatively high loading capacities.
NE has been chosen as a target for inhibition by synthetic compounds because it is a widely recognized serine protease, which has been shown to be associated with a variety of lung diseases including CF [327]. DX-890, a small protein inhibitor of NE, has been observed to be tolerable in rats and humans after phase 1 clinical trial [328]. This compound was shown to be involved in IL-8 release from CF neutrophils and to reduce neutrophil transmigration through the epithelial barrier. Outcome of phase 3 clinical trials will reveal the usefulness of DX-890 as a therapeutic measure for a variety of lung diseases.
In a guinea pig model system, the dual MMP9/MMP12 inhibitor, AZ11557272 was found to be protective toward cigarette smoke-induced emphysema [329]. This compound markedly reduces number of inflammatory cells in bronchoalveolar lavage (BAL) fluid and has also been shown to decrease smoke-induced air space enlargement, which suggests that MMP-2 and MMP-12 could be the potential targets for therapeutic intervention in COPD. AS112108, another dual MMP-9/MMP-12 inhibitor, has been shown to inhibit the early inflammatory responses with a decrease in neutrophil numbers [330]. AS111793, a selective MMP-12 inhibitor, elicited dose-dependent inhibition in the levels of neutrophils and macrophages in bronchoalveolar lavage (BAL) fluid, and also on the concentration of several inflammation markers that express after cigarette smoke exposure [331]. However, it did not inhibit lung inflammation observed by lipopolysaccharide (LPS). Understanding the mechanism of these antiproteases will eventually lead us to gain an insight into the basic biochemical mechanisms that regulate COPD.
MMP-1 has been considered as one of the target proteases in lung cancer [332]. The generation of MMP-1 deficient mouse model suggested pro-tumorigenic role of the enzyme [333]. Further studies on MMP-1 and other MMPs in knockout mice will be required to evaluate the functional redundancy and relative relevance of MMP-1 and other MMPs in cell proliferation, regulation of inflammatory cells, and different stages of cancer progression.
References
Lecaille F, Lalmanach G, Andrault PM (2016) Antimicrobial proteins and peptides in human lung diseases: a friend and foe partnership with host proteases. Biochimie 122:151–168
Geraghty P, Rogan MP, Greene CM et al (2007) Neutrophil elastase up-regulates cathepsin B and matrix metalloprotease-2 expression. J Immunol 178:5871–5878
Imai K, Yokohama Y, Nakanishi I et al (1995) Matrix metalloproteinase 7 (matrilysin) from human rectal carcinoma cells. Activation of the precursor, interaction with other matrix metalloproteinases and enzymic properties. J Biol Chem 270:6691–6697
Ferry G, Lonchampt M, Pennel L et al (1997) Activation of MMP-9 by neutrophil elastase in an in vivo model of acute lung injury. FEBS Lett 402:111–115
Shamamian P, Schwartz JD, Pocock BJ et al (2001) Activation of progelatinase A (MMP-2) by neutrophil elastase, cathepsin G, and proteinase-3: a role for inflammatory cells in tumor invasion and angiogenesis. J Cell Physiol 189:197–206
Nakamura H, Yoshimura K, McElvaney NG et al (1992) Neutrophil elastase in respiratory epithelial lining fluid of individuals with cystic fibrosis induces interleukin-8 gene expression in a human bronchial epithelial cell line. J Clin Invest 89:1478–1484
Benarafa C, Priebe GP, Remold-O’Donnell E (2007) The neutrophil serine protease inhibitor serpinb1 preserves lung defense functions in Pseudomonas aeruginosa infection. J Exp Med 204:1901–1909
D’Alonzo GE, Barst RJ, Ayres SM et al (1991) Survival in patients with primary pulmonary hypertension. Results from a national prospective registry. Ann Intern Med 115:343–349
Perkett EA, Lyons RM, Moses HL et al (1990) Transforming growth factor-beta activity in sheep lung lymph during the development of pulmonary hypertension. J Clin Invest 86:1459–1464
Meyrick B, Reid L (1983) Pulmonary hypertension. Anatomic and physiologic correlates. Clin Chest Med 4:199–217
Archer SL, Nelson DP, Weir EK (1989) Detection of activated O2 species in vitro and in rat lungs by chemiluminescence. J Appl Physiol 67:1912–1921
Farrukh IS, Sciuto AM, Spannhake EW et al (1986) Leukotriene D4 increases pulmonary vascular permeability and pressure by different mechanisms in the rabbit. Am Rev Respir Dis 134:229–232
Farrukh IS, Michael JR, Summer WR et al (1985) Thromboxane-induced pulmonary vasoconstriction: involvement of calcium. J Appl Physiol 58:34–44
Freeman BA, Topolosky MK, Crapo JD (1982) Hyperoxia increases oxygen radical production in rat lung homogenates. Arch Biochem Biophys 216:477–484
Gurtner GH, Michael JR, Farrukh IS et al (1985) Mechanism of hyperoxia-induced pulmonary vascular paralysis: effect of antioxidant pretreatment. J Appl Physiol 59:953–958
Farrukh IS, Michael JR, Peters SP et al (1988) The role of cyclooxygenase and lipoxygenase mediators in oxidant-induced lung injury. Am Rev Respir Dis 137:1343–1349
Feddersen CO, Chang S, Czartalomna J et al (1990) Arachidonic acid causes cyclooxygenase-dependent and -independent pulmonary vasodilation. J Appl Physiol 68:1799–1808
Gurtner GH, Knoblauch A, Smith PL et al (1983) Oxidant- and lipid-induced pulmonary vasoconstriction mediated by arachidonic acid metabolites. J Appl Physiol 55:949–954
Gurtner GH, Burke-Wolin T (1991) Interactions of oxidant stress and vascular reactivity. Am J Physiol 260:L207–L211
Seeger W, Wolf H, Graubert E et al (1983) Influence of aprotinin and gabexate mesilate on arachidonic acid release by the Ca-ionophore A 23187 in the lung. Adv Exp Med Biol 156:553–567
White RP (1988) Pharmacodynamic effects of tosyl-arginine methyl ester (TAME) on isolated human arteries. Gen Pharmacol 19:387–392
Chakraborti S, Gurtner GH, Michael JR (1989) Oxidant-mediated activation of phospholipase A2 in pulmonary endothelium. Am J Physiol 257:L430–L437
de Haas GH, Postema NM, Nieuwenhuizen W, van Deenen LL (1968) Purification and properties of an anionic zymogen of phospholipase A from porcine pancreas. Biochim Biophys Acta 159(1):118–129
Chakraborti S, Roy S, Mandal A et al (2012) Role of PKCα-p38MAPK-Giα axis in NADPH oxidase derived O2—mediated activation of cPLA2 under U46619 stimulation in pulmonary artery smooth muscle cells. Arch Biochem Biophys 523:169–180
Chakraborti T, Das S, Chakraborti S (2005) Proteolytic activation of protein kinase Calpha by peroxynitrite in stimulating cytosolic phospholipase A2 in pulmonary endothelium: involvement of a pertussis toxin sensitive protein. Biochemistry (USA) 44:5246–5257
Chakraborti S, Chowdhury A, Chakraborti T (2015) Cross-talk between p(38)MAPK and G iα in regulating cPLA 2 activity by ET-1 in pulmonary smooth muscle cells. Mol Cell Biochem 400:107–123
Chakraborti S, Mandal A, Das S, Chakraborti T (2004) Inhibition of Na+/Ca2+ exchanger by peroxynitrite in microsomes of pulmonary smooth muscle: role of matrix metalloproteinase-2. Biochim Biophys Acta 1671:70–78
Chakraborti S, Mandal A, Das S, Chakraborti T (2005) Role of MMP-2 in PKCdelta-mediated inhibition of Na+ dependent Ca2+ uptake in microsomes of pulmonary smooth muscle: involvement of a pertussis toxin sensitive protein. Mol Cell Biochem 280:107–117
Chakraborti T, Ghosh SK, Michael JR et al (1996) Role of an aprotinin-sensitive protease in the activation of Ca2+-ATPase by superoxide radical O −∙2 in microsomes of pulmonary vascular smooth muscle. Biochem J 317:885–890
Ghosh SK, Chakraborti T, Michael JR et al (1996) Oxidant-mediated proteolytic activation of Ca2+-ATPase in microsomes of pulmonary smooth muscle. FEBS Lett 387:171–174
Chakraborti T, Das S, Mandal M et al (2002) Role of Ca2+ dependent metalloprotease-2 in stimulating Ca2+ ATPase activity under peroxynitrite treatment in bovine pulmonary artery smooth muscle cell membrane. IUBMB Life 53:167–173
Bouchard RA, Bose D (1992) Contribution of sarcolemmal sodium-calcium exchange and intracellular calcium release to force development in isolated canine ventricular muscle. J Gen Physiol 99:931–960
Blaustein MP (1988) Sodium/calcium exchange and the control of contractility in cardiac muscle and vascular smooth muscle. J Cardiovasc Pharmacol 5:S56–S68
Vieillard-Baron A, Frisdal E, Raffestin B et al (2003) Inhibition of matrix metalloproteinases by lung TIMP-1 gene transfer limits monocrotaline-induced pulmonary vascular remodeling in rats. Hum Gene Ther 14:861–869
Stenmark KR, Mecham RP (1997) Cellular and molecular mechanisms of pulmonary vascular remodeling. Ann Rev Physiol 59:89–144
Stamenkovic I (2003) Extracellular matrix remodelling: the role of matrix metalloproteinases. J Pathol 200:448–464
Woessner JF Jr (1991) Matrix metalloproteinases and their inhibitors in connective tissue remodeling. FASEB J 5:2145–2154
Chakraborti S, Mandal M, Das S, Mandal A, Chakraborti T (2003) Regulation of matrix metalloproteinases: an overview. Mol Cell Biochem 253:269–285
Mandal M, Das S, Chakraborti T, Mandal A, Chakraborti S (2003) Identification, purification and partial characterization of tissue inhibitor of matrix metalloproteinase-1 (TIMP-1) in bovine pulmonary artery smooth muscle. Mol Cell Biochem 254:145–155
Mandal M, Mandal A, Das S, Chakraborti T, Chakraborti S (2003) Identification, purification and partial characterization of tissue inhibitor of matrix metalloproteinase-2 in bovine pulmonary artery smooth muscle. Mol Cell Biochem 254:275–287
Roy S, Chakraborti T, Chowdhury A, Chakraborti S (2013) Role of PKC-α in NF-κB-MT1-MMP-mediated activation of proMMP-2 by TNF-α in pulmonary artery smooth muscle cells. J Biochem 153:289–302
Roy S, Samanta K, Chakraborti T et al (2011) Role of TGF-β1 and TNF-α in IL-1β mediated activation of proMMP-9 in pulmonary artery smooth muscle cells: involvement of an aprotinin sensitive protease. Arch Biochem Biophys 513:61–69
Palese P (2004) Influenza: old and new threats. Nat Med 10:S82–S87
Yewdell J, Garcia-Sastre A (2002) Influenza virus still surprises. Curr Opin Microbiol 5:414–418
Horimoto T, Kawaoka Y (2005) Influenza: lessons from past pandemics, warnings from current incidents. Nat Rev Microbiol 3:591–600
Garten RJ, Davis CT, Russell CA et al (2009) Antigenic and genetic characteristics of swine-origin 2009 A(H1N1) influenza viruses circulating in humans. Science 325:197–201
Blanquer J, Blanquer R, Borra’s R et al (1991) Aetiology of community acquired pneumonia in Valencia, Spain: a multicentre prospective study. Thorax 46:508–511
Lauderdale TL, Chang FY, Ben RJ et al (2005) Etiology of community acquired pneumonia among adult patients requiring hospitalization in Taiwan. Respir Med 99:1079–1086
Garten W, Klenk HD (2008) Cleavage activation of the influenza virus hemagglutinin and its role in pathogenesis. In Avian influenza: monographs in virology, vol 27. Karger, Basel, Switzerland
Klenk HD, Garten W (1994) Host cell proteases controlling virus pathogenicity. Trends Microbiol 2:39–43
Steinhauer DA (1999) Role of hemagglutinin cleavage for the pathogenicity of influenza virus. Virology 258:1–20
Skehel JJ, Wiley DC (2000) Receptor binding and membrane fusion in virus entry: the influenza hemagglutinin. Annu Rev Biochem 69:531–569
Klenk HD, Rott R, Orlich M et al (1975) Activation of influenza A viruses by trypsin treatment. Virology 68:426–439
Lazarowitz SG, Choppin PW (1975) Enhancement of the infectivity of influenza A and B viruses by proteolytic cleavage of the hemagglutinin polypeptide. Virology 68:440–454
Goto H, Kawaoka Y (1998) A novel mechanism for the acquisition of virulence by a human influenza A virus. Proc Natl Acad Sci U S A 95:10224–10228
Lazarowitz SG, Goldberg AR, Choppin PW (1973) Proteolytic cleavage by plasmin of the HA polypeptide of influenza virus: host cell activation of serum plasminogen. Virology 56:172–180
LeBouder F, Morello E, Rimmelzwaan GF et al (2008) Annexin II incorporated into influenza virus particles supports virus replication by converting plasminogen into plasmin. J Virol 82:6820–6828
Kido H, Okumura Y, Yamada H et al (2007) Proteases essential for human influenza virus entry into cells and their inhibitors as potential therapeutic agents. Curr Pharm Des 13:405–414
Tashiro M, Ciborowski P, Klenk HD et al (1987) Role of Staphylococcus protease in the development of influenza pneumonia. Nature 325:536–537
Scheiblauer H, Reinacher M, Tashiro M et al (1992) Interactions between bacteria and influenza A virus in the development of influenza pneumonia. J Infect Dis 166:783–791
Bahgat MM, Błazejewska P, Schughart K (2011) Inhibition of lung serine proteases in mice: a potentially new approach to control influenza infection. Virol J 8:27
Böttcher E, Matrosovich T, Beyerle M et al (2006) Proteolytic activation of influenza viruses by serine proteases TMPRSS2 and HAT from human airway epithelium. J Virol 80:9896–9898
Chaipan C, Kobasa D, Bertram S et al (2009) Proteolytic activation of the 1918 influenza virus hemagglutinin. J Virol 83:3200–3211
Steinhoff M, Buddenkotte J, Shpacovitch V et al (2005) Proteinase-activated receptors: transducers of proteinase-mediated signaling in inflammation and immune response. Endocr Rev 26:1–43
Vergnolle N (2008) Proteinase-activated receptors (PARs) in infection and inflammation in the gut. Int J Biochem Cell Biol 40:1219–1227
Riteau B, de Vaureix C, Lefevre F (2006) Trypsin increases pseudorabies virus production through activation of the ERK signalling pathway. J Gen Virol 87:1109–1112
Chignard M, Pidard D (2006) Neutrophil and pathogen proteinases versus proteinase-activated receptor-2 lung epithelial cells: more terminators than activators. Am J Respir Cell Mol Biol 34:394–398
Lan RS, Stewart GA, Goldie RG et al (2004) Altered expression and in vivo lung function of protease-activated receptors during influenza A virus infection in mice. Am J Physiol 286:L388–L398
Khoufache K, LeBouder F, Morello E et al (2009) Protective role for protease-activated receptor-2 against influenza virus pathogenesis via an IFN-gamma-dependent pathway. J Immunol 182:7795–7802
Ovcharenko AV, Zhirnov OP (1994) Aprotinin aerosol treatment of influenza and paramyxovirus bronchopneumonia of mice. Antiviral Res 23:107–118
Lee MG, Kim KH, Park KY et al (1996) Evaluation of anti-influenza effects of camostat in mice infected with non-adapted human influenza viruses. Arch Virol 141:1979–1989
Manzano-Leon N, Quintana R, Sanchez B (2013) Variation in the composition and in vitro proinflammatory effect of urban particulate matter from different sites. J Biochem Mol Toxicol 27:87–97
Heijink IH, de Bruin HG, Dennebos R et al (2016) Cigarette smoke-induced epithelial expression of WNT-5B: implications for COPD. Eur Respir J ERJ-01541-2015
Chadwick D, Goode JA (eds) (2001) Chronic obstructive pulmonary disease: pathogenesis to treatment. Novartis Foundation Symposium 234, vol 234. Novartis Foundation. ISBN: 0-471-49437-2
Stockley JA, Stockley RA (2016) Pulmonary physiology of chronic obstructive pulmonary disease, cystic fibrosis, and alpha-1 antitrypsin deficiency. Ann Am Thorac Soc 2:S118–S122
Zuo L, Pannell BK, Zhou T et al (2016) Historical role of alpha-1-antitrypsin deficiency in respiratory and hepatic complications. Gene 589:118–122
Ganrot PO, Laurell CB, Eriksson S (1967) Obstructive lung disease and trypsin inhibitors in α1-antitrypsin deficiency. Scand J Clin Lab Invest 19:205–208
Kueppers F, Briscoe WA, Bearn AG (1964) Hereditary deficiency of serum α-l-antitrypsin. Science 146:1678–1679
Okada Y, Watanabe S, Nakanishi I et al (1988) Inactivation of tissue inhibitor of metalloproteinases by neutrophil elastase and other serine proteinases. FEBS Lett 229:157–160
Hunninghake GW, Davidson JM, Rennard S (1981) Elastin fragments attract macrophage precursors to diseased sites in pulmonary emphysema. Science 212:925–927
Kessenbrock K, Dau T, Jenne DE (2010) Tailor-made inflammation: how neutrophil serine proteases modulate the inflammatory response. J Mol Med 89:23–28
Korkmaz B, Horwitz MS, Jenne DE et al (2011) Neutrophil elastase, proteinase 3, and cathepsin G as therapeutic targets in human diseases. Pharmacol Rev 62:726–759
Pham CT (2008) Neutrophil serine proteases fine-tune the inflammatory response. Int J Biochem Cell Biol 40:1317–1333
Kramps JA, Franken C, Dijkman JH (1984) ELISA for quantitative measurement of low-molecular-weight bronchial protease inhibitor in human sputum. Am Rev Respir Dis 129:959–963
Vogelmeier C, Hubbard RC, Fells GA et al (1991) Anti-neutrophil elastase defense of the normal human respiratory epithelial surface provided by the secretory leukoprotease inhibitor. J Clin Invest 87:482–488
Schalkwijk J, Wiedow O, Hirose S (1999) The trappin gene family: proteins defined by an N-terminal transglutaminase substrate domain and a C-terminal four-disulphide core. Biochem J 340:569–577
Hochstrasser K, Albrecht GJ, Schonberger OL et al (1981) An elastase-specific inhibitor from human bronchial mucus: isolation and characterization. Hoppe-Seyler’s Z Physiol Chem 362:1369–1375
Takubo Y, Guerassimov A, Ghezzo H et al (2002) Alpha1-antitrypsin determines the pattern of emphysema and function in tobacco smoke-exposed mice: parallels with human disease. Am J Respir Crit Care Med 166:1596–1603
Janoff A, Carp H, Lee DK (1980) Inactivation of alpha 1-proteinase inhibitor and bronchial mucous proteinase inhibitor by cigarette smoke in vitro and in vivo. Bull Eur Physiopathol Respir 16:321–340
Simon RH, Gross TJ, Edwards JA et al (1992) Fibrin degradation by rat pulmonary alveolar epithelial cells. Am J Physiol 262:L482–L488
Blasi F, Carmeliet P (2002) uPAR: a versatile signaling orchestrator. Nat Rev Mol Cell Biol 3:932–943
Zhang Y, Xiao W, Jiang Y et al (2012) Levels of components of the urokinase-type plasminogen activator system are related to chronic obstructive pulmonary disease parenchymal destruction and airway remodelling. J Int Med Res 40:976–985
Ostridge K, Williams N, Kim V et al (2016) Relationship between pulmonary matrix metalloproteinases and quantitative CT markers of small airways disease and emphysema in COPD. Thorax 71:126–132
Navratilova Z, Kolek V, Petrek M (2016) Matrix metalloproteinases and their inhibitors in chronic obstructive pulmonary disease. Arch Immunol Ther Exp (Warsz) 64:177–193
Churg A, Wang RD, Tai H et al (2003) Macrophage metalloelastase mediates acute cigarette smoke-induced inflammation via tumor necrosis factor-alpha release. Am J Respir Crit Care Med 167:1083–1089
Lagente V, Le Quement C, Boichot E (2009) Macrophage metalloelastase (MMP-12) as a target for inflammatory respiratory diseases. Expert Opin Ther Targets 13:287–295
Aida Y, Shibata Y, Abe S et al (2014) Inhibition of elastase-pulmonary emphysema in dominant-negative MafB transgenic mice. Int J Biol Sci 10:882–894
Haq I, Lowrey GE, Kalsheker N et al (2011) Matrix metalloproteinase-12 (MMP-12) SNP affects MMP activity, lung macrophage infiltration and protects against emphysema in COPD. Thorax 66:970–976
Navratilova Z, Zatloukal J, Kriegova E et al (2012) Simultaneous up-regulation of matrix metalloproteinases 1, 2, 3, 7, 8, 9 and tissue inhibitors of metalloproteinases 1, 4 in serum of patients with chronic obstructive pulmonary disease. Respirology 17:1006–1012
Raulo SM, Sorsa TA, Kiili MT et al (2001) Evaluation of collagenase activity, matrix metalloproteinase-8, and matrix metalloproteinase-13 in horses with chronic obstructive pulmonary disease. Am J Vet Res 62:1142–1148
Roderick JT, Cheryl LF, Laura M et al (2006) Matrix metalloproteinases promote inflammation and fibrosis in asbestos-induced lung injury in mice. Am J Respir Cell Mol Biol 35:289–297
Deshmukh HS, Shaver C, Case LM et al (2008) Acrolein-activated matrix metalloproteinase-9 contributes to persistent mucin production. Am J Respir Cell Mol Biol 38:446–454
Lim S, Roche N, Oliver BG et al (2000) Balance of matrix metalloprotease-9 and tissue inhibitor of metalloprotease-1 from alveolar macrophages in cigarette smokers. Regulation by interleukin-10. Am J Respir Crit Care Med 162:1355–1360
Deshmukh HS, McLachlan A, Atkinson JJ et al (2009) Matrix metalloproteinase-14 mediates a phenotypic shift in the airways to increase mucin production. Am J Respir Crit Care Med 180:834–845
Quan TE, Cowper SE, Bucala R (2006) The role of circulating fibrocytes in fibrosis. Curr Rheumatol Rep 8:145–150
Katzenstein AL, Myers JL (1998) Idiopathic pulmonary fibrosis: clinical relevance of pathologic classification. Am J Respir Crit Care Med 157:1301–1315
Moles A, Tarrats N, Fernández-Checa JC et al (2009) Cathepsins B and D drive hepatic stellate cell proliferation and promote their fibrogenic potential. Hepatology 49:1297–1307
Imai K, Hiramatsu A, Fukushima D et al (1997) Degradation of decorin by matrix metalloproteinases: identification of the cleavage sites, kinetic analyses and transforming growth factor-beta1 release. Biochem J 322:809–814
van den Brûle S, Misson P, Bühling F et al (2005) Overexpression of cathepsin K during silica-induced lung fibrosis and control by TGF-beta. Respir Res 6:84
Scabilloni JF, Wang L, Antonini JM et al (2005) Matrix metalloproteinase induction in fibrosis and fibrotic nodule formation due to silica inhalation. Am J Physiol Lung Cell Mol Physiol 288:L709–L717
Perdereau C, Godat E, Maurel MC et al (2006) Cysteine cathepsins in human silicotic bronchoalveolar lavage fluids. Biochim Biophys Acta 1762:351–356
Rowe SM, Miller S, Sorscher EJ (2005) Cystic fibrosis. N Engl J Med 352:1992–2001
Gibson GJ, Loddenkemper R, Lundbäck B (2013) Respiratory health and disease in Europe: the new European Lung White Book. Eur Respir J 42:559–563
Tilly BC, Winter MC, Ostedgaard LS et al (1992) Cyclic AMP-dependent protein kinase activation of cystic fibrosis transmembrane conductance regulator chloride channels in planar lipid bilayers. J Biol Chem 267:9470–9473
Downey DG, Bell SC, Elborn JS (2009) Neutrophils in cystic fibrosis. Thorax 64:81–88
Kreda SM, Davis CW, Rose MC (2012) CFTR, mucins, and mucus obstruction in cystic fibrosis. Cold Spring Harb Perspect Med 2:a009589
Voynow JA, Rubin BK (2009) Mucins, mucus, and sputum. Chest 135:505–512
Shao MX, Nadel JA (2005) Neutrophil elastase induces MUC5AC mucin production inhuman airway epithelial cells via a cascade involving protein kinase C, reactive oxygen species, and TNF-alpha-converting enzyme. J Immunol 175:4009–4016
Song JS, Cho KS, Yoon HK et al (2005) Neutrophil elastase causes MUC5AC mucin synthesis via EGF receptor, ERK and NF-kB pathways in A549 cells. Korean J Intern Med 20:275–283
Fischer BM, Voynow JA (2002) Neutrophil elastase induces MUC5AC gene expression in airway epithelium via a pathway involving reactive oxygen species. Am J Respir Cell Mol Biol 26:447–452
Amitani R, Wilson R, Rutman A (1991) Effects of human neutrophil elastase and Pseudomonas aeruginosa proteinases on human respiratory epithelium. Am J Respir Cell Mol Biol 4:26–32
Mall MA (2008) Role of cilia, mucus, and airway surface liquid in mucociliary dysfunction: lessons from mouse models. J Aerosol Med Pulm Drug Deliv 21:13–24
Åstrand AB, Hemmerling M, Root J et al (2015) Linking increased airway hydration, ciliary beating, and mucociliary clearance through ENaC inhibition. Am J Physiol Lung Cell Mol Physiol 308:L22–L32
Gaggar A, Hector A, Bratcher PE et al (2011) The role of matrix metalloproteinases in cystic fibrosis lung disease. Eur Respir J 38:721–727
Gaggar A, Li Y, Weathington N et al (2007) Matrix metalloprotease-9 dysregulation in lower airway secretions of cystic fibrosis patients. Am J Physiol Lung Cell Mol Physiol 293:L96–L104
Gaggar A, Jackson PL, Noerager BD et al (2008) A novel proteolytic cascade generates an extracellular matrix-derived chemoattractant in chronic neutrophilic inflammation. J Immunol 180:5662–5669
Matthew S, Twigg SB, Philip L et al (2015) The role of serine proteases and antiproteases in the cystic fibrosis lung. Mediat Inflamm 2015:10
Kim HY, DeKruyff RH, Umetsu DT (2010) The many paths to asthma: phenotype shaped by innate and adaptive immunity. Nat Immunol 11:577–584
Braman SS (2006) The global burden of asthma. Chest 130:4S–12S
Barnes PJ (2008) Immunology of asthma and chronic obstructive pulmonary disease. Nat Rev Immunol 8:183–192
Holgate ST (2008) Pathogenesis of asthma. Clin Exp Allergy 38:872–897
Agrawal DK, Shao Z (2010) Pathogenesis of allergic airway inflammation. Curr Allergy Asthma Rep 10:39–48
Broide DH (2008) Immunologic and inflammatory mechanisms that drive asthma progression to remodeling. J Allergy Clin Immunol 121:560–570
Cataldo D, Munaut C, Noel A et al (2000) MMP2 and MMP9 linked gelatinolytic activity in the sputum from patients with asthma and chronic obstructive pulmonary disease. Int Arch Allergy Immunol 123:259–267
Corry DB, Rishi K, Kanellis J et al (2002) Decreased allergic lung inflammatory cell egression and increased susceptibility to asphyxiation in MMP2 deficiency. Nat Immunol 3:347–353
Greenlee KJ, Corry DB, Engler DA et al (2006) Proteomic identification of in vivo substrates for matrix metalloproteinases 2 and 9 reveals a mechanism for resolution of inflammation. J Immunol 177:7312–7321
Greenlee KJ, Werb Z, Kheradmand F (2007) Matrix metalloproteinases in lung: multiple, multifarious, and multifaceted. Physiol Rev 87:69–98
McMillan SJ, Kearley J, Campbell JD (2004) Matrix metalloproteinase9 deficiency results in enhanced allergen induced airway inflammation. J Immunol 172:2586–2594
Tsai YS, Tseng YT, Chen PS et al (2016) Protective effects of elafin against adult asthma. Allergy Asthma Proc 37:15–34
Rijken DC, Sakharov DV (2001) Basic principles in thrombolysis: regulatory role of plasminogen. Thromb Res 103:S41–S49
Pesci A, Foresi A, Bertorelli G et al (1993) Histochemical characteristics and degranulation of mast cells in epithelium and lamina propria of bronchial biopsies from asthmatic and normal subjects. Am Rev Respir Dis 147:684–689
Cho SH, Tam SW, Demissie-Sanders S et al (2000) Production of plasminogen activator inhibitor-1 by human mast cells and its possible role in asthma. J Immunol 165:3154–3161
Cho SH, Lee SH, Kato A et al (2015) Cross-talk between mast cells and bronchial epithelial cells in plasminogen activator inhibitor-1 production via transforming growth factor β1. Am J Respir Cell Mol Biol 52:88–95
Vignola AM, Riccobono L, Mirabella A et al (1998) Sputum metalloproteinase-9/tissue inhibitor of metalloproteinase-1 ratio correlated with airflow obstruction in asthma and chronic bronchitis. Am J Respir Crit Care Med 158:1945–1950
Mautino G, Henriquet C, Gougat C et al (1999) Increased expression of tissue inhibitor of metalloproteinase-1 and loss of correlation with matrix metalloproteinase-9 by macrophages in asthma. Lab Invest 79:39–47
Gibson PG, Simpson JL, Saltos N (2001) Heterogeneity of airway inflammation in persistent asthma. Chest 119:1329–1336
Van Den Steen PE, Proost P, Wuyts A et al (2000) Neutrophil gelatinase B potentiates interleukin-8 tenfold by aminoterminal processing, whereas it degrades CTAP-III, PF-4, and GRO—and leaves RANTES and MCP-2 intact. Blood 96:2673–2681
George L, Brightling CE (2016) Eosinophilic airway inflammation: role in asthma and chronic obstructive pulmonary disease. Ther Adv Chronic Dis 7:34–51
Renauld JC (2001) New insights into the role of cytokines in asthma. J Clin Pathol 54:577–589
Hodge S, Hodge G, Holmes M et al (2005) Increased airway epithelial and T-cell apoptosis in COPD remains despite smoking cessation. Eur Respir J 25:447–454
Lokau J, Nitz R, Agthe M et al (2016) Proteolytic cleavage governs interleukin-11 trans signaling. Cell Rep 14:1761–1773
Trautmann A, Kruger K, Akdis M et al (2005) Apoptosis and loss of adhesion of bronchial epithelial cells in asthma. Int Arch Allergy Immunol 138:142–150
Kuwano K (2007) Epithelial cell apoptosis and lung remodeling. Cell Mol Immunol 4:419–429
Solarewicz-Madejek K, Basinski TM, Crameri R et al (2009) T cells and eosinophils in bronchial smooth muscle cell death in asthma. Clin Exp Allergy 39:845–855
Lieberman J (2003) The ABCs of granule-mediated cytotoxicity: new weapons in the arsenal. Nat Rev Immunol 3:361–370
Lopez M, Salvaggio JE (1985) Mold-sensitive asthma. Clin Rev Allergy 3:183–196
Agarwal R (2011) Severe asthma with fungal sensitization. Curr Allergy Asthma Rep 11:403–413
Downs SH, Mitakakis TZ, Marks GB et al (2001) Clinical importance of Alternaria exposure in children. Am J Respir Crit Care Med 164:455–459
Kouzaki H, Iijima K, Kobayashi T et al (2011) The danger signal, extracellular ATP, is a sensor for an airborne allergen and triggers IL-33 release and innate Th2-type responses. J Immunol 186:4375–4387
Prefontaine D, Nadigel J, Chouiali F et al (2010) Increased IL-33 expression by epithelial cells in bronchial asthma. J Allergy Clin Immunol 125:752–754
Prefontaine D, Lajoie-Kadoch S, Foley S et al (2009) Increased expression of IL-33 in severe asthma: evidence of expression by airway smooth muscle cells. J Immunol 183:5094–5103
Snelgrove RJ, Gregory LG, Peiró T et al (2014) Alternaria-derived serine protease activity drives IL-33-mediated asthma exacerbations. J Allergy Clin Immunol 134:583–592
Park MK, Cho MK, Kang SA et al (2014) Acanthamoeba protease activity promotes allergic airway inflammation via protease-activated receptor 2. PLoS ONE 9:e92726
Widmer F, Hayes PJ, Whittaker RG et al (2000) Substrate preference profiles of proteases released by allergenic pollens. Clin Exp Allergy 30:571–576
Hammad H, Lambrecht BN (2008) Dendritic cells and epithelial cells: linking innate and adaptive immunity in asthma. Nat Rev Immunol 8:193–204
Matheson N, Schmidt J, Travis J (1995) Isolation and properties of an angiotensin II-cleaving peptidase from mesquite pollen. Am J Respir Cell Mol Biol 12:441–448
Medical Section of the American Lung Association (1998) The official conference report of the American Thoracic Society and Approved by the ATS board of Directors. Respiratory health hazards in agriculture. Am J Respir Crit Care Med 158:S1–S76
Kirkhorn SR, Garry VF (2000) Agricultural lung diseases. Environ Health Perspect 108:705–712
Langley RL (2011) Consequences of respiratory exposures in the farm environment. N C Med J 72:477–480
Poole JA, Romberger DJ (2012) Immunological and inflammatory responses to organic dust in agriculture. Curr Opin Allergy Clin Immunol 12:126–132
May S, Romberger DJ, Poole JA (2012) Respiratory health effects of large animal farming environments. J Toxicol Environ Health B Crit Rev 15:524–541
Dodmane PR, Schulte NA, Heires AJ et al (2011) Airway epithelial epidermal growth factor receptor mediates hogbarn dust-induced cytokine release but not Ca2 response. Am J Respir Cell Mol Biol 45:882–888
Romberger DJ, Heires AJ, Nordgren TM et al (2015) Proteases in agricultural dust induce lung inflammation through PAR-1 and PAR-2 activation. Am J Physiol Lung Cell Mol Physiol 309:L388–L399
Kouzaki H, O’Grady SM, Lawrence CB et al (2009) Proteases induce production of thymic stromal lymphopoietin by airway epithelial cells through protease-activated receptor-2. J Immunol 183:1427–1434
Page K, Strunk VS, Hershenson MB (2003) Cockroach proteases increase IL-8 expression in human bronchial epithelial cells via activation of protease activated receptor (PAR)-2 and extracellular-signal-regulated kinase. J Allergy Clin Immunol 112:1112–1118
Post S, Heijink IH, Petersen AH et al (2014) Protease-activated receptor-2 activation contributes to house dust mite-induced IgE responses in mice. PLoS ONE 9:e91206
Liu C, Li Q, Zhou X et al (2013) Human airway trypsin-like protease induces mucin5AC hypersecretion via a protease activated receptor 2-mediated pathway in human airway epithelial cells. Arch Biochem Biophys 535:234–240
Matsushima R, Takahashi A, Nakaya Y et al (2006) Human airway trypsin-like protease stimulates human bronchial fibroblast proliferation in a protease-activated receptor-2-dependent pathway. Am J Physiol Lung Cell Mol Physiol 290:L385–L395
Asokananthan N, Graham PT, Stewart DJ et al (2002) House dust mite allergens induce proinflammatory cytokines from respiratory epithelial cells: the cysteine protease allergen, Derp 1, activates protease-activated receptor (PAR)-2 and inactivates PAR-1. J Immunol 169:4572–4578
Bernton H, Brown H (1964) Insect allergy-preliminary studies of the cockroach. J Allergy 35:506–513
Arruda LK, Vailes LD, Ferriani VPL et al (2001) Cockroach allergens and asthma. Curr Rev Allergy Clin Immunol 107:419–428
Crain EF, Walter M, O’Connor GT et al (2002) Home and allergic characteristics of children with asthma in seven U.S. urban communities and design of an environmental intervention: the inner-city asthma study. Env Health Persp 110:939–945
Bhat RK, Page K, Tan A et al (2003) German cockroach extract increases bronchial epithelial cell interleukin-8 expression. Clin Exp Allergy 33:35–42
Hughes VS, Page K (2007) German cockroach frass proteases cleave promatrix metalloproteinase-9. Exp Lung Res 33:135–150
Page K, Ledford JR, Zhou P et al (2010) Mucosal sensitization to German cockroach involves protease-activated receptor-2. Respir Res 11:62
Page K (2012) Role of cockroach proteases in allergic disease. Curr Allergy Asthma Rep 12:448–455
Young RP, Hopkins RJ (2011) How the genetics of lung cancer may overlap with COPD. Respirology 16:1047–1055
Brzóska K, Bartłomiejczyk T, Sochanowicz B et al (2014) Matrix metalloproteinase 3 polymorphisms as a potential marker of enhanced susceptibility to lung cancer in chronic obstructive pulmonary disease subjects. Ann Agric Environ Med 21:546–551
Safranek J, Pesta M, Holubec L et al (2009) Expression of MMP-7, MMP-9, TIMP-1 and TIMP-2 mRNA in lung tissue of patients with non-small cell lung cancer (NSCLC) and benign pulmonary disease. Anticancer Res 29:2513–2517
Fang S, Jin X, Wang R et al (2005) Polymorphisms in the MMP1 and MMP3 promoter and non-small cell lung carcinoma in North China. Carcinogenesis 26:481–486
Turpeenniemi-Hujanen T (2005) Gelatinases (MMP-2 and-9) and their natural inhibitors as prognostic indicators in solid cancers. Biochimie 87:287–297
Chen MH, Cui SX, Cheng YN et al (2008) Galloyl cyclic-imide derivative CH1104I inhibits tumour invasion through suppressing matrix metalloproteinase activity. Anticancer Drugs 19:957–965
Qian Q, Wang Q, Zhan P et al (2010) The role of matrix metalloproteinase 2 on the survival of patients with non-small cell lung cancer: a systematic review with meta-analysis. Cancer Invest 28:661–669
Pulukuri SM, Rao JS (2008) Matrix metalloproteinase-1 promotes prostate tumour growth and metastasis. Int J Oncol 32:757–765
Houghton AM, Grisolano JL, Baumann ML et al (2006) Macrophage elastase (matrix metalloproteinase-12) suppresses growth of lung metastases. Cancer Res 66:6149–6155
Nie GY, Hampton A, Li Y et al (2003) Identification and cloning of two isoforms of human high-temperature requirement factor A3 (HtrA3), characterization of its genomic structure and comparison of its tissue distribution with HtrA1 and HtrA2. Biochem J 371:39–48
Clausen T, Southan C, Ehrmann M (2002) The HtrA family of proteases: implications for protein composition and cell fate. Mol Cell 10:443–455
Esposito V, Campioni M, De Luca A et al (2006) Analysis of HtrA1 serine protease expression in human lung cancer. Anticancer Res 26:3455–3459
Oka C, Tsujimoto R, Kajikawa M et al (2004) HtrA1 serine protease inhibits signaling mediated by TGF, family proteins. Development 131:1041–1053
Hirotaka O, Takahashi T (2002) Genetic alterations of multiple tumor suppressors and oncogenes in the carcinogenesis and progression of lung cancer. Oncogene 21:7421–7434
Shen ZT, Shen JS, Ji XQ et al (2016) TGF-β1 -rs1982073 polymorphism contributes to radiation pneumonitis in lung cancer patients: a meta-analysis. J Cell Mol Med. doi:10.1111/jcmm.12933
Chien J, Ota T, Aletti G et al (2009) Serine protease HtrA1 associates with microtubules and inhibits cell migration. Mol Cell Biol 2009:4177–4187
Cilenti L, Kyriazis GA, Soundarapandian MM et al (2005) Omi/HtrA2 protease mediates cisplatin-induced cell death in renal cells. Am J Physiol Renal Physiol 288:371–379
Egger L, Schneider J, Rhême C et al (2003) Serine proteases mediate apoptosis-like cell death and phagocytosis under caspase-inhibiting conditions. Cell Death Differ 10:1188–1203
Leist M, Jaattela M (2001) Four deaths and a funeral: from caspases to alternative mechanisms. Nat Rev Mol Cell Biol 2:589–598
Wolf BB, Green DR (2002) Apoptosis: letting slip the dogs of war. Curr Biol 5:R177–R179
Jorgensen ED, Dozmorov I, Frank MB et al (2004) Global gene expression analysis of human bronchial epithelial cells treated with tobacco condensates. Cell Cycle 3:1154–1168
Beleford D, Liu Z, Rattan R et al (2010) Methylation induced gene silencing of HtrA3 in smoking-related lung cancer. Clin Cancer Res 16:398–409
Fujii-Kuriyama Y, Mimura J (2005) Molecular mechanisms of AhR functions in the regulation of cytochrome P450 genes. Biochem Biophys Res Commun 338:311–317
Safe S, Wang F, Porter W et al (1998) Ah receptor agonists as endocrine disruptors: antiestrogenic activity and mechanisms. Toxicol Lett 102:343–347
Hockings JK, Thorne PA, Kemp MQ et al (2006) The ligand status of the aromatic hydrocarbon receptor modulates transcriptional activation of BRCA-1 promoter by estrogen. Cancer Res 66:2224–2232
Chien J, Campioni M, Shridhar V et al (2009) HtrA serine proteases as potential therapeutic targets in cancer. Curr Cancer Drug Targets 9:451–468
Korkmaz B, Moreau T, Gauthier F (2008) Neutrophil elastase, proteinase 3 and cathepsin G: physicochemical properties, activity and physiopathological functions. Biochimie 90:227–242
Reece ST, Loddenkemper C, Askew DJ et al (2010) Serine protease activity contributes to control of Mycobacterium tuberculosis in hypoxic lung granulomas in mice. J Clin Invest 120:3365–3376
Elkington P, Shiomi T, Breen R et al (2011) MMP-1 drives immunopathology in human tuberculosis and transgenic mice. J Clin Invest 121:1827–1833
Walker NF, Clark SO, Oni T et al (2012) Doxycycline and HIV infection suppress tuberculosis-induced matrix metalloproteinases. Am J Resp Crit Care Med 185:989–997
Elkington PT, Armiento JM, Friedland JS (2011) Tuberculosis immunopathology: the neglected role of extracellular matrix destruction. Sci Transl Med 3:71–76
Seddon J, Kasprowicz V, Walker NF et al (2013) Procollagen III N terminal propeptide and desmosine are released by matrix destruction in pulmonary tuberculosis. J Infect Dis 208:1571–1579
Ugarte-Gil CA, Elkington P, Gilman RH et al (2013) Induced sputum MMP-1, -3 and -8 concentrations during treatment of tuberculosis. PLoS ONE 8:e61333
Singh S, Saraiva L, Elkington PT et al (2014) Regulation of matrix metalloproteinase-1, -3, and -9 in Mycobacterium tuberculosis-dependent respiratory networks by the rapamycin-sensitive PI3K/p70(S6K) cascade. FASEB J 28:85–93
Leco KJ, Waterhouse P, Sanchez OH et al (2001) Spontaneous air space enlargement in the lungs of mice lacking tissue inhibitor of metalloproteinases-3 (TIMP-3). J Clin Invest 108:817–829
Kübler A, Luna B, Larsson C et al (2015) Mycobacterium tuberculosis dysregulates MMP/TIMP balance to drive rapid cavitation and unrestrained bacterial proliferation. J Pathol 235:431–444
Peiris JS, Yuen KY, Osterhaus AD et al (2003) The severe acute respiratory syndrome. N Engl J Med 349:2431–2441
Peiris JS, Guan Y, Yuen KY (2004) Severe acute respiratory syndrome. Nat Med 10:S88–S97
Hudson LD, Milberg JA, Anardi D et al (1995) Clinical risks for development of the acute respiratory distress syndrome. Am J Respir Crit Care Med 151:293–301
Ware LB, Matthay MA (2000) The acute respiratory distress syndrome. N Engl J Med 342:1334–1349
Raiden S, Nahmod K, Nahmod V et al (2002) Nonpeptide antagonists of AT1 receptor for angiotensin II delay the onset of acute respiratory distress syndrome. J Pharmacol Exp Ther 303:45–51
Marshall RP, Webb S, Bellingan GJ et al (2002) Angiotensin converting enzyme insertion/deletion polymorphism is associated with susceptibility and outcome in acute respiratory distress syndrome. Am J Respir Crit Care Med 166:646–650
Jerng JS, Yu CJ, Wang HC et al (2006) Polymorphism of the angiotensin-converting enzyme gene affects the outcome of acute respiratory distress syndrome. Crit Care Med 34:1001–1006
Hamming I, Timens W, Bulthuis ML et al (2004) Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol 203:631–637
Kuba K, Imai Y, Rao S et al (2005) A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat Med 11:875–879
Guyot N, Zani ML, Berger P et al (2005) Proteolytic susceptibility of the serine protease inhibitor trappin-2 (pre-elafin): evidence for tryptase-mediated generation of elafin. Biol Chem 386:391–399
Molhuizen HO, Alkemade HA, Zeeuwen PL et al (1993) SKALP/elafin: an elastase inhibitor from cultured human keratinocytes. Purification, cDNA sequence, and evidence for transglutaminase cross-linking. J Biol Chem 268:12028–12032
Nara K, Ito S, Ito T et al (1994) Elastase inhibitor elafin is a new type of proteinase inhibitor which has a transglutaminase-mediated anchoring sequence termed ‘cementoin’. J Biochem 115:441–448
Petty TL (1991) Protease mechanisms in the pathogenesis of acute lung injury. Ann N Y Acad Sci 624:267–277
Wang Z, Beach D, Su L et al (2008) A genome-wide expression analysis in blood identifies pre-elafin as a biomarker in ARDS. Am J Respir Cell Mol Biol 38:724–732
Wang Z, Chen F, Zhai R et al (2009) Plasma neutrophil elastase and elafin imbalance is associated with acute respiratory distress syndrome (ARDS) development. PLoS ONE 4:e4380
Tejera P, Wang Z, Zhai R et al (2009) Genetic polymorphisms of peptidase inhibitor 3 (elafin) are associated with ARDS. Am J Respir Cell Mol Biol 41:696–704
Sixt SU, Adamzik M, Spyrka D et al (2009) Alveolar extracellular 20S proteasome in patients with acute respiratory distress syndrome. Am J Respir Crit Care Med 179:1098–1106
Roth GA, Moser B, Krenn C et al (2005) Heightened levels of circulating 20S proteasome in critically ill patients. Eur J Clin Invest 35:399–403
Kerrin A, Weldon S, Chung AHK et al (2013) Proteolytic cleavage of elafin by 20S proteasome may contribute to inflammation in acute lung injury. Thorax 68:315–321
Merritt TA, Cochrane CG, Holcomb K et al (1983) Elastase and alpha 1-proteinase inhibitor activity in tracheal aspirates during respiratory distress syndrome. Role of inflammation in the pathogenesis of bronchopulmonary dysplasia. Clin Invest 72:656–666
Janoff A (1985) Elastases and emphysema. Current assessment of the protease antiprotease hypothesis. Am Rev Respir Dis 132:417–433
Taggart CC, Greene CM, Carroll TP et al (2005) Elastolytic proteases: inflammation resolution and dysregulation in chronic infective lung disease. Am J Respir Crit Care Med 171:1070–1076
Kawabata K, Suzuki M, Sugitani M et al (1991) ONO-5046, a novel inhibitor of human neutrophil elastase. Biochem Biophys Res Commun 177:814–820
Inoue Y, Omodani T, Shiratake R et al (2009) Development of a highly water-soluble peptide-based human neutrophil elastase inhibitor; AE-3763 for treatment of acute organ injury. Bioorg Med Chem 17:7477–7486
Chowdhury D, Lieberman J (2008) Death by a thousand cuts: granzyme pathways ofprogrammed cell death. Annu Rev Immunol 26:389–420
Boivin WA, Cooper DM, Hiebert PR et al (2009) Intracellular versus extracellular granzyme B in immunity and disease: challenging the dogma. Lab Invest 89:1195–1220
Metkar SS, Menaa C, Pardo J et al (2008) Human and mouse granzyme A induce a proinflammatory cytokine response. Immunity 29:720–733
Hendel A, Hiebert PR, Boivin WA et al (2010) Granzymes in age-related cardiovascular and pulmonary diseases. Cell Death Differ 17:596–606
Pardo J, Aguilo JI, Anel A et al (2009) The biology of cytotoxic cell granule exocytosis pathway: granzymes have evolved to induce cell death and inflammation. Microbes Infect 11:452–459
García-Laorden MI, Stroo I, Blok DC et al (2016) Granzymes A and B regulate the local inflammatory response during Klebsiella pneumoniae pneumonia. J Innate Immun 8:258–268
Latge JP (1999) Aspergillus fumigatus and aspergillosis. Clin Microbiol Rev 12:310–350
Segal BJ (2009) Aspergillosis. N Engl J Med 360:1870–1884
Woodcock AA, Steel N, Moore CB et al (2006) Fungal contamination of bedding. Allergy 61:140–142
Moutaouakil M, Monod M, Prevost MC et al (1993) Identification of the 33-kDa alkaline protease of Aspergillus fumigates in vitro and in vivo. J Med Microbiol 39:393–399
Kunert J, Kopecek P et al (2000) Multiple forms of the serine proteinase ALP of Aspergillus fumigatus. Mycoses 43:339–347
Monod M, Paris S, Sanglard D et al (1993) Isolation and characterization of a secreted metalloprotease of Aspergillus fumigatus. Infect Immun 61:4099–5104
Reichard U, Eiffert H, Ruchel R (1994) Purification and characterization of an extracellular aspartic proteinase of Aspergillus fumigatus. J Med Vet Mycol 32:427–436
Kolattukudy PE, Lee JD, Rogers LM et al (1993) Evidence for possible involvement of an elastolytic serine protease in aspergillosis. Infect Immun 61:2357–2368
Markaryan A, Morozova I, Yu H et al (1994) Purification and characterization of an elastinolytic metalloprotease from Aspergillus fumigatus and immunoelectron microscopic evidence of secretion of this enzyme by the fungus invading the murine lung. Infect Immun 62:2149–2157
McDonagh A, Fedorova ND, Crabtree J et al (2008) Sub-telomere directed gene expression during initiation of invasive aspergillosis. PLoS Pathog 4:e1000154
Bouchara JP, Sanchez M, Esnault K et al (1999) Interactions between Aspergillus fumigatus and host matrix protein. Contrib Microbiol 2:167–181
Asano Y (2010) Future treatments in systemic sclerosis. J Dermatol 37:54–70
Roy R, Wewer UM, Zurakowski D et al (2004) ADAM 12 cleaves extracellular matrix proteins and correlates with cancer status and stage. J Biol Chem 279:51323–51330
Shi Z, Xu W, Loechel F et al (2000) ADAM 12, a disintegrin metalloprotease, interacts with insulin-like growth factor-binding protein-3. J Biol Chem 275:18574–18580
Loechel F, Fox JW, Murphy G et al (2000) ADAM 12-S cleaves IGFBP-3 and IGFBP-5 and is inhibited by TIMP-3. Biochem Biophys Res Commun 278:511–515
Jones JI, Clemmons DR et al (1995) Insulin-like growth factors and their binding proteins: biological actions. Endocr Rev 16:3–34
Harsha A, Stojadinovic O, Brem H et al (2008) ADAM12: a potential target for the treatment of chronic wounds. J Mol Med (Berl) 86:961–969
Coalson JJ (2003) Pathology of new bronchopulmonary dysplasia. Semin Neonatol 8:73–81
Madurga A, Mizíková I, Ruiz-Camp J et al (2013) Recent advances in late lung development and the pathogenesis of bronchopulmonary dysplasia. Am J Physiol Lung Cell Mol Physiol 305:893–905
Bruce MC, Schuyler M, Martin RJ et al (1992) Risk factors for the degradation of lung elastic fibers in the ventilated neonate. Implications for impaired lung development in bronchopulmonary dysplasia. Am Rev Respir Dis 146:204–212
Altiok O, Yasumatsu R, Bingol-Karakoc G et al (2006) Imbalance between cysteine proteases and inhibitors in a baboon model of bronchopulmonary dysplasia. Am J Respir Crit Care Med 173:318–326
Davies PL, Spiller OB, Beeton ML et al (2010) Relationship of proteinases and proteinase inhibitors with microbial presence in chronic lung disease of prematurity. Thorax 65:246–251
Bry K, Hogmalm A, Backstrom E (2010) Mechanisms of inflammatory lung injury in the neonate: lessons from a transgenicmouse model of bronchopulmonary dysplasia. Semin Perinatol 34:211–221
Chilosi M, Pea M, Martignoni G et al (2009) Cathepsin-k expression in pulmonary lymphangioleiomyomatosis. Mod Pathol 22:161–166
Smith GN Jr, Mickler EA, Payne KK et al (2007) Lung transplant metalloproteinase levels are elevated prior to bronchiolitis obliterans syndrome. Am J Transplant 7:1856–1861
Riise GC, Ericson P, Bozinovski S et al (2010) Increased net gelatinase but not serine protease activity in bronchiolitis obliterans syndrome. J Heart Lung Transplant 29:800–807
Iwata T, Chiyo M, Yoshida S et al (2008) Lung transplant ischemia reperfusion injury: metalloprotease inhibition down-regulates exposure of type V collagen, growth-related oncogene-induced neutrophil chemotaxis, and tumour necrosis factor-a expression. Transplantation 85:417–426
Khatwa UA, Kleibrink BE, Shapiro SD et al (2010) MMP-8 promotes polymorphonuclear cell migration through collagen barriers in obliterative bronchiolitis. J Leukocyte Biol 87:69–77
Fernandez FG, Campbell LG, Liu W et al (2005) Inhibition of obliterative airway disease development in murine tracheal allografts by matrix metalloproteinase-9 deficiency. Am J Transplant 5:671–683
Yoshida S, Iwata T, Chiyo M et al (2007) Metalloproteinase inhibition has differential effects on alloimmunity, autoimmunity, and histopathology in the transplanted lung. Transplantation 83:799–808
King RJ, Clements JA (1972) Surface active materials from dog lung. I. Method of isolation. Am J Physiol 223:707–714
LeVine AM, Kurak KE, Wright JR et al (1999) Surfactant protein-a binds group b streptococcus enhancing phagocytosis and clearance from lungs of surfactant protein-a-deficient mice. Am J Respir Cell Mol Biol 20:279–286
Crouch E, Hartshorn K, Ofek I (2000) Collectins and pulmonary innate immunity. Immunol Rev 173:52–65
Hartshorn KL, Crouch E, White MR et al (1998) Pulmonary surfactant proteins a and d enhance neutrophil uptake of bacteria. Am J Physiol 274:L958–L969
Sano H, Sohma H, Muta T et al (1999) Pulmonary surfactant protein a modulates the cellular response to smooth and rough lipopolysaccharides by interaction with cd14. J Immunol 163:387–395
Garcia-Verdugo I, Descamps D, Chiganard M et al (2010) Protease/anti-protease net work and modulation of mucus production and surfactant activity. Biochimie 92:1608–1617
Murakami S, Iwaki D, Mitsuzawa H et al (2002) Surfactant protein A inhibits peptidoglycan-induced tumor necrosis factor-alpha secretion in u937 cells and alveolar macrophages by direct interaction with toll-like receptor 2. J Biol Chem 277:6830–6837
Kantyka T, Pyrc K, Gruca M et al (2013) Staphylococcus aureus proteases degrade lung surfactant protein A potentially impairing innate immunity of the lung. J Innate Immun 5:251–260
Reid KBM (1998) Interactions of surfactant protein D with pathogens, allergens and phagocytes. Biochem Biophys Acta 1408:290–295
Restrepo CI, Dong Q, Savov J et al (1999) SP-D stimulates phagocytosis of Pseudomonas aeruginosa by alveolar macrophages. Am J Respir Cell Mol Biol 21:576–585
Crouch EC (1998) Structure, biological properties, and expression of surfactant protein D (SP-D). Biochem Biophys Acta 1408:278–289
Kuan SF, Rust K, Crouch EC (1992) Interactions of surfactant protein D with bacterial lipopolysaccharides. Surfactant protein D is an Escherichia coli-binding protein in bronchoalveolar lavage. J Clin Invest 90:97–106
Madan T, Eggleton P, Kishore U et al (1997) Binding of pulmonary surfactant protein A and D to Aspergillus fumigatus conidia enhance phagocytosis and killing by human neutrophils and alveolar macrophages. Infect Immun 65:3171–3179
Persson A, Chang D, Crouch E (1990) Surfactant protein D is a divalent cation-dependent carbohydrate-binding protein. J Biol Chem 265:5755–5760
Pikaar JC, Voorhut WF, van Golde LM et al (1995) Opsonic activities of surfactant protein A and D in phagocytosis of gram-negative bacterias by alveolar macrophages. J Infect Dis 172:481–489
Birrer P, McElvaney G, Rüdelberg A et al (1994) Protease-antiprotease imbalance in the lungs of children with cystic fibrosis. Am J Respir Crit Care Med 151:207–213
von Bredow C, Birrer P, Griese M (2001) Degradation of surfactant protein A and other bronchoalveolar lavage fluid proteins in patients with cystic fibrosis. Eur Respir J 17:716–722
Beatty AL, Malloy JL, Wright JR (2005) Pseudomonas aeruginosa degrades pulmonary surfactant and increases conversion in vitro. Am J Respir Cell Mol Biol 32:128–134
Malloy JL, Veldhuizen RA, Thibodeaux BA et al (2005) Pseudomonas aeruginosa protease IV degrades surfactant proteins and inhibits surfactant host defense and biophysical functions. Am J Physiol Lung Cell Mol Physiol 288:409–418
Alcorn JF, Wright JR (2004) Degradation of pulmonary surfactant protein D by Pseudomonas aeruginosa elastase abrogates innate immune function. J Biol Chem 279:30871–30879
Mariencheck WI, Alcorn JF, Palmer SM et al (2003) Pseudomonas aeruginosa elastase degrades surfactant proteins A and D. Am J Respir Cell Mol Biol 28:528–537
Hirche TO, Crouch EC, Espinola M et al (2004) Neutrophil serine proteinases inactivate surfactant protein D by cleaving within a conserved subregion of the carbohydrate recognition domain. J Biol Chem 279:27688–27698
Liau DF, Yin NX, Huang J et al (1996) Effects of human polymorphonuclear leukocyte elastase upon surfactant proteins in vitro. Biochim Biophys Acta 1302:117–128
Griese M, Wiesener A, Lottspeich F et al (2003) Limited proteolysis of surfactant protein D causes a loss of its calcium-dependent lectin functions. Biochim Biophys Acta 1638:157–163
Burgel PR, Nadel JA et al (2004) Roles of epidermal growth factor receptor activation in epithelial cell repair and mucin production in airway epithelium. Thorax 59:992–996
Burgel PR, Nadel JA et al (2008) Epidermal growth factor receptor-mediated innate immune responses and their roles in airway diseases. Eur Respir J32:1068–1081
Hewson CA, Edbrooke MR, Johnston SL et al (2004) PMA induces the MUC5AC respiratory mucin in human bronchial epithelial cells, via PKC, EGF/TGF-alpha, Ras/Raf, MEK, ERK and Sp1-dependent mechanisms. J Mol Biol 344:683–695
Wu DY, Wu R, Chen Y et al (2007) PMA stimulates MUC5B gene expression through an Sp1-based mechanism in airway epithelial cells. Am J Respir Cell Mol Biol 37:589–597
Lundgren JD, Rieves RD, Mullol J et al (1994) The effect of neutrophil proteinase enzymes on the release of mucus from feline and human airway cultures. Respir Med 88:511–518
Kohri K, Ueki IF, Nadel JA et al (2002) Neutrophil elastase induces mucin production by ligand-dependent epidermal growth factor receptor activation. Am J Physiol Lung Cell Mol Physiol 283:531–540
Chokki M, Yamamura S, Eguchi H et al (2004) Human airway trypsin-like protease increases mucin gene expression in airway epithelial cells. Am J Respir Cell Mol Biol 30:470–478
Miki M, Nakamura Y, Takahashi A et al (2003) Effect of human airway trypsin-like protease on intracellular free Ca2+ concentration in human bronchial epithelial cells. J Med Invest 50:95–107
Englert N (2004) Fine particles and human health—a review of epidemiological studies. Toxicol Lett 149:235–242
Mukae H, Hogg JC, English D et al (2000) Phagocytosis of particulate air pollutants by human alveolar macrophages stimulates the bone marrow. Am J Physiol Lung Cell Mol Physiol 279:L924–L931
van Eeden SF, Tan WC, Suwa T et al (2001) Cytokines involved in the systemic inflammatory response induced by exposure to particulate matter air pollutants (PM(10)). Am J Respir Crit Care Med 164:826–830
Sun J (2010) Matrix metalloproteinases and tissue inhibitor of metalloproteinases are essential for the inflammatory response in cancer cells. J Signal Transduct 2010:985132
Wang J, Zhang H, Su C et al (2014) Dexamethasone ameliorates H2S-induced acute lung injury by alleviating matrix metalloproteinase-2 and -9 expression. PLoS ONE 9:e94701
Ong CW, Elkington PT, Friedland JS (2014) Tuberculosis, pulmonary cavitation and matrix metalloproteinases. Am J Respir Crit Care Med 190:9–18
Ishii T, Abboud RT, Wallace AM et al (2014) Alveolar macrophage proteinase/antiproteinase expression in lung function and emphysema. Eur Respir J 43:82–91
Kumar M, Phougat N, Ruhil S et al (2013) Genomics of chronic obstructive pulmonary disease (COPD); exploring the SNPs of protease–antiprotease pathway. Curr Genomics 14:204–213
Morales-Bárcenas R, Chirino YI, Sánchez-Pérez Y et al (2015) Particulate matter (PM10) induces metalloprotease activity and invasion in airway epithelial cells. Toxicol Lett 237:167–173
Taggart CC, Lowe GJ, Greene CM et al (2001) Cathepsin B, L, and S cleave and inactivate secretory leucoprotease inhibitor. J Biol Chem 276:33345–33352
Quinn DJ, Weldon S, Taggart CC (2010) Antiproteases as therapeutics to target inflammation in cystic fibrosis. Open Respir Med J 4:20–31
Gibbons A, McElvaney NG, Cryan SA (2010) A dry powder formulation of liposome-encapsulated recombinant secretory leukocyte protease inhibitor (rSLPI) for inhalation: preparation and characterisation. AAPS Pharm Sci Tech 11:1411–1421
Delacourt C, erigault SH, Delclaux C et al (2002) Protection against acute lung injury by intravenous or intratracheal pretreatment with EPI-HNE-4, a new potent neutrophil elastase inhibitor. Am J Respir Cell Mol Biol 26:290–297
Dunlevy FK, Martin SL, de Courcey F et al (2012) Anti-inflammatory effects of DX-890, a human neutrophil elastase inhibitor. J Cyst Fibros 11:300–304
Churg A, Wang R, Wang X et al (2007) Effect of an MMP-9/MMP-12 inhibitor on smoke-induced emphysema and airway remodelling in guinea pigs. Thorax 62:706–713
le Quémen C, Lagente V, Guénon I, Muzio V, Gillon J-Y, Boichot E (2008) Anti-inflammatory properties of MMP inhibitors in experimental models of chronic obstructive pulmonary disease and lung inflammation. In: Lagente V, Boichot E (eds) Matrix metalloproteinases in tissue remodelling and inflammation. Springer, New York, pp 57–69
Le Quément C, Guénon I, Gillon JY et al (2008) The selective MMP-12 inhibitor, AS111793 reduces airway inflammation in mice exposed to cigarette smoke. Br J Pharmacol 154:1206–1215
Jawad MU, Garamszegi N, Garamszegi SP et al (2010) Matrix metalloproteinase 1: role in sarcoma biology. PLoS ONE 5:e14250
Fanjul-Fernández M, Folgueras AR, Fueyo A et al (2013) Matrix metalloproteinase MMP-1a is dispensable for normal growth and fertility in mice and promotes lung cancer progression by modulating inflammatory responses. J Biol Chem 288:14647–14656
Acknowledgements
This work was supported by DST-SERB (Govt. of India) and also the DST-PURSE programme of the University of Kalyani.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Chakraborti, S., Sarkar, J., Pramanik, P.K., Chakraborti, T. (2017). Role of Proteases in Lung Disease: A Brief Overview. In: Chakraborti, S., Chakraborti, T., Dhalla, N. (eds) Proteases in Human Diseases. Springer, Singapore. https://doi.org/10.1007/978-981-10-3162-5_16
Download citation
DOI: https://doi.org/10.1007/978-981-10-3162-5_16
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-3161-8
Online ISBN: 978-981-10-3162-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)