Abstract
Background
Neurological infection is an important cause of critical illness, yet little is known on the epidemiology of neurological infections requiring critical care.
Methods
We analysed data on all adults with proven or probable neurological infection admitted to UK (NHS) critical care units between 2001 and 2020 reported to the Intensive Care National Audit and Research Centre. Diagnoses, physiological variables, organ support and clinical outcomes were analysed over the whole period, and for consecutive 5-year intervals within it. Predictors of in-hospital mortality were identified using a backward stepwise regression model.
Results
We identified 20,178 critical care admissions for neurological infection. Encephalitis was the most frequent presentation to critical care, comprising 6725 (33.3%) of 20,178 cases. Meningitis– bacterial, viral or unspecified cases - accounted for 10,056 (49.8%) of cases. In-hospital mortality was high, at 3945/19,765 (20.0%) overall. Over the four consecutive 5-year periods, there were trends towards higher Glasgow Coma Scale scores on admission, longer critical care admissions (from median 4 [IQR 2–8] to 5 days [IQR 2–10]), and reduced in-hospital mortality (from 24.9 to 18.1%). We identified 12 independent predictors of in-hospital death which when used together showed good discrimination between patients who die and those who survive (AUC = 0.79).
Conclusions
Admissions with neurological infection to UK critical care services are increasing and the mortality, although improving, remains high. To further improve outcomes from severe neurological infection, novel approaches to the evaluation of risk stratification, monitoring and management strategies are required.
Key points
• Meningitis comprised 50% and encephalitis comprised 33% of neurological infections requiring critical care admission.
• During the 20-year study period, there was a progressive trend of increasing neurological infection admissions to critical care, and a reduction in the overall mortality rate.
Similar content being viewed by others
Introduction
Neurological infections are major causes of mortality and morbidity [1]. Worldwide, meningitis and encephalitis are estimated to cause 290,000 and 108,000 deaths per year, respectively [1, 2]. The mortality rates of neurological infections, which can exceed 50% in some settings, are higher than those of most other infection types [2,3,4]. Neurological infections frequently present as emergencies, which may rapidly progress to death, or permanent neurological disability in survivors [3, 5, 6]. In the UK, patients with neurological infection are managed in both specialist and non-specialist hospitals. Uncertainties in risk stratification, diagnostic approaches, monitoring strategies, and optimal management particularly of severe disease and raised intracranial pressure (ICP), likely contribute to lengthy hospital admissions and a substantial healthcare burden.
Patients with neurological infection often require admission to critical care units for close monitoring and physiological support, aiming to minimise secondary brain injury [6, 7]. Support includes close neurological monitoring, seizure prophylaxis, and airway protection in those with depressed consciousness. Less commonly, neurological infection may develop in individuals already in critical care, particularly those who have undergone invasive neurological procedures [8].
There are limited data to guide management of neurological infections beyond use of anti-infectives and corticosteroids [7, 9,10,11,12,13]. Raised ICP is often suspected in severe disease, yet its incidence in primary neurological infection is uncertain given invasive ICP monitoring is not routine. Improved outcomes with ICP-guided management have been described in some settings [14, 15]. In an observational study of 52 adults with bacterial meningitis in Sweden, mortality at 2 months was significantly lower in the group receiving ICP-guided therapy (target ICP < 20 mmHg and cerebral perfusion pressure > 50 mmHg), vs. standard care [15]. Cases were not randomly assigned to groups, and the extent to which the study results were related to ICP control, as opposed to other inter-group differences, is uncertain. Non-invasive ICP monitoring may make such an approach more widely deliverable.
Neurological infections account for a minority of critical care admissions, but are associated with high mortality. In the 24-hour point prevalence study EPIC III, conducted at 1150 centres in 88 countries in 2017, of 8135 patients in intensive care with suspected or proven infection, 314 (3.9%) had neurological infection [16]. Their in-hospital mortality was 29.0%. Few studies have captured the full spectrum of neurological infection in critical care [17, 18]. In a prospective multicenter international cohort study of meningoencephalitis in intensive care, of 591 cases with identified aetiologies there were 247/591 (41.8%) cases of acute bacterial meningitis and 140/591 (23.7%) cases of infectious encephalitis [19]. The characteristics, management and outcomes of neurological infection in UK critical care have not been well characterized.
A National Infection Trainees Collaborative for Audit and Research study of community acquired meningitis in the UK and Ireland estimated that 50% patients with confirmed bacterial meningitis required critical care admission [20]. To address this knowledge gap, and characterize adult neurological infection admissions to UK critical care, we analyzed records from the Intensive Care National Audit and Research Centre (ICNARC) [21], an independent charity that collects and provides national-level data of UK critical care admissions. In this retrospective cohort study, we investigated the burden, type, presentation, severity, organ support requirements, and clinical outcomes, of adult neurological infection admissions to UK critical care units over a 20-year period.
Methods
Case selection
Data were retrieved for adults (≥ 18 years) admitted to UK critical care units, with reasons for critical care admission (primary, secondary, and ultimate reasons using the ICNARC Coding Method, in the ICNARC Case Mix Programme [CMP] database) coded as encephalitis, meningococcal meningitis, meningitis unspecified, bacterial meningitis not meningococcal, intracranial abscess, infected cerebrospinal fluid (CSF) shunt, viral meningitis, and tuberculous meningitis. Cerebral malaria cases could not be separated from non-cerebral malaria, and were excluded. Cases of spinal infection were not included. One or more diagnoses were provided for each case, assigned either at critical care admission or discharge. Following ICNARC methodology, the primary reason for admission was considered to be the most important underlying condition or reason for admission to the critical care unit, assessed and recorded at admission to (and during the first 24 h in) the critical care unit. For cases where ‘meningitis unspecified’ was recorded with a second neurological infection diagnosis, the more precise diagnosis was used. For cases with more than one neurological infection diagnosis (other than ‘meningitis unspecified’) e.g., ‘encephalitis’ plus ‘viral meningitis’, the diagnosis listed as the ‘primary reason for admission’ was used. If this was not available, the ‘secondary reason for admission’ was taken, or the ‘ultimate reason for admission’ if neither was available.
ICNARC data were collected under existing ethical approvals (Supplementary Material 1). Ethical approval for this study was granted by the London School of Hygiene and Tropical Medicine Research Ethics Committee (26,550).
Clinical data
Data were identifed based on the ICNARC data collection form 3.1 (2015). The following parameters were obtained: age, gender, weight, height, date of hospital admission, date of critical care admission, source of critical care admission, reason for critical care admission, and comorbidities. Lowest and highest values during the first 24 h in critical care were obtained for: temperature, blood pressure, heart rate, respiratory rate and urine output; hemoglobin concentration, white cell count and platelet count; and serum concentrations of bicarbonate, sodium, potassium, glucose, lactate, urea, and creatinine. Arterial blood gas parameters for lowest pH and lowest PaO2 were obtained. Details of pupil reactivity, lowest Glasgow Coma Scale (GCS) score, period of sedation and paralysis, and highest level of physiological support provided, the number of days of organ support (by organ system), dates of critical care unit discharge, and hospital discharge, were also obtained.
‘Days of neurological support’ was defined as the number of calendar days (including part days) during which any neurological support was administered. Neurological support was defined as: a requirement for monitoring for central nervous system depression prejudicing the airway and its protective reflexes; invasive neurological monitoring or treatment; continuous intravenous medication to control seizures; continuous cerebral monitoring; or therapeutic hypothermia.
Statistical analysis
Data were summarized using medians for continuous variables and counts (with percentages) for discrete and ordinal variables. Requirement for neurological support, disability and mortality outcomes were described for all cases, and then stratified by admission brain infection type, and four consecutive 5-year admission periods (2001–2005, 2006–2010, 2011–2015, 2016–2020) chosen prior to data analysis.
We aimed to identify independent predictors of mortality by building a mortality prediction model. We considered all variables listed in Table 1 for inclusion, but excluded variables for which data were missing in > 1,000 cases (with the exception of GCS which was judged likely to be a very strong predictor of mortality). For variables with multiple measurements within a 24-hour period, we investigated associations with both the lowest and highest values, as defined by the ICNARC risk prediction model [22]. We expected some variables to have non-linear relationships with mortality (e.g., body temperature). To assess the appropriate form with which to include these variables we first assessed univariate associations of all continuous predictors using restricted cubic splines with 3 knots. We identified which variables were significant predictors by including them in a logistic regression model with a backwards stepwise selection procedure, with p values ≥ 0.05 eliminated at each iteration. We report odds ratios (OR) of death during hospital admission, and 95% confidence intervals (CIs). The ability of variables to discriminate between death and survival was assessed using the C-statistic, equivalent to the area under the receiver operating characteristic (ROC) curve. Analysis was performed using STATA version 17 (StataCorp LLC, College Station, TX) and R version 4.2.2 (R Foundation for Statistical Computing, Vienna, Austria) (Supplementary Material 2).
Results
Critical care admission
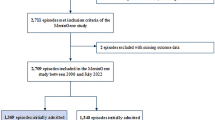
Neurological infection represented 20,178 (0.7%) of 2,808,359 adult admissions to all UK critical care units participating in the ICNARC CMP from 1st January 2001 to 31st January 2020. Critical care observation time was for a total of 416.3 years (20,169 cases). The median age at critical care unit admission was 53 years (interquartile range [IQR] 37–66), and 11,086 cases (54.9%) were male. Neurological infection diagnoses requiring critical care admission were: encephalitis, 6725 (33.3%); meningitis unspecified, 5360 (26.6%): bacterial meningitis non-meningococcal, 3105 (15.4%); intracranial abscess, 2730 (13.5%); meningococcal meningitis, 1096 (5.4%); infected CSF shunt, 665 (3.3%); viral meningitis, 363 (1.8%); tuberculous meningitis, 132 (0.7%). Additional admission diagnoses of status epilepticus or uncontrolled seizures, secondary hydrocephalus and tuberculosis are described in Supplementary Material 3. The emergency department was the most common source of admission (6961 [42.3%]). Critical care admission data, including clincial and laboratory parameters evaluated during the first 24 h in critical care, are described in Table 1.
Organ support
The median duration of critical care unit stay was 4 days (IQR 2–9). The median durations of level 3 support (two or more organ systems, or advanced respiratory support) [23] and level 2 support (single organ) were 3 days (IQR 1–7), and 2 days (IQR (0–3), respectively. 6624/16,449 (40.3%) required at least one day of neurological support. The neurological infections that most commonly required neurological support were CSF shunt infection (398/569 [69.9%] cases), intracerebral abscess (1200/2400 [50.0%] cases), and tuberculous meningitis (65/132 [49.2%] cases). Overall, 325/547 (59.4%) of cases with an CSF shunt infection, 1051/2228 (47.2%) of cases with an intracranial abscess, and 51/116 (44.0%) of cases with tuberculous meningitis, were expected to require at least minor assistance with some daily activities in the two weeks following hospital discharge.
Outcomes
The median length of hospital stay was 20 days (IQR 10–44). Overall, 3945/19,765 (20.0%) cases with neurological infection died in the hospital in which the critical care unit was based (Table 2). The neurological infection type associated with highest mortality during hospital admission was tuberculous meningitis (44/127 [33.3%]). For those discharged from hospital alive, at least minor assistance was required with daily activities in the first two weeks in 325/547 cases (59.4%) of CSF shunt infection, 1051/2228 cases (47.2%) of intracranial abscess, and 51/116 cases (44.0%) of tuberculous meningitis.
Analysis by consecutive time periods
Neurological infection admissions to critical care reported to ICNARC increased from 2742 in 2001–2005 to 7339 in 2015–2020. The number of critical care units reporting data to the ICNARC CMP also increased, from a median of 170 units to 283 units over the same time period. In-hospital mortality was 24.9% in 2001–2005, falling progressively to 18.1% in 2016–2020, and there was a similar reduction in the requirement for neurological support (Table 3; Fig. 1). Lowest GCS, days to critical care admission, and days of critical care stay, are described for the 5-year intervals of this study in Table 3. Over the four consecutive 5-year periods, the proportion of cases dying during the admission fell; the admission GCS increased; and the duration of admissions lengthened (Table 4).
Mortality prediction
A total of 10,403 admissions had complete information on all variables considered for inclusion (listed in the footnote to Table 5). Among this subset of patients, we identified 12 variables each of which were independently and significantly associated with increased risk of mortality in multivariate models. The odds of death during hospital admission were lower for females (OR vs. males: 0.84, 95% CI 0.75, 0.94, p < 0.01), and increased with age (OR 1.2 [CI 1.2, 1.3] per decade older, p < 0.001). Three or more co-morbidities (from the list shown in Table 1) strongly predicted death during hospital admission (OR vs. 0 or 1 comorbidities 2.4, 95% CI 1.8, 3.4, p < 0.001). For each 1-point increase in the lowest GCS score in the first 24 h of admission, the odds of death reduced by 0.84 (95% CI 0.82, 0.85, p < 0.001). Tachycardia (> 90 beats/minute), hypotension (systolic blood pressure < 110mmHg), leukopenia (< 25 × 109/L and tachypnea were each associated with increased mortality. Low serum urea (below 10mmol/l) was associated with reduced risk. For several variables there was a U-shaped association with mortality. For minimum body temperature the lowest risk occurred at 37oC, and for maximum body temperature it occurred at 38oC. For urine output the lowest risk was at 2.5 L/day, and for serum sodium it was at 140mmol/L. Inclusion of all 12 measurements in a mortality prediction model gave a C-statistic of 0.79 (Fig. 2), indicating that an admission who dies has a higher predicted risk than an admission who survives on 79% of occasions.
Discussion
Harnessing the high-quality ICNARC database, we present the largest descriptive analysis of neurological infection amongst adults admitted to critical care units in the UK over 20 years. Taken together, encephalitis was the most common diagnosis requiring admission to critical care. Meningitis diagnoses– including bacterial, viral, and unspecified cases - accounted for almost 50% of admissions. Typically, patients were admitted to critical care within 24 h of presentation, most commonly from the emergency department. Critical care stays were short, and almost half of cases required neurological support. Over 20 years there was a trend towards higher admission GCS scores, longer critical care stays, and reduced in-hospital mortality.
Neurological infections such as bacterial meningitis and viral encephalitis in adults are uncommon causes of admission to hospital in the UK, but have disproportionately high morbidity and mortality burden. In our cohort, which is expected to include individuals with the most severe disease, the in-hospital mortality of 20% was similar to the 23.9% mortality rate reported from a broader, multinational observational study of patients admitted to 167 ICUs in 17 European countries [24]. UK data are sparse; a recent study of community acquired meningitis admissions to UK and Ireland hospitals reported 3% in-hospital mortality [20]. However, patients with encephalitis, cryptococcal tuberculous and nosocomial meningitis were excluded from the analysis, and only 192 (13.1%) patients required ICU admission.
An analysis of mortality by sequential 5-year time periods demonstrated a progressive reduction of mortality over time; from 24.9% in 2001–2005, to 18.1% in 2016–2020. While a change in ICU admission criteria cannot be excluded, this is consistent with improved outcomes from neurological infection over the past 25 years [25], and is likely to be associated with significant improvements in diagnostics, monitoring, treatments, and critical care management over this time period. Furthermore, the Global Burden of Disease Study for neurological disorders (195 countries, 1990–2016) demonstrated both mortality, and disability-adjusted life-years, are improving for meningitis and encephalitis [25], The health improvements responsible for these data may also be driving fewer severe neurological infection presentations to critical care.
Our data describe a steady increase in the reporting of neurological infection critical care admissions to the ICNARC CMP. This contrasts with the reduction in disease severity, particularly severe meningococcal disease, which might be anticipated from the introduction of national vaccination programmes which started with MenC in 1999 [26]. The proportion of UK critical care neurological infection admissions diagnosed with meningococcal meningitis has actually remained stable during the period of this analysis. Overall, critical care admissions were longer, which may reflect an increasing need for prolonged critical care support, a more cautious approach to step-down, or improved survival amongst the study population. The observed increase in admissions may be explained by several factors which are not mutually exclusive. These include improved data collection with an increase in the number of critical care units reporting data over this period; lower thresholds for critical care admission of neurological infections, perhaps reflecting an increase in the number of critical care beds available in the UK (from 3.8 to 5.9 per 100,000 people between 2011 and 2020) [27]; improved recognition and diagnosis of neurological infection in the community or in-hospital, through better diagnostics (especially CSF multiplex PCR), improved brain imaging for encephalitis and rapid neurological reporting, and clinical practice guidelines for neurological infection [8, 27, 28], and finally an increase in severe neurological infections in a growing and ageing UK adult population [28]. An increase in admission GCS during the study period may also support lower thresholds for critical care admission of neurological infections.
In our analysis, encephalitis was the most common neurological infection diagnosis, accounting for more cases than meningococcal meningitis and non-meningococcal bacterial meningitis combined. This is consistent with estimates describing a higher incidence of encephalitis than bacterial meningitis in the UK [29, 30]. Our study is large and the first to describe the UK burden of neurological infections requiring critical care. However, due to its observational and retrospective design, it has limitations. We describe a requirement for neurological support in ~ 40% cases, but the nature of support required by individual patients is unknown. The ICNARC coding method did not specifically identify cryptococcal disease as a cause of meningitis, and these presentations may be represented by other meningitis codes in the data. Our analysis assumes all meningitis and encephalitis cases are caused by infection, and might erroneously include some non-infectious causes. Neurological infection diagnoses are dependent upon accurate clinical coding, and may not reflect microbiological data. Misclassification of cases is possible, particularly for distinction between viral meningitis and encephalitis, and between viral and bacterial meningitis. Prior to 2006, specific meningitis codes were not used, and meningitis cases were grouped together. We combine data for neurological infections that differ in pathophysiology and treatment. No disability data were reported for 2001–2005, so the time-period trend data in this respect are limited. Data were reported to ICNARC on a voluntary basis. Increasing participation in the ICNARC CMP over the study period affects evaluation of changing dynamics over this time. Consecutive 5-year intervals were chosen to give four equal-length periods that would contain sufficient cases to draw conclusions; however, a different approach (e.g., 2-year groups) may have revealed different trends.
Notwithstanding its limitations, our analysis provides a comprehensive picture of the burden of neurological infections managed in UK critical care units, including changes in its characteristics and outcomes over a 20-year period. Our analysis shows that mortality remains high. In this context, the gaps in the evidence to inform management strategies are notable, particularly the uncertainties in risk stratification, the optimal use and benefits of intracranial pressure monitoring and case management. These knowledge gaps hamper our ability to select the most appropriate cases for critical care; to allocate them rationally to specialist and non-specialist units; and to provide the care they need to secure the best possible outcomes.
Data availability
The data that support the findings of this study are available from the Intensive Care National Audit and Research Centre (ICNARC) but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of ICNARC.
References
Zunt JR, Kassebaum NJ, Blake N, et al. Global, regional, and national burden of meningitis, 1990–2016: a systematic analysis for the global burden of Disease Study 2016. Lancet Neurol. 2018;17:1061.
World Health Organization. Geneva. Defeating meningitis by 2030 global road map (version 26 October 2020). https://www.who.int/publications/m/item/defeating-meningtis-by-2030-global-road-map-(version-20-october-2020) (accessed July 16, 2021).
Pasquier E, Kunda J, de Beaudrap P, et al. Long-term mortality and disability in cryptococcal meningitis: a systematic literature review. Clin Infect Dis. 2018;66:1122–32.
Wilkinson RJ, Rohlwink U, Misra UK, et al. Tuberculous meningitis. Nat Rev Neurol. 2017;13:581–98.
Dyckhoff-Shen S, Koedel U, Pfister H-W, Klein M. SOP: emergency workup in patients with suspected acute bacterial meningitis. Neurological Research and Practice 2021 3:1 2021; 3: 1–7.
Donovan J, Figaji A, Imran D, Phu NH, Rohlwink U, Thwaites GE. The neurocritical care of tuberculous meningitis. Lancet Neurol. 2019;18:771–83.
McGill F, Heyderman RS, Michael BD, et al. The UK joint specialist societies guideline on the diagnosis and management of acute meningitis and meningococcal sepsis in immunocompetent adults. J Infect. 2016;72:405–38.
Vengamma B, Rajguru M, Prasad BCM, Ramesh Chandra VV. Central nervous system infections in the intensive care unit. J Clin Sci Res 2014;: 106–13.
van de Beek D, de Gans J, Tunkel AR, Wijdicks EFM. Community-acquired bacterial meningitis in adults. N Engl J Med. 2006;354:44–53.
Brouwer MC, Mcintyre P, Prasad K, van de Beek D. Corticosteroids for acute bacterial meningitis. Cochrane Database of Systematic Reviews. 2015;CD004405. https://doi.org/10.1002/14651858.CD004405.pub5.
Thwaites GE, Bang ND, Dung NH, et al. Dexamethasone for the treatment of tuberculous meningitis in adolescents and adults. N Engl J Med. 2004;351:1741–51.
Prasad K, Singh MB, Ryan H. Corticosteroids for managing tuberculous meningitis. Cochrane Database of Systematic Reviews. 2016. https://doi.org/10.1002/14651858.CD002244.pub4. Published online April 28.
Beardsley J, Wolbers M, Kibengo FM, et al. Adjunctive dexamethasone in HIV-Associated Cryptococcal Meningitis. N Engl J Med. 2016;374:542–54.
Lindvall P, Ahlm C, Ericsson M, Gothefors L, Naredi S, Koskinen L-OD. Reducing intracranial pressure may increase survival among patients with bacterial meningitis. Clin Infect Dis. 2004;38:384–90.
Glimåker M, Johansson B, Halldorsdottir H, et al. Neuro-intensive treatment targeting intracranial hypertension improves outcome in severe bacterial meningitis: an intervention-control study. PLoS ONE. 2014;9:e91976.
Vincent JL, Sakr Y, Singer M, et al. Prevalence and outcomes of infection among patients in Intensive Care Units in 2017. JAMA. 2020;323:1478.
Fernandes D, Gonçalves-Pereira J, Janeiro S, Silvestre J, Bento L, Póvoa P. Acute bacterial meningitis in the intensive care unit and risk factors for adverse clinical outcomes: retrospective study. J Crit Care. 2014;29:347–50.
Valdoleiros SR, Torrão C, Freitas LS, Mano D, Gonçalves C, Teixeira C. Nosocomial meningitis in intensive care: a 10-year retrospective study and literature review. Acute and Critical care. 2022;37:61–70.
Sonneville R, de Montmollin E, Contou D, et al. Clinical features, etiologies, and outcomes in adult patients with meningoencephalitis requiring intensive care (EURECA): an international prospective multicenter cohort study. Intensive Care Med. 2023;49:517–29.
Ellis J, Harvey D, Defres S, et al. Clinical management of community-acquired meningitis in adults in the UK and Ireland in 2017: a retrospective cohort study on behalf of the National Infection Trainees Collaborative for Audit and Research (NITCAR). BMJ Open. 2022;12:e062698.
Intensive Care National Audit & Research Centre. Home. 2022. https://www.icnarc.org/ (accessed Dec 13, 2022).
Harrison DA, Parry GJ, Carpenter JR, Short A, Rowan K. A new risk prediction model for critical care: the Intensive Care National Audit & Research Centre (ICNARC) model. Crit Care Med. 2007;35:1091–8.
Intensive Care Society. Levels of care. 2021. https://ics.ac.uk/resource/levels-of-care.html (accessed Feb 7, 2023).
Capuzzo M, Volta CA, Tassinati T, et al. Hospital mortality of adults admitted to Intensive Care Units in hospitals with and without Intermediate Care units: a multicentre European cohort study. Crit Care. 2014;18. https://doi.org/10.1186/S13054-014-0551-8.
Feigin VL, Nichols E, Alam T, et al. Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the global burden of Disease Study 2016. Lancet Neurol. 2019;18:459.
Subbarao S, Campbell H, Ribeiro S, et al. Invasive meningococcal Disease, 2011–2020, and impact of the COVID-19 pandemic, England. Emerg Infect Dis. 2021;27:2495.
Our World in Data. Intensive care beds per 100,000 people., 2020. 2020. https://ourworldindata.org/grapher/intensive-care-beds-per-100000 (accessed Feb 9, 2023).
Kadambari S, Okike I, Ribeiro S, et al. Seven-fold increase in viral meningo-encephalitis reports in England and Wales during 2004–2013. J Infect. 2014;69:326–32.
McGill F, Griffiths MJ, Bonnett LJ, et al. Incidence, aetiology, and sequelae of viral meningitis in UK adults: a multicentre prospective observational cohort study. Lancet Infect Dis. 2018;18:992–1003.
Granerod J, Cousens S, Davies NWS, Crowcroft NS, Thomas SL. New estimates of incidence of Encephalitis in England. Emerg Infect Dis. 2013;19:1455.
Acknowledgements
This publication is based on data derived from the Intensive Care National Audit & Research Centre (ICNARC) Case Mix Programme Database. The Case Mix Programme is the national, comparative audit of patient outcomes from adult critical care coordinated by ICNARC. We thank all the staff in the critical care units participating in the Case Mix Programme. For more information on the representativeness and quality of these data, please contact ICNARC. Disclaimer: The views and opinions expressed therein are those of the authors and do not necessarily reflect those of ICNARC.
Funding
The acquisition of these study data was funded by Professor David Moore (Professor of Infectious Diseases and Tropical Medicine, LSHTM), who was not involved in the study design, in the collection, analysis, or interpretation of the data, or in the writing of the manuscript.
Author information
Authors and Affiliations
Contributions
JD, AWH, ECW and RSH designed the study. JD acquired the data. JD, AG, and JG analysed the data. JD wrote the first draft of the manuscript. JD, JG, AWH, ECW, and RSH revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was performed in accordance with relevant guidelines and regulations, and in accordance with the Declaration of Helsinki.
ICNARC data were collected by ICNARC under existing ethical approvals. As available on https://www.icnarc.org/About/Information-Standards/Information-Security/Privacy-Policy, ICNARC have approval to hold patient identifiable data under Sect. 251 of the NHS Act 2006. Approval was granted by the Confidentiality Advisory Group (CAG) within the Health Research Authority (HRA) (previously National Information Governance Board). The approval number for the CMP was PIAG 2–10(f)/2005. Ethical approval for this study was granted by the London School of Hygiene and Tropical Medicine Research Ethics Committee (26550). Individual participant content was waived by the London School of Hygiene and Tropical Medicine Research Ethics Committee due to retrospective nature of the study. An information sharing agreement was signed with ICNARC.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Donovan, J., Glover, A., Gregson, J. et al. A retrospective analysis of 20,178 adult neurological infection admissions to United Kingdom critical care units from 2001 to 2020. BMC Infect Dis 24, 132 (2024). https://doi.org/10.1186/s12879-024-08976-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12879-024-08976-z