Abstract
Background
In Kenya, diarrhoeal disease is the third leading cause of child mortality after malaria and pneumonia, accounting for nearly 100 deaths daily. We conducted a cross-sectional study in Mukuru informal settlements to determine the bacteria associated with diarrhea and their ASTs to provide data essential for implementing appropriate intervention measures.
Methods
Diarrheagenic children (≤ 5 years) were purposively recruited from outpatient clinics of Municipal City Council, Mukuru kwa Reuben, Medical Missionaries of Mary, and Mama Lucy Kibaki Hospital, Nairobi. A total of 219 stool samples were collected between May 2021 and August 2021. Stool culture was done on MacConkey and Salmonella Shigella agar, while the recovered bacteria were identified using VITEK®2GNID and polymerase chain reaction (PCR) used for E. coli pathotyping. Antibiotic Susceptibility Testing was done using VITEK®2AST-GN83.
Results
At least one bacterial organism was recovered from each of the 213 (97%) participants, with 115 (56%) participants having only one bacterial type isolated, 90 (43%) with two types of bacteria, and 2 (1%) with three types of bacteria recovered. The most predominant bacteria recovered was 85% (93/109) non-pathogenic E.coli and 15% (16/109)of pathogenic E.coli, with 2 (1%) were Enterohemorrhagic E.coli (EHEC), 6 (3%) were Enteroaggregative E.coli (EAEC), and 8 (4%) were Enteropathogenic E.coli (EPEC). Other potentially pathogenic bacteria included Enterobacter sp (27.8%), Klebsiella sp 33(11%), and Citrobacter sp 15(4.7%). Pathogenic isolates such as Salmonella 7 (2%), Proteus mirabilis 16 (6%), Providencia alcalifaciens 1 (0.3%), and Shigella 16 (4.7%) were detected. Isolates such as Pantoea spp 2(0.67%), Raoultella planticola 1(0.33%), and Kluyvera 6(2%) rarely reported but implicated with opportunistic diarrhoeal disease were also recovered. Ampicillin, cefazolin, and sulfamethoxazole-trimethoprim were the least effective antimicrobials at 64%, 57%, and 55% resistance, respectively, while meropenem (99%), amikacin (99%), tazobactam piperacillin (96%), and cefepime (95%) were the most effective. Overall, 33(21%) of all enterics recovered were multidrug-resistant.
Conclusion
The study documented different bacteria potentially implicated with childhood diarrhea that were not limited to E. coli, Shigella, and Salmonella, as previously observed in Kenya. The strains were resistant to the commonly used antibiotics, thus narrowing the treatment options for diarrheal disease.
Similar content being viewed by others
Background
Diarrhoeal disease is Kenya’s third leading cause of pediatric deaths after malaria and pneumonia [1]. According to UNICEF 2022, diarrhoeal diseases account for an estimated 1300 deaths per day in children below five years of age [2]. This burden is higher in informal settlements, characterized by insufficient toilet facilities, inadequate sewage drainage infrastructure, overcrowding, and poor sanitation practices. It is estimated that 48% of Kenya’s population lacks access to safe water and has limited sanitation amenities, resulting in improper disposal of fecal waste in the informal settlements [3, 4].
Although parasites and viruses have been associated with diarrhea diseases, bacteria are the most common aetiological agents, especially Escherichia coli, Clostridium botulinum, Shigella, Staphylococcus aureus, Campylobacter jejuni, Vibrio cholera, and Salmonella [5]. Children aged 5 years and below are at higher risk of foodborne bacterial infections caused by Campylobacter jejuni, Vibrio cholerae, E.coli, Salmonella,andShigellaspecies[6]. Approximately, Shigella causes about 125 million diarrhea episodes and 160,000 deaths every years, with a third are related to young children [7], while diarrheagenic E.coli causes 70,000 deaths in children under 5 years of age in Africa [8]. These bacteria are ingested along with unsafe food and water. They can also be contracted by touching animals suffering from diarrhea, polluted environments, or contact with a person’s stool. Acute diarrhea caused by these bacteria is often characterized by a massive loss of water that forms a large proportion of children’s body weight, leading to rapid dehydration that can lead to death. The severity of acute water loss is aggravated by children using more water daily due to their higher metabolic rates than adults [9].
The primary care for diarrhea involves the replacement of lost electrolytes and water through oral rehydration solution (ORS) [10]. The World Health Organization does not recommend antibiotic treatment of diarrhea in children, but traveler’s diarrhea, commonly associated with enterotoxigenic and enteroaggregative E. coli, non-typhoidal Salmonella, and Shigella spp, requires proper antibiotic therapy to shorten the bacterial excretion and manage the clinical symptoms [11, 12]. In our Kenyan settings, antibiotic treatment of diarrhea involves using oral metronidazole, azithromycin, ciprofloxacin, and amoxiclav [13]. The parenteral antibiotic intervention involves the use of ceftriaxone or ciprofloxacin injections [14] The continued use and misuse of these antimicrobials has resulted in high levels of resistance against commonly available antimicrobials. These enteric bacteria interact within the gut with the potential to transmit and exchange resistance genes, leading to the emergence of multidrug-resistant strains [15]. Diarrhea caused by multidrug-resistant bacteria can result in longer hospitalization, poor treatment outcomes, and increased mortality [16]. Despite drug-resistant enteric bacteria being a significant cause of children’s morbidity and mortality, the full extent of the burden of disease is not well known. This study sought to investigate the common enteric bacterial agents associated with childhood diarrhea and their corresponding antibiotic susceptibility patterns.
Methodology
Study design and site
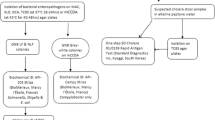
A cross-sectional study design was employed to conduct the study between May 2021 and November 2021 in Mukuru informal settlement, the second-largest informal set-up in Kenya,. The participants were purposively recruited from outpatient clinics located in this informal settlement, in Municipal City Council (MCC), Mukuru kwa Reuben (MR), Medical Missionaries of Mary (MMM), and Mama Lucy Kibaki Hospital (MLKH) Nairobi, the referral facility for patients from MCC, MR, and MMM. MCC, MMM, and MR are level 1/ 2 facilities and thus do not have inpatient facilities, while MLKH operates as an outpatient and inpatient hospital with a 135-bed capacity and a 35-bed children’s ward on the ground floor of the inpatient wing. The ward is divided into an acute care and chronic care unit managing various medical conditions in children aged 1 month to 12 years of age. The clinics were selected because they have the capacity to handle over 1000 cases of diarrheal infections per month, they offer affordable services like a child welfare clinic, and the fact that the research team has long-standing professional relationships with the management of these hospitals.
Study recruitment
A healthcare worker at each of the clinics identified patients presenting mainly with abdominal pain and intense and frequent urge for bowel movement, accompanied by other symptoms including vomiting, fever, dehydration, weight loss, and loose stool. Participants who had taken antibiotics within the previous 7 days to treat other illnesses were excluded from the study, as confirmed by the clinician and the guardian/parent of the child. The parents/guardians of the participants meeting the inclusion criteria were interviewed to obtain the history of their illness and then asked to provide a stool sample or a rectal swab. However, we did not recruit inpatient children and those who were being given parenteral antibiotics.
Sampling
According to the Kenya Bureau of Statistics 2019 census data, 11.2% of Kenya’s population is between 0 and 4 years of age. Using these statistics, we estimated that from a total population of 150,000 people in Mukuru kwa Reuben and Njenga, 16, 800 of its population is below the age of 5 years (http://housingfinanceafrica.org/documents/2019-kenya-population-and-housing-census-reports/). A purposive sampling method (children presenting exclusively with diarrhea were recruited) was used to calculate the working sampling size from our target population of.
16, 800 children. Fisher’s sample size calculation method was used to establish the sample.
size in the study [17].
N = Z 2 P (l-P)/d 2.
Where N = Minimal sample size:
Z = Standard normal deviation corresponding to 95% confidence interval (= 1.96);
P = Estimated prevalence of diarrhoea in children under 5 in Kenya, which in this case is 15% [18].
d = degree of precision (5%).
= (1.96) 2 0.15(1-0.15)/ (0.05) 2 = 195.9216
N = 196 minimum fecal samples.
219 Stool samples per recruitment site were collected depending on how many children presented mainly with diarrhea and had met our inclusion criteria during the participants recruitment period.
Stool sample collection
The fecal sample was collected in a poly pot by the guardian or the parent following guidance from the attending nurse. For children over two years, a cotton rectal swab was used and moistened to avoid discomfort to the child. The collected stool samples were labeled with unique identifiers, kept in Cary Blair transport media, transported to the Centre for Microbiology laboratory in a cool box, and processed within 24 h.
Bacterial isolation and identification
Direct plating on MacConkey agar (Oxoid) and Salmonella Shigella agar (Oxoid)media was done in the lab. Enrichment was also done on Selenite Faecal broth(Oxoid) to optimize the recovery of Salmonella and incubated at 37 ℃ overnight [19]. The non-duplicate enteric colonies were identified using colony morphology, Gram staining, VITEK 2 system (bioMérieux), and PCR [20]. The following NTCT and ATCC organisms were used as positive controls for the detection of the enteric pathogens: NCTC 13,420 to help in the identification of Acinetobacter baumannii, NCTC 10,975 to identify Proteus mirabilis, NCTC 12,028 to identify Morganella morganii, NCTC 8900 to identify Serratia marcescens, E.coli ATCC 25,922 and S. aureus ATCC 25,923. For the recovered E. coli isolates, PCRs were done to identify pathotypes of E.coli associated with opportunistic pediatric diarrhea. The targeted genes for intestinal E. coli pathotypes included bundle-forming pilus(bfpA) of enteropathogenic E.coli (EPEC), Shiga toxin (stx1) of enterohemorrhagic E.coli (EHEC), transcriptional activator (aggR) of enteroaggregative E.coli (EAEC), heat-labile toxin (LT) of enterotoxigenic E.coli (ETEC), heat-stable toxin (sth) of ETEC, stp of ETEC, and afimbrial adhesins(afa) of diffusely adherent E.coli (DAEC) [21]. All E. coli isolates and commercially bought positive controls for each of the genes tested had their DNA extracted using the boiling method previously described by [22]. Briefly, a loopful of pure culture was emulsified in 1 ml distilled DNase/RNase-free water and boiled at 95 °C for 12 min in a heating block. Centrifuging for 5 min at 14 000 rotations per minute did the separation of bacterial cell debris and DNA. The extracted DNA’s quality and concentration were measured using 1% gel electrophoresis stained using SYBR green [23]. The DNA templates were amplified using specific oligonucleotide primer pairs, Table 1. Qiagen master mix was used, where the final volume was 25 µL in a master mix containing one µl forward primer(0.2µM), one µl reverse primer (0.2µM),11 µl PCR water,11 µl PCR mix (QIAGEN, Germany) with the PCR components such as; Taq DNA Polymerase(2.5units), PCR Buffer(1x), MgCl2 (0.2µM), and ultrapure dNTPs(200µM),) followed by addition of 1 µl template DNA. DNA amplification was performed using the following thermal profile for 35 cycles: initial denaturation for 5 min at 95 ℃, followed by final denaturation for 30 s at 94 ℃, variable annealing (shown in Table 1) for 1 min, initial extension for 2 min 30 s at 72 ℃ followed by final extension for 7 min at 72 ℃. 10 µl of amplicons were analyzed by electrophoresis on 1.5% agarose gel. The sizes of the amplicons were determined by comparing them with a 100 bp DNA ladder.
Antimicrobial susceptibility testing
Antibiotic susceptibility testing for 157 non-duplicate representative isolates of each genus was done using the VITEK 2®AST-GN83card on VITEK 2® automated platform. The minimum inhibitory concentration (MIC) interpretations were done using Clinical and Laboratory Standards Institute (CLSI 2022) and the VITEK 2 ® Advanced Expert System(AES), and the analysis using WHONET software followed. The following antimicrobial agents were tested: ampicillin (AMP), amoxicillin/clavulanic acid (AMC), ampicillin/ Sulbactam (SAM), piperacillin/tazobactam (TZP), cefazolin (CZO), cefuroxime(CXM), cefuroxime axetil (CXA), cefoxitin (FOX), cefotaxime (CTX), ceftazidime (CAZ), ceftriaxone(CRO), cefepime(FEP), aztreonam(ATM), meropenem (MEM), amikacin (AMK), gentamicin (GEN), ciprofloxacin (CIP, nitrofurantoin (NIT), and trimethoprim-sulfamethoxazole (SXT). The MICs generated were used to cluster the isolates into various resistance profiles ranging from fully sensitive, intermediate, and resistant. Isolates exhibiting resistance to 3 or more classes or subclasses of antibiotics were scored as Multidrug-resistant (MDR) as defined by the European Center for Disease Control (ECDC) [24], while those that were resistant to penicillins, 1st,2nd, 3rd cephalosporins and aztreonam were scored as ESBLs(Extended Spectrum Beta-lactamase). Isolates, both intermediate and resistant to meropenem, were scored as carbapenemases, while isolates with combined resistance to β-lactams, fluoroquinolone, and aminoglycosides such as gentamicin were denoted as βFQA-strains [24].
Data analysis
Data was analyzed using the Statistical Package for Social Sciences (SPSS), version 20. Descriptive analysis was used to show the distribution of recovered bacterial enteric per recruitment site, gender, and age group. Antimicrobial-resistant patterns were interpreted and analysed using the M 100 Clinical and Laboratory Standards Institute (CLSI 2022) and the VITEK 2® Advanced Expert System (AES). And the Antimicrobial resistant patters were analyzed using WHONET 2023 https://whonet.org/. Moreover the statistical significance of distribution of recovered bacteria isolates between gender, age group and recruitment site was done using Fisher’s exact test.
Results
Patients’ demographic characteristics
Of the 219 participants recruited, 117(53%) were male. The participants in age category 12–24 were significantly higher than other age categories, p < 0.001, as shown in Table 2.
Bacterial isolation
At least one bacterial organism was recovered from 213 (97%) participants, with 115(56%) participants having only one bacterial type isolated and 43.2% (92/213) polyculture, with 90(43%) with two types of bacteria recovered and the common combination being Enterobacter cloacae and E.coli, and 2(1%) with three types of bacteria recovered (Shigella, Proteus mirabilis and Enterobacter cloacae combination while the other combination being Salmonella, Proteus mirabilis and Klebsiella Pnuemoniae). The most predominant bacteria recovered was 85%(93/109) non-pathogenic E.coli and 15% (16/109)of pathogenic E.coli, with 2 (1%) were Enterohemorrhagic E.coli (EHEC), 6 (3%) were Enteroaggregative E.coli (EAEC), and 8 (4%) were Enteropathogenic E.coli(EPEC). Other potentially pathogenic bacteria included Enterobacter sp (27.8%), Klebsiella sp 33(11%), and Citrobacter sp 15(4.7%), with specific species of each genus shown in Fig. 1. Other pathogenic isolates such as Salmonella spp 7 (2%), Proteus mirabilis 16 (6%), Providencia alcalifaciens 1 (0.3%), and Shigella spp 16 (4.7%) were also isolated. Rarely reported but implicated with opportunistic diarrhoeal disease diseases isolates such as Pantoea spp 2 (0.67%), Raoultella planticola 1(0.33%), and Kluyvera spp 5 (2) were also recovered, as depicted in Fig. 1 below.
Antibiotic resistance patterns
Overall, ampicillin (AMP), sulfamethoxazole-trimethoprim (STX), and cefazolin (CZO) were the least effective antimicrobials, with resistance at 64%, 54% & 51%, respectively. Similarly, relatively high resistance towards cefoxitin (33%) and ampicillin-sulbactam (31%) was also recorded. Meropenem, amikacin, tazobactam piperacillin, and cefepime were the most effective, with less than 5% resistance. For antibiotics commonly used for the treatment of diarrhea resistance levels were; amoxiclav (31%), ceftriaxone (14%), and ciprofloxacin (11%), Fig. 2. The resistance levels towards ceftazidime, ceftriaxone, and cefepime range between 5 and 14%. Among antibiotic-inhibitor combinations, amoxicillin-clavulanic acid and ampicillin-sulbactam resistance were at 31%, while for tazobactam piperacillin resistance was (4%).
Genus-specific analysis showed that E coli isolates were resistant to at least 1 of the 20 antibiotics tested, and two (3.6%) non-pathogenic E. coli isolates were resistant to meropenem, as shown in Table 3. E.coli, Enterobacter spp & Klebsiella spp were the most resistant to broad-spectrum antimicrobials such as ceftazidime, ceftriaxone ciprofloxacin, gentamicin, and cefepime, ranging between 6 and 33%. Resistance to all 20 antibiotics was high in the age group 12–24 months, while isolates from participants 0–11 months were the least resistant. Notably, meropenem resistance in the non-pathogenic E.coli was recorded in two recruitment sites, MMM and MCC. Gentamicin resistance was noted in isolates recovered from the MMM (7.4%), MCC (14.3%), and MR (15%) and not in the referral hospital MLK, while amikacin resistance was only recorded in MLK (4.8%) hospital. The isolates recovered as polyculture slightly differed in antibiotic susceptibility patterns for all tested antibiotics. For instance, one patient had Klebsiella pneumoniae, and E.coli recovered from their stool. Their ASTs patterns differed, where Klebsiella pneumoniae was fully susceptible to aztreonam, cefepime, and piperacillin-tazobactam, but E.coli showed resistance ranging between 1 and 6%.
A total of 33(21%) bacterial isolates were multidrug-resistant (MDR) organisms resistant to three or more classes of antibiotics, with the most common MDR phenotype being a combination of ampicillin, amoxiclav, ampicillin-sulbactam, aztreonam, cefazolin, and sulfamethoxazole-trimethoprim at 13%. Of the 33 MDR isolates, 26 (78.8%) were E.coli, with 2 EPEC and 1 EAEC, 2(6%) were Klebsiella pneumoniae, 1(3%) were Klebsiella oxytoca, 1(3%) Proteus mirabillis,1(3%) Kluyvera cryoscrescence and 2(6%) was Enterobacter cloacae. Moreover, 3(1.9%) non-pathogenic E.coli, each from MMM, MCC, and MR, were extensively drug-resistant (XDR) to 6/7 classes of antibiotics tested. A total of 22(14%) were Expanded Spectrum Beta Lactamase (ESBL) producers, presented by 18 E.coli, 1 Klebsiella oxytoca, 1 Klebsiella pneumoniae, 1 Kluyvera cryoscrescence and 1 Enterobacter cloacae from all the recruitment sites, while 2(1%) non-pathogenic E.coli were carbapenemase producers from MMM and MLK, and 8(5%),7 E.coli and1 Klebsiella pneumoniae from the 4 hospitals had Beta-lactam, Fluoro/Quinolones, Aminoglycosides(BFQA) resistance phenotype, as shown in Fig. 3.
Antibiotic-resistant patterns (%) for the recovered bacteria enterics. (n/157) represents the number of bacteria enterics resistant to the antimicrobials out of 157 bacteria subjected to AST. * ampicillin and ampicillin sulbactam resistance were out 100 and 99, respectively, since the Vitek 2 VITEK 2® automated platform did not test it against organisms with intrinsic resistance, including Citrobacter freundii, Enterobacter cloacae, Enterobacter aerogenes, and Morganella morganii
Discussion
The recovery of at least one bacterial enteric from 213/219(97%) implies bacterial etiologies are a significant cause of diarrhea illnesses in children under five years. 43.2% (92/213) of participants were polyculture, with the recovery of the normal intestinal flora alongside pathogenic bacteria enterics associated with childhood diarrhea. The most dominant pathogenic E.coli isolation rate at 15% % (16/109), with 1% EHEC, 4% EPEC, and 3% EAEC, confirms E.coli as a leading cause of diarrhea in under-five years child in Mukuru. Our finding differed from the Zelelie et al. study conducted in Ethiopia among children under five years of age with acute diarrhea and hospitalized or visited the institutions as an outpatient, where the EPEC isolation rate was 13%, 6% EHEC, and 6% EAEC [25]. Moreover, this study’s findings contrast Webale et al. (2020) study conducted in children under five years visiting Mbagathi Hospital, where 36.4% wre pathogenic E.coli, with 20.9% EAEC, 10.2% Enterotoxigenic E.coli (ETEC), 4% EPEC, and 0.5% Enteroinvasive E.coli (EIEC) [26]. Similarly, a study by Impwi et al. (2016) carried out in Mukuru informal settlement in diarrheagenic children under five years had a different isolation rate of E.coli, where 27.1% was EAEC, 37.4% EPEC, 27.1% ETEC, 4.2% EHEC,and 4.2% EIEC [27].In the neighboring Uganda, the isolation rate of EPEC was 6.7%, 14.2% EAC, and 3% EAEC, from a study conducted among inpatient children under 5 years [28], disagreeing with our findings. The varying results across different geographical locations and regions could imply pathogenic E.coli burden varies with locations. The variations could also have been attributed to different inclusion criteria in the study, for instance, Zelelie et al.’s study recruited children who were interacting with calves, while the study conducted in Uganda had inpatient children only. This suggests a need for standardized testing worldwide for diarrhea in children for better surveillance and comparison in the future.
The study also identified rarely reported isolates but implicated with diarrhea diseases such as Pantoea spp, Raoultella planticola, and Kluyvera, while other previous studies in Kenya did not [15, 17, 29]. The current study used VITEK 2®system (bioMérieux) for comprehensive bacterial identification, while these previous studies used the basic biochemical and serotyping methods, signifying a more sensitive automated identification method is superior and should be adopted for robust identification and better treatment plans. Our findings had a lower Pantoea spp isolate rate than a cross-sectional study conducted in two Nigerian healthcare centers among infants with 3.85% [30]. The Raoultella planticola isolation rate in the current study was lower than Afum’s and colleagues’ cross-sectional study conducted among diarrheagenic patients in Ghana, with a 2% isolaion rate [31]. The variations in the isolation rate could be attributed to the burden varying with locations.
The study identified Enterobacter spp as the second dominant bacterial enterics. Among them, Enterobacter cloacae was the most dominant(24.5%). These findings differ from previous studies, which identified Shigella spp as the second most dominant [26, 27, 29]. Usually, Enterobacter cloacae, together with Citrobacter freundii, Enterobacter cloacae complex, Enterobacter aerogenes, and Morganella morganii, are a gut-normal flora that becomes pathogenic when the bacterial load increases, and they acquire virulence genes from other pathogenic isolates. Our study, however, had a 5.2% Shigella isolation rate, a lower rate than 83.8% from a study conducted in Kenya during two different climatic seasons [32]. The differences in the isolation rates could imply changes in trends of bacteria associated with childhood diarrhea. Other possibilities could be related to the study’s comprehensive identification system, recruitment of patients with only acute bloody diarrhea, and climatic seasons aspects in the inclusion criteria.
The study recovered Proteus mirabilis among the bacterial associated with diarrhoea in children under five years. The isolation rate(5.2%) was inconsistent with a 14.5% rate from a study conducted in the same informal settlement four years ago from the day of sample collection [27]. These findings agree with a study conducted in Accra, Ghana, where Proteus mirabilis was isolated at 5.7%, slightly higher than our study’s rate of 5.2% [33]Thse findings imply that Proteus mirabilis is a common cause of childhood diarrhea, which also agrees with findings from Yu et al. (2015). The isolation rate of Salmonella spp (2.3%) agreed with the 2.4% isolation rate from similar studies in Mbagathi Hospital [26].. However, this increased from 1.5% in 2016 [26], suggesting changes in patterns of bacteria in circulation in the informal settlement.
A significant proportion of the recovered bacterial enterics were resistant to commonly used antibiotics to manage diarrhea. There was 64% resistance to ampicillin, 55% to sulfamethoxazole-trimethoprim, and 57% to cefazolin. It is important to note that some of the recovered bacteria, such as Citrobacter freundii, Enterobacter cloacae, Enterobacter aerogenes, and Morganella morganii, contain intrinsic resistance to ampicillin [34] hence the VITEK 2®system (bioMérieux) did not test for it. The high resistance to AMP, SXT (Septrin ™), and CZO could be attributed to the fact that they are readily available in informal settings as over-the-counter drugs [35]. Our findings are consistent with Impwi et al. (2016), which documented high resistance to ampicillin and sulfamethoxazole-trimethoprim [27]. This implies that the resistance levels have been constant in these settings, and there should be a change in treatment options available for better management of bacterial infections.
Additionally, the study documented a 33% resistance toward cefoxitin, differing from Impwi et al. (2016) study conducted in the Mukuru informal settlement, showing a 64% resistance [27]. This variation could have resulted from the recruitment differences where Impwi and colleagues recruited children under five without diarrhea while our study recruited diarrheagenic pediatrics. Moreover, the four-year difference could have allowed for more antimicrobial stewardship in the settings. The high-level resistance to cefoxitin could be associated with the intrinsic resistance of some of the recovered enterics, such as Citrobacter freundii, Enterobacter cloacae, and Enterobacter aerogenes [34].
There was 1% carbapenem resistance for non-pathogenic E.coli. These findings are slightly lower than Dela et al. (2022) findings that documented a 4% meropenem resistance by E.coli recovered from diarrheagenic patients in Accra, Ghana [36]. Although the differences in the recruitment criteria and the testing methods applied in the studies could have accounted for the variations in resistance, the results imply that resistance varies across geographical locations and regions. The findings are worrying as commensal E. coli is harboring carbapenem resistance, and horizontal gene transfer can spread resistance to other pathogenic microorganisms, resulting in severe bacterial infections with reduced treatment options. Additionally, resistance to aminoglycosides would signify differences in antibiotics used in hospitals, as amikacin is majorly given as an injection, and this could imply that resistance recorded in MLKH, a referral hospital, uses it. Also, co-resistance between beta-lactams, fluoro/quinolones, and aminoglycosides suggests limited treatment options for childhood diarrhoea, with severe consequences, including increased mortality rates.
Study limitations
The data was collected from children that had visited the clinics to seek treatments for diarrhoea; therefore, a true community prevalence of diarrhoea in children in the Mukuru informal settlement could not be reflected. However, these groups form an important part that can be used to extrapolate what happens in the community, as they are commonly treated with readily available antibiotics. The study was also conducted immediately after the COVID-19 lockdowns, which limited the sample size as people were still hesitant to visit the hospitals. Due to logistical constraints, only 157 samples were subjected to Antimicrobial Susceptibility Testing, and only E.coli isolates had their pathogenicity confirmed.
Conclusion
The study documented different bacteria implicated as aetiologies of childhood diarrhoea that were not limited to E.coli, Shigella, and Salmonella, as previously observed in Kenya. The presence of Meropenem-resistant non-pathogenic E.coli suggests the possible emergence of carbapenem-resistant strains in the Mukuru informal settlement. Recovery of MDR strains suggests emerging limited treatment options for childhood diarrhoea, with severe consequences, including increased mortality rates. These findings, therefore, indicate the need for more robust standardized surveillance to confirm the patterns observed. Increasing resistance towards amoxiclav implies narrowed treatment options for diarrhoea, posing a public health threat.
Data availability
The data that support the findings of this study are available from the corresponding author, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are, however, available from the authors upon reasonable request and with permission of NACOSTI.
References
Khan DSA, Naseem R, Salam RA, Lassi ZS, Das JK, Bhutta ZA. Interventions for high-burden infectious diseases in Children and adolescents: a Meta-analysis. Pediatrics 2022;149. https://doi.org/10.1542/PEDS.2021-053852C.
UNICEF. Diarrhoea - UNICEF DATA. UNICEF Official Page; 2022.
Mbae CK, Nokes DJ, Mulinge E, Nyambura J, Waruru A, Kariuki S. Intestinal parasitic infections in children presenting with diarrhoea in outpatient and inpatient settings in an informal settlement of Nairobi, Kenya. BMC Infect Dis. 2013;13:1–11. https://doi.org/10.1186/1471-2334-13-243/TABLES/5.
A System of Health Accounts. A System of Health Accounts 2011. https://doi.org/10.1787/9789264116016-EN.
Zhang SX, Zhou YM, Tian LG, Chen JX, Tinoco-Torres R, Serrano E et al. Antibiotic resistance and molecular characterization of diarrheagenic Escherichia coli and non-typhoidal Salmonella strains isolated from infections in Southwest China. Infect Dis Poverty 2018;7. https://doi.org/10.1186/S40249-018-0427-2/ASSET/1FF4DD78-E272-4B0C-A564-691EC19B0B40/ASSETS/GRAPHIC/2095-5162-07-03-103-F002.PNG.
Moharana SS, Panda RK, Dash M, Chayani N, Bokade P, Pati S, et al. Etiology of childhood diarrhoea among under five children and molecular analysis of antibiotic resistance in isolated enteric bacterial pathogens from a tertiary care hospital, eastern odisha, India. BMC Infect Dis. 2019;19:1–9. https://doi.org/10.1186/S12879-019-4501-6/TABLES/5.
Baker S, The HC. Recent insights into Shigella: a major contributor to the global diarrhoeal disease burden. Curr Opin Infect Dis. 2018;31. https://doi.org/10.1097/QCO.0000000000000475.
WHO. World Health organization. The top 10 causes of death Available from: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-ofdeath. Https://WwwWhoInt/News-Room/Fact-Sheets/Detail/the-Top-10-Causes-of-Death 2020.
Jéquier E, Constant F. Water as an essential nutrient: the physiological basis of hydration. Eur J Clin Nutr 2010. 2009;64:2. https://doi.org/10.1038/ejcn.2009.111.
Grenov B, Lanyero B, Nabukeera-Barungi N, Namusoke H, Ritz C, Friis H, et al. Diarrhea, dehydration, and the Associated Mortality in children with complicated severe Acute Malnutrition: a prospective cohort study in Uganda. J Pediatr. 2019;210. https://doi.org/10.1016/j.jpeds.2019.03.014.
World Health Organization. Technical updates of the guidelines on the Integrated Management of Childhood Illness (IMCI). Production 2005.
Tribble DR. Antibiotic therapy for acute watery diarrhea and dysentery. Mil Med. 2017;182. https://doi.org/10.7205/MILMED-D-17-00068.
Rhee C, Aol G, Ouma A, Audi A, Muema S, Auko J et al. Inappropriate use of antibiotics for childhood diarrhea case management - Kenya, 2009–2016. BMC Public Health 2019;19. https://doi.org/10.1186/s12889-019-6771-8.
Guarino A, Bruzzese E, Giannattasio A. Antibiotic treatment of acute gastroenteritis in children. F1000Res. 2018;7. https://doi.org/10.12688/F1000RESEARCH.12328.1/DOI.
Tang Y, Fang L, Xu C, Zhang Q. Antibiotic resistance trends and mechanisms in the foodborne pathogen, Campylobacter. Anim Health Res Rev. 2017;18:87–98. https://doi.org/10.1017/S1466252317000135.
Neupane R, Bhathena M, Das G, Long E, Beard J, Solomon H, et al. Antibiotic resistance trends for common bacterial aetiologies of childhood diarrhoea in low- and middle-income countries: a systematic review. J Glob Health. 2023;13. https://doi.org/10.7189/JOGH.13.04060.
Jung SH. Stratified Fisher’s exact test and its sample size calculation. Biom J. 2014;56. https://doi.org/10.1002/bimj.201300048.
Samuel SK, Moses NM, Emily TJ. Prevalence of Enterobacteriaceae isolated from Childhood Diarrhoea in Mukuru Slums, Nairobi- Kenya. J Adv Microbiol. 2019. https://doi.org/10.9734/jamb/2019/v17i330142.
de Boer MD, de Boer RF, Lameijer A, Sterne E, Skidmore B, Suijkerbuijk AWM et al. Selenite enrichment broth to improve the sensitivity in molecular diagnostics of Salmonella. J Microbiol Methods 2019;157. https://doi.org/10.1016/j.mimet.2018.12.018.
Kohlerschmidt DJ, Mingle LA, Dumas NB, Nattanmai G. Identification of aerobic gram-negative Bacteria. Practical Handb Microbiol. 2021. https://doi.org/10.1201/9781003099277-7.
Kademane A, Dixit M, Vasundhara. A Comprehensive Review of the Pathogenesis and virulence factors of E. Coli. Salud Ciencia Y Tecnologia. 2023;3. https://doi.org/10.56294/saludcyt2023411.
Abdelhai H. M. Comparative study of Rapid DNA extraction methods of pathogenic Bacteria. Am J Bioscience Bioeng 2016;4. https://doi.org/10.11648/j.bio.20160401.11.
Pooniya S, Lalwani S, Raina A, Millo T, Dogra T, Das. Quality and quantity of extracted deoxyribonucleic acid (DNA) from preserved soft tissues of Putrefied Unidentifiable Human Corpse. J Lab Physicians. 2014;6. https://doi.org/10.4103/0974-2727.129088.
Magiorakos AP, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, Giske CG, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18. https://doi.org/10.1111/j.1469-0691.2011.03570.x.
Zelelie TZ, Eguale T, Yitayew B, Abeje D, Alemu A, Seman A, et al. Molecular epidemiology and antimicrobial susceptibility of diarrheagenic Escherichia coli isolated from children under age five with and without diarrhea in Central Ethiopia. PLoS ONE. 2023;18. https://doi.org/10.1371/journal.pone.0288517.
Webale MK, Wanjala C, Guyah B, Shaviya N, Munyekenye GO, Nyanga PL et al. Epidemiological patterns and antimicrobial resistance of bacterial diarrhea among children in Nairobi City, Kenya. Gastroenterol Hepatol Bed Bench 2020;13. https://doi.org/10.22037/ghfbb.v13i3.1910.
Impwi PM, Wambugu P, Kimang’a AN, Kariuki S. Pathogenic e.Coli and other pathogenic gram negative enteric strains from foecal samples of children without diarrhoea living in mukuru slums, Nairobi. East Afr Med J 2016;93.
Masiga F, Kigozi E, Najjuka CF, Kajumbula H, Kateete DP. Diarrhoeagenic Escherichia coli isolated from children with acute diarrhoea at Rakai hospital, Southern Uganda. Afr Health Sci 2022;22. https://doi.org/10.4314/ahs.v22i1.67.
Olipher M, Johnstone M, Anne M, Joseph M, Tom M, Mwau M. Antibacterial spectrum and susceptibility of bacterial pathogens causing Diarrheal illnesses: Cross Sectional Study of Patients Visiting Health Facility in Lake Victoria Region - Kenya. East Afr Sci. 2020;2:45–58. https://doi.org/10.24248/EASCI-D-18-00013.
Pius Okiki A, Pius OA, Olubunmi OC. Medicine and Nursing www.iiste.orgISSN.vol.19.2015.
Afum T, Asandem DA, Asare P, Asante-Poku A, Mensah GI, Musah AB, et al. Diarrhea-causing Bacteria and their antibiotic resistance patterns among Diarrhea patients from Ghana. Front Microbiol. 2022;13. https://doi.org/10.3389/fmicb.2022.894319.
Njuguna C, Njeru I, Mgamb E, Langat D, Makokha A, Ongore D, et al. Enteric pathogens and factors associated with acute bloody diarrhoea, Kenya. BMC Infect Dis. 2016;16:1–10. https://doi.org/10.1186/S12879-016-1814-6/TABLES/4.
Dela H, Egyir B, Majekodunmi AO, Behene E, Yeboah C, Ackah D, et al. Diarrhoeagenic E. Coli occurrence and antimicrobial resistance of Extended Spectrum Beta-Lactamases isolated from diarrhoea patients attending health facilities in Accra, Ghana. PLoS ONE. 2022;17:e0268991. https://doi.org/10.1371/JOURNAL.PONE.0268991.
Magiorakos A-P, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, Giske CG et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance background emergence of resistance to multiple antimicrobial agents in pathogenic bac. Clin Microbiol Infect 2012;18.
Mukokinya MA, Opanga S, Oluka M, Godman B. Dispensing of antimicrobials in Kenya: a cross-sectional pilot study and its implications. J Res Pharm Pract. 2018;7:77. https://doi.org/10.4103/JRPP.JRPP_17_88.
Dela H, Egyir B, Majekodunmi AO, Behene E, Yeboah C, Ackah D, et al. Diarrhoeagenic E. Coli occurrence and antimicrobial resistance of Extended Spectrum Beta-Lactamases isolated from diarrhoea patients attending health facilities in Accra, Ghana. PLoS ONE. 2022;17. https://doi.org/10.1371/journal.pone.0268991.
Acknowledgements
We express our heartfelt gratitude to German Research Foundation (Deutsche Forschungsgemeinschaft) for financial support to carry out the research. We also appreciate the health facilities that allowed us to conduct our study on their premises. Finally, our heart goes to the participants, whom the study couldn’t have been possible without them.
Funding
This work was supported by German Research Foundation (DFG) ref DG: WI1436/13 − 1 awarded to Prof. Samuel Kariuki of KEMRI under MISSIoN project.
Author information
Authors and Affiliations
Contributions
SK led the isolation, identification, and antibiotic susceptibility testing team and has drafted this manuscript. In addition, JM was involved in the crucial components of the study’s methodology formulation and actualization and reviewed and edited the initial drafts of this paper. JNM reviewed and edited the initial drafts of this paper. ES supervised, reviewed, and edited the initial draft. SMK is the one who received the funding, he also supervised, reviewed, and edited the initial draft.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
Ethical approval for the study was obtained from the Scientific Ethical Review Committee (SERU) of Kenya Medical Research Institute (No. KEMRI/SERU/CMR/P00155/4195). A research license, NACOSTI/P/21/12,323, was obtained from the National Commission for Science, Technology, and Innovation (NACOSTI). Informed consent to collect stool samples from children (participants) was obtained from the parents and guardians before recruiting the participants for the study, and no penalties for those who declined to participate. A unique study identification number and barcode label was applied to the sample immediately after collection for anonymity, and the information collected was confidential. All the methods and procedures carried out in this study were in accordance with the Helsinki Declaration.
Consent for publication
Not applicable.
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Kiiru, S., Maina, J., Mwaniki, J.N. et al. Enteric bacterial agents associated with diarrhea and their antimicrobial sensitivity profiles in children under 5 years from mukuru informal settlement, Nairobi, Kenya. BMC Infect Dis 24, 237 (2024). https://doi.org/10.1186/s12879-024-09114-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12879-024-09114-5