Abstract
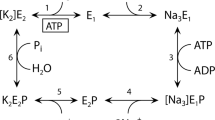
Extensive studies have been performed on structure-function relationship of the Na+ /K+-ATPase. Nevertheless, current ideas on basic processes underlying coupled ATP hydrolysis, energy transduction and cation transport are deficient in molecular details. This is mainly due to the lack of reliable data on the spatial protein structure (1). The three-dimensional structure of the Na+/K+-ATPase cannot yet be solved by X-ray analysis. However, significant information on the spatial organization of the protein molecule may be derived by other experimental approaches such as limited proteolysis, affinity modification, labeling by permeable and impermeable reagents, analysis with specific antibodies and spectroscopic methods. A combination of these techniques has been used to identify functional domains and to probe the folding of subunit polypeptide chains in the membrane. It is generally accepted that the beta subunit spans the membrane only once (1,4). In contrast, the transmembrane folding of the catalytic alpha subunit has been the subject of considerable controversy. Several topological models differ in the number and location of the membrane-spanning segments within the COOH-terminal half of the polypeptide chain and therefore the orientation of the COOH-terminus (2,3,5,7).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Horisberger J-D, Lemas V, Kraechenbuhl JP, Rossier BC (1991) Structure-function relationship of Na+/K+-ATPase. Ann Rev. Physiol 53: 565–584
Kawakami K, Noguchi S, Noda M, Takahashi H, Ohta T, Kawamura M, Nojima H, Nogano K, Hirose T, Inayama S, Hayashida H, Miyata T, Numa S (1985) Primary structure of the α-subunit of Torpedo Californica Na+/K+-ATPase deduced from cDNA sequence. Nature (London) 316: 733–736
Karlish SJD, Goldshleger R, Tal D-M, Capasso JM, Hoving S, Stein WD (1992) Identificaton of the cation binding domain of Na+/K+-ATPase. Acta Physiol Scand 146: 69–76
Modyanov N, Lutsenko S, Chertova E, Efremov R (1991) Architecture of the sodium pump molecule: probing the folding of the hydrophobic domain. In: Kaplan J, De Weer P (Eds) The Sodium Pump: Structure, Mechanism, and Regulation. pp 99–115
Ovchinnikov Yu, Arsenyan S, Broude N, Petrukhin K, Grishin A, Aldanova N, Arzamazova N, Arystarkhova E, Melkov A, Smirnov Yu, Guryev S, Monastyrskaya G, Modyanov N (1985) Primary structure of the α-subunit of the pig kidney Na+/K+-ATPase deduced from cDNA sequence. Proc Acad Sci USSR 285: 1491–1495
Ovchinnikov YuA, Arzamazova NM, Arystarkhova EA, Gevondyan NM, Aldanova NA, Modyanov NN (1987) Detailed structural analysis of exposed domains of membrane-bound Na+/K+-ATPase. A model of transmembrane arrangement. FEBS Letters 217: 269–274
Shull GE, Schwartz A, Lingrel JB (1985) Amino-acid sequence of the catalytic subunit of the Na+/K+ATPase deduced from a complementary DNA. Nature (London) 316: 691–695
Vladimirova (Arzamazova) NM, Potapenko NA, Levina NB, Modyanov NN (1991) Na+/K+-ATPase isoforms in different areas of calf brain. Biomedical science 2: 68–78
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1994 Dietrich Steinkopff Verlag GmbH & Co. KG, Darmstadt
About this chapter
Cite this chapter
Vladimirova, N.M., Efendiyev, R.E., Potapenko, N.A., Modyanov, N.N. (1994). Determination of sidedness of COOH-terminus of Na+/K+ ATPase alpha subunit by vectorial labeling. In: Bamberg, E., Schoner, W. (eds) The Sodium Pump. Steinkopff. https://doi.org/10.1007/978-3-642-72511-1_61
Download citation
DOI: https://doi.org/10.1007/978-3-642-72511-1_61
Publisher Name: Steinkopff
Print ISBN: 978-3-642-72513-5
Online ISBN: 978-3-642-72511-1
eBook Packages: Springer Book Archive