Abstract
Background
Patients on extracorporeal membrane oxygenation (ECMO) are often among the most severely ill in the intensive care unit. They are often receiving broad-spectrum antibiotics; they have multiple entry points for pathogens; and their immune system is impaired by blood circuit interaction. These factors are thought to predispose them to fungal infections. We thus aimed to evaluate the prevalence, risk factors, and prognosis of fungal infections in adults on ECMO.
Methods
We conducted a retrospective cohort study using the Extracorporeal Life Support Organization registry, which compiles data on ECMO use from hundreds of international centers. We included all adult patients from 2006 to 2016 on any mode of ECMO with either a diagnosis of fungal infection or a positive fungal culture.
Results
Our study comprised 2129 adult patients (10.8%) with fungal colonization or infection. Aspergillus involvement (colonization or infection) was present in 272 patients (1.4%), of whom 35.7% survived to hospital discharge. There were 245 patients (1.2%) with Candida invasive bloodstream infection, with 35.9% survival. Risk factors for Aspergillus involvement included solid organ transplant (OR 1.83; p = 0.008), respiratory support (OR 2.75; p < 0.001), and influenza infection (OR 2.48; p < 0.001). Risk factors for candidemia included sepsis (OR 1.60; p = 0.005) and renal replacement therapy (OR 1.55; p = 0.007). In multivariable analysis, Aspergillus involvement (OR 0.40; p < 0.001) and candidemia (OR 0.47; p < 0.001) were both independently associated with decreased survival.
Conclusions
The prevalence of Aspergillus involvement and Candida invasive bloodstream infection were not higher in patients on ECMO than what has been reported in the general intensive care population. Both were independently associated with a reduced survival. Aspergillus involvement was strongly associated with ECMO for respiratory support and influenza.
Similar content being viewed by others
Background
Critically ill patients are at increased risk of fungal infections. Candida and Aspergillus are the most frequently isolated fungi in the intensive care unit (ICU). In fact, Candida is the third most prevalent microorganism in patients with infections the ICU [1]. It is responsible for 12% of ICU-acquired bloodstream infections [2]. Nosocomial Candida bloodstream infection (CBSI) is associated with increased morbidity and mortality as well as a prolonged hospital length of stay compared with other bloodstream infections [3]. Crude mortalities of 40–60% and attributable mortalities of 5–71% have been reported in patients with Candida infection in the ICU [4,5,6]. Invasive aspergillosis (IA) has typically been described in patients with hematological malignancies and profound immunosuppression, but recent evidence has suggested that it may be present in a significant proportion of critically ill patients without these conventional risk factors [7]. Researchers in previous studies have reported a 1–2% prevalence of Aspergillus [8,9,10,11] in the respiratory tract of mechanically ventilated patients and up to 8% in those with acute respiratory distress syndrome (ARDS) [12]. In an autopsy series of patients who died of ARDS, a prevalence of 12.5% was described [13]. Even in the absence of hematological malignancy, isolation of Aspergillus in critically ill patients has been associated with mortality as high as 86% [14].
In the last decade, use of extracorporeal membrane oxygenation (ECMO) has significantly increased in the adult population [15]. It is now routinely used to support critically ill patients with severe respiratory or cardiac failure in whom conventional therapies have failed. The severity of their illness and blood circuit interactions are thought to impair their immune system. They have catheters, cannulas, and oxygenators that can become colonized with microorganisms. They are often treated with broad-spectrum antibacterial agents. These factors are thought to predispose them to fungal colonization and infection. Recently reported data suggest that up to 15% of all bloodstream infections while on ECMO are of fungal origin [16]. A 6.4% prevalence of CBSI in an Australian center has been reported [17]. In another study, yeast DNA was retrieved in 7% of oxygenators after weaning [18].
All of these studies were performed in single centers. Fungal infection rates can vary greatly between units, depending on the type of patients treated and differences in local antibiotic and antifungal prescription practices. Robust multicenter data are lacking. Therefore, the objective of the present study was to establish the prevalence, risk factors, and prognosis of fungal infection and colonization in an adult ECMO population composed of a large international cohort of patients.
Methods
Study design
We performed a retrospective cohort study using data from the Extracorporeal Life Support Organization (ELSO) registry. The registry compiles data on ECMO use in more than 300 international centers after approval by local institutional review boards. For each ECMO run, participating centers complete a standardized data sheet containing patient demographics, diagnosis and procedure information, ECMO technique, physiological and microbiological data, complications, and outcomes (see Additional file 1: Appendix 1). After approval by the ELSO Registry Committee, limited de-identified datasets are released to participating centers for research purposes without the need for further approval from individual centers. This study was conducted in accordance with the amended Declaration of Helsinki.
Population
We included all consecutive adult patients with fungal infection or colonization from January 2006 to September 2016 on any mode of extracorporeal life support. These patients were identified as those having either a diagnosis of fungal infection according to the International Classification of Diseases, Ninth Revision, or any positive fungal culture (see Additional file 1: Appendix 2).
Statistical analysis
Analyses were performed using IBM SPSS Statistics for Mac software version 24.0 (IBM, Armonk, NY, USA). Categorical variables were summarized using frequencies and percentages, and continuous variables were summarized using mean and SD. Pearson’s chi-square test was used to test for univariate associations of categorical variables, and the Mann-Whitney U test was used for continuous variables. Missing variables were excluded from analyses.
To determine factors independently associated with Aspergillus involvement (colonization or infection) and Candida bloodstream infection, we performed a multivariable logistic regression using a stepwise backward selection model with a p value less than 0.20 for inclusion and greater than 0.15 for exclusion. In order to determine if Aspergillus involvement and CBSI were independently associated with a lower survival rate, we also used a multivariable logistic regression with a stepwise backward selection model with the same conditions for inclusion and exclusion. All of the models were validated by bootstrapping with 1000 samples. The variables entered into the models can be found in Additional file 1: Appendixes 5–7.
Results
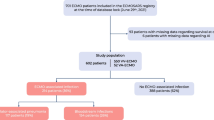
There were 19,697 adult patients in ELSO registry during the study period. Of these, 2129 had either a diagnosis of fungal infection or a positive fungal culture, resulting in a prevalence of 10.8% (95% CI 10.4–11.2%) of infection and colonization. Their mean age was 48.5 years. The majority (67.3%) of patients received ECMO for respiratory support, and a majority (57.8%) were on venovenous ECMO (Table 1). The overall survival of patients with fungus did not significantly differ from the survival of those without (49.7% vs 48.7%; p = 0.559). However, two main subgroups emerged as having a different survival in univariate analyses: patients with an aspergillosis diagnosis or any Aspergillus-positive culture (35.7%; p < 0.001) and those with Candida in blood culture (35.9%; p < 0.001) (Fig. 1). There were 272 patients with Aspergillus involvement in the registry, which resulted in a prevalence of 1.4% (95% CI 1.2–1.5%), and 245 patients with CBSI, which resulted in a prevalence of 1.4% 1.2% (95% CI 1.1–1.4%) (Table 2). Positive Candida cultures in sites other than blood did not affect survival, no matter how many sites were affected (see Additional file 1: Appendix 4).
Survival of patients with Aspergillus involvement and Candida bloodstream infection compared with the overall survival in the Extracorporeal Life Support Organization (ELSO) registry during study the period. *Statistically significant difference compared with overall survival in the ELSO registry during the study period
In multivariable analysis, male sex, hematological malignancy, influenza, solid organ transplant, and ECMO for respiratory support were associated with an increased risk of Aspergillus involvement. Increased weight, nonviral pneumonia, aspiration pneumonitis, ARDS, and acute respiratory failure were associated with a decreased risk of Aspergillus involvement (Table 3). With bootstrapping, the associations with hematological malignancies and ARDS were not statistically significant (see Additional file 1: Appendix 5). Although steroid administration was positively associated with Aspergillus involvement in univariate analysis (OR 1.50, 95% CI 1.07–2.10, p = 0.017), this association did not reach statistical significance in the multivariable backward selection model. There was no significant association between Aspergillus involvement and chronic pulmonary condition, tobacco use, tuberculosis, or human immunodeficiency virus (HIV).
Advanced age, increased weight, sepsis, and renal replacement therapy (RRT) were independent risk factors for CBSI. Although increased weight, nonviral pneumonia, intra-aortic balloon pump, and cardiopulmonary bypass were associated with the risk of CBSI in the backward selection model, the association could not be validated by bootstrapping (p > 0.05) (Table 4; see also Additional file 1: Appendix 6]. Diabetes, steroids, femoral cannulation, laparotomy, and pancreatitis were not significantly associated with CBSI. The registry does not collect information on antibiotic or parenteral nutrition use.
As hypothesized, Aspergillus involvement (OR 0.40, p < 0.001) and CBSI (OR 0.47, p < 0.001) were both independently associated with decreased survival. The other factors associated with worse survival in multivariable analysis were advanced age, hematological malignancy, acute kidney injury (AKI), HIV, ARDS, sepsis, nitric oxide, ECMO for cardiac support, and extracorporeal cardiopulmonary resuscitation. However, increased weight, aspiration pneumonitis, influenza, solid organ transplant, nonviral pneumonia, and neuromuscular blockers were independently associated with increased survival in our cohort (Table 5). The associations with AKI, sepsis, and solid organ transplant were not statistically significant with bootstrapping (see also Additional file 1: Appendix 7).
Discussion
We found a 10.8% prevalence of fungal infection or colonization in the ELSO registry, including 1.3% of patients with Aspergillus involvement and 1.2% of patients with CBSI. These prevalence rates are lower than expected in a population of mechanically ventilated critically ill patients with indwelling catheters and an ICU length of stay generally exceeding 1 week. Aspergillus involvement was associated with classic factors that cause immunosuppression, such as hematological malignancy and solid organ transplant, as well as with influenza and ECMO for respiratory support. Not surprisingly, CBSI was associated with typical risk factors such as sepsis and RRT. The survival of patients with any Aspergillus involvement (37.5%) was significantly lower than that of the rest of the ELSO population, even after adjusting for other predictive factors in multivariable analysis (OR 0.41). Similarly, CBSI was associated with poor survival (35.9%) independently (OR 0.47).
The prevalence of fungus-positive samples in general ICU patients with a length of stay more than 7 days has been reported to be as high as 59.7% with systematic sampling, much higher than in our cohort [19]. However, cultures reported in the ELSO registry are performed at the discretion of clinicians rather than systematically. It is highly probable that fungi were simply undetected in a significant number of patients. This could have resulted in a significant underestimation of the overall prevalence of fungal colonization and infection as well as of the prevalence of specific infections. Moreover, information about antifungal agent administration was not available. A more liberal use of antifungal prophylaxis in this very sick population of patients could also have contributed to a lower rate of fungus-positive samples.
The overall prevalence of any microbiological or histological evidence of Aspergillus in a medical ICU has been reported to be 6.9%, and a combined prevalence of proven, probable, or possible IA has been reported to be 5.8% [14]. These data are more in line with the 7.2% prevalence found in an Australian ICU in a review of their ECMO cases between 2005 and 2011 [20]. There was a much lower prevalence of Aspergillus involvement in the ELSO registry. The first caveat in interpreting the rates of Aspergillus involvement is that microscopic examination and culture of respiratory tract specimens have a combined sensitivity of only 50% for IA [21]. Second, discriminating between colonization and infection is always challenging without histology. Biopsies are rarely performed in ECMO patients, because they are at high risk of bleeding owing to systemic anticoagulation, platelet and coagulation factor consumption, and increased fibrinolysis associated with the extracorporeal circuit. Nonneutropenic patients most often do not display the characteristic radiological signs of IA [22]. Furthermore, many patients had respiratory failure with already abnormal chest imaging results before developing IA. Newer tests such as galactomannan and PCR of serum or bronchoalveolar lavage specimens have been developed to improve the diagnostic yield for IA [23]. However, results of such tests, if performed, were not collected in the database. Blot and colleagues have described and validated an algorithm for use in diagnosing aspergillosis in critically ill individuals [24]. However, the ELSO registry did not collect all the variables needed to apply their definitions. In addition, whether Aspergillus isolated in the lower respiratory tract of critically ill patients can be viewed as a contaminant or colonizer remains a matter of debate [25, 26].
Meersseman and colleagues reported an overall survival of 20% in patients without hematological malignancy with evidence of Aspergillus involvement [14]. The higher survival that we found could be due to the fact that ECMO patients, although acutely very ill, are younger and have fewer comorbidities than the general ICU population. Patients with Aspergillus involvement in the ELSO registry had a mean age of 46 years, and only 8.5% of them had chronic pulmonary disease, as compared with a mean age of 61 years and 42% with chronic pulmonary disease in the Meersseman study.
The strong association between influenza and Aspergillus found in our cohort has been described before in case series and case-control studies. Coinfection rates ranging from 29% to 75% have been reported [27, 28]. Proposed mechanisms for this increased susceptibility to Aspergillus invasion include both local and systemic effects [29]. Indeed, influenza induces tracheitis and bronchitis and impairs normal ciliary function [30]. The virus also impairs phagocytosis and induces anti-inflammatory cytokine production leading to T-cell dysfunction and apoptosis [31,32,33].
In recent large multicentric cohort studies of unselected patients admitted to mixed ICUs, the prevalence of candidemia ranged from 0.33% to 0.69% [3, 34]. Although the rate we found was higher than the one reported in these large studies, in a cohort of patients with severe critical illness and a long ICU length of stay, we expected a much higher rate. A prevalence of 3.3% has been reported in patients with a length of stay in the ICU more than 7 days [19]. Moreover, a prevalence of 6.4% of CBSI was found in a retrospective review of ECMO cases in an Australian center [17]. In keeping with these findings, yeast DNA has been detected in 7% of patients on oxygenators [18]. In theory, this should translate to positive fungal cultures at some point. The discrepancy could be explained by undersampling or a more liberal use of antifungal prophylaxis.
In contrast, CBSI survival was strikingly worse than previously described in the literature. León and colleagues found an overall survival of 43.4% in critically ill patients with invasive Candida infection hospitalized for more than 7 days [19]. Others have even found a survival of 57–60% in general ICU cohorts [3, 34]. Patients on ECMO could simply have a higher illness severity than these cohorts. It is also possible that the presence of foreign material makes it more difficult to eradicate Candida, as seen with prosthetic valve endocarditis.
Multiple hospitals across the globe in very different settings participate in the ELSO registry. These centers collect data on all their ECMO cases. This confers strong external validity to our study. However, the internal validity of the study is affected by multiple factors partly inherent to its retrospective nature. For instance, microbiological sampling was nonsystematic. This may have caused underestimation of the prevalence of fungal infections. This also weakened the analysis of risk factors by making them highly susceptible to observer bias. Indeed, fungal cultures may have been performed more often in patients with presenting factors that were suspected by clinicians to increase the risk of fungal infection. Moreover, limited data were available on potential confounding factors such as antifungal therapy and prophylaxis. This may have decreased certain associations with risk factors if antifungal therapy was prescribed more often in those patients. Such limitations could be avoided or minimized in a prospective study.
Conclusions
Patients on ECMO do not seem to develop fungal colonization or infection more frequently than other critically ill patients. Aspergillus involvement and CBSI were independently associated with decreased survival. CBSI mortality was higher than described in the general ICU population. Aspergillus involvement was associated with respiratory ECMO and influenza. Clinicians should maintain a high index of suspicion in this subgroup, and efforts should be made to establish an early diagnosis. It remains unclear, however, if treating the fungus would improve survival.
Abbreviations
- AKI:
-
Acute kidney injury
- ARDS:
-
Acute respiratory distress syndrome
- CBSI:
-
Candida bloodstream infection
- ECMO:
-
Extracorporeal membrane oxygenation
- ECPR:
-
Extracorporeal cardiopulmonary resuscitation
- ELSO:
-
Extracorporeal Life Support Organization
- HIV:
-
Human immunodeficiency virus
- IA:
-
Invasive aspergillosis
- ICU:
-
Intensive care unit
- RRT:
-
Renal replacement therapy
References
Vincent JL, Rello J, Marshall J, et al. International study of the prevalence and outcomes of infection in intensive care units. JAMA. 2009;302:2323–9.
Hidron AI, Edwards JR, Patel J, et al. NHSN annual update: antimicrobial-resistant pathogens associated with healthcare-associated infections: annual summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2006–2007. Infect Control Hosp Epidemiol. 2008;29:996–1011. A published erratum appears in Infect Control Hosp Epidemiol. 2009;30:107
Kett DH, Azoulay E, Echeverria PM, et al. Candida bloodstream infections in intensive care units: analysis of the extended prevalence of infection in intensive care unit study. Crit Care Med. 2011;39:665–70.
Méan M, Marchetti O, Calandra T. Bench-to-bedside review: Candida infections in the intensive care unit. Crit Care. 2008;12:204.
Guery BP, Arendrup MC, Auzinger G, et al. Management of invasive candidiasis and candidemia in adult non-neutropenic intensive care unit patients: part II. Treatment. Intensive Care Med. 2009;35:206–14.
Delaloye J, Calandra T. Invasive candidiasis as a cause of sepsis in the critically ill patient. Virulence. 2014;5:161–9.
Koulenti D, Garnacho-Montero J, Blot S. Approach to invasive pulmonary aspergillosis in critically ill patients. Curr Opin Infect Dis. 2014;27:174–83.
Bassetti M, Mikulska M, Repetto E, et al. Invasive pulmonary aspergillosis in intensive care units: is it a real problem? J Hosp Infect. 2010;74:186–7.
Garnacho-Montero J, Amaya-Villar R, Ortiz-Leyba C, et al. Isolation of Aspergillus spp. from the respiratory tract in critically ill patients: risk factors, clinical presentation and outcome. Crit Care. 2005;9:R191–9.
Vandewoude K, Blot S, Benoit D, et al. Invasive aspergillosis in critically ill patients: analysis of risk factors for acquisition and mortality. Acta Clin Belg. 2004;59:251–7.
Vandewoude KH, Blot SI, Depuydt P, et al. Clinical relevance of Aspergillus isolation from respiratory tract samples in critically ill patients. Crit Care. 2006;10:R31.
Contou D, Dorison M, Rosman J, et al. Aspergillus-positive lower respiratory tract samples in patients with the acute respiratory distress syndrome: a 10-year retrospective study. Ann Intensive Care. 2016;6:52.
de Hemptinne Q, Remmelink M, Brimioulle S, et al. ARDS: a clinicopathological confrontation. Chest. 2009;135:944–9.
Meersseman W, Vandecasteele SJ, Wilmer A, et al. Invasive aspergillosis in critically ill patients without malignancy. Am J Respir Crit Care Med. 2004;170:621–5.
Karagiannidis C, Brodie D, Strassmann S, et al. Extracorporeal membrane oxygenation: evolving epidemiology and mortality. Intensive Care Med. 2016;42:889–96.
Kim DW, Yeo HJ, Yoon SH, et al. Impact of bloodstream infections on catheter colonization during extracorporeal membrane oxygenation. J Artif Organs. 2015;19:128–33.
Aubron C, Cheng AC, Pilcher D, et al. Infections acquired by adults who receive extracorporeal membrane oxygenation risk factors and outcome. Infect Control Hosp Epidemiol. 2015;34:24–30.
Kuehn C, Orszag P, Burgwitz K, et al. Microbial adhesion on membrane oxygenators in patients requiring extracorporeal life support detected by a universal rDNA PCR test. ASAIO J. 2013;59:368–73.
León C, Álvarez-Lerma F, Ruiz-Santana S, et al. Fungal colonization and/or infection in non-neutropenic critically ill patients: results of the EPCAN observational study. Eur J Clin Microbiol Infect Dis. 2009;28:233–42.
Aubron C, Pilcher D, Leong T, et al. Aspergillus sp. isolated in critically ill patients with extracorporeal membrane oxygenation support. Scand J Infect Dis. 2013;45:715–21.
Hope WW, Walsh TJ, Denning DW. Laboratory diagnosis of invasive aspergillosis. Lancet Infect Dis. 2005;5:609–22.
Meersseman W, Lagrou K, Maertens J, et al. Invasive aspergillosis in the intensive care unit. Clin Infect Dis. 2007;45:205–16.
Meersseman W, Lagrou K, Maertens J, et al. Galactomannan in bronchoalveolar lavage fluid. Am J Respir Crit Care Med. 2008;177:27–34.
Blot SI, Taccone FS, Van den Abeele AM, et al. A clinical algorithm to diagnose invasive pulmonary aspergillosis in critically ill patients. Am J Respir Crit Care Med. 2012;186:56–64.
Alshabani K, Haq A, Miyakawa R, et al. Invasive pulmonary aspergillosis in patients with influenza infection: report of two cases and systematic review of the literature. Expert Rev Respir Med. 2014;9:89–96.
Khasawneh F, Mohamad T, Moughrabieh MK, et al. Isolation of Aspergillus in critically ill patients: a potential marker of poor outcome. J Crit Care. 2006;21:322–7.
Guervilly C, Roch A, Ranque S, et al. A strategy based on galactomannan antigen detection and PCR for invasive pulmonary aspergillosis following influenza A (H1N1) pneumonia. J Infect. 2012;65:470–3.
Crum-Cianflone NF. Invasive aspergillosis associated with severe influenza infections. Open Forum Infect Dis. 2016;3:171–8.
Lewis M, Kallenbach J, Ruff P, et al. Invasive pulmonary aspergillosis complicating influenza A pneumonia in a previously healthy patient. Chest. 1985;87:691–3.
Gill JR, Sheng ZM, Ely SF, et al. Pulmonary pathologic findings of fatal 2009 pandemic influenza A/H1N1 viral infections. Arch Pathol Lab Med. 2010;134:235–43.
Adalja AA, Sappington PL, Harris SP, et al. Isolation of Aspergillus in three 2009 H1N1 influenza patients. Influenza Other Respir Viruses. 2011;5:225–9.
Hinshaw VS, Olsen CW, Dybdahl-Sissoko N, et al. Apoptosis: a mechanism of cell killing by influenza A and B viruses. J Virol. 1994;68:3667–73.
Xie D, Bai H, Liu L, et al. Apoptosis of lymphocytes and monocytes infected with influenza virus might be the mechanism of combating virus and causing secondary infection by influenza. Int Immunol. 2009;21:1251–62.
Shahin J, Allen EJ, Patel K, et al. Predicting invasive fungal disease due to Candida species in non-neutropenic, critically ill, adult patients in United Kingdom critical care units. BMC Infect Dis. 2016;16:480.
Acknowledgements
The data were provided by Peter Rycus, executive administrator of the Extracorporeal Life Support Organization registry. We thank Peter Rycus, the ELSO registry, Gail Faulkner, and the rest of the Leicester ECMO team for their help and support.
Availability of data and materials
The data that support the findings of this study are available from the Extracorporeal Life Support Organization (ELSO), but restrictions apply to the availability of these data, which were used under license for the present study and so are not publicly available. Data are available from the authors, however, upon reasonable request and with permission of the ELSO.
Author information
Authors and Affiliations
Contributions
YAC and HY contributed to study design, data analysis and interpretation, and writing of the manuscript. RP contributed to the writing of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
After approval by the ELSO Registry Committee, limited de-identified datasets are released to participating centers for research purposes without the need for further approval from individual centers. No patient consent is required. This study was conducted in accordance with the amended Declaration of Helsinki.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Additional file
Additional file 1:
Appendix 1. ELSO Registry Case Report Form (document). Appendix 2. Patient selection (list). Appendix 3. Details of Aspergillus involvement (table). Appendix 4. Survival of patients according to number of cultures other than blood positive for Candida (table). Appendix 5. Multiple logistic regression for Aspergillus (table). Appendix 6. Multiple logistic regression for Candida bloodstream infection (table). Appendix 7. Multiple logistic regression for survival (table). Appendix 8. Case distribution by year (figure). (PDF 1090 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Cavayas, Y.A., Yusuff, H. & Porter, R. Fungal infections in adult patients on extracorporeal life support. Crit Care 22, 98 (2018). https://doi.org/10.1186/s13054-018-2023-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13054-018-2023-z