Abstract
Background
Trueperella pyogenes (T. pyogenes) is a bacterium that colonizes the skin and mucosal surfaces of various domestic and wild animals. It rarely leads to infections in humans, with only a few descriptions available in the literature.
Case presentation
A 71-year-old Swiss farmer with a history of recurring basal cell carcinoma and metastasized pancreatic neuroendocrine tumor presented with signs of sepsis after a three-day history of general weakness, malaise and fever. Clinical and echocardiographic findings, as well as persistent bacteremia were consistent with mitral valve endocarditis caused by T. pyogenes. The patient’s condition gradually improved under antibiotic treatment with piperacillin/tazobactam (empiric therapy of sepsis), and later penicillin G based on resistance testing. He was discharged after 13 days and continued outpatient antibiotic therapy with ceftriaxone, resulting in a total antibiotic treatment duration of six weeks. This is the first literature review of T. pyogenes endocarditis in humans. Among nine cases of T. pyogenes endocarditis, three patients had documented contact with farm animals and five had an underlying condition that compromised the immune system. While antibiotic resistance of T. pyogenes is an emerging concern, susceptibility to beta-lactam antibiotics seems to persist. The mortality of T. pyogenes endocarditis described in the literature was high, with 66% of patients not surviving the disease.
Conclusions
T. pyogenes is a rare causative organism of infectious endocarditis in humans and descriptions are mainly restricted to case reports. In our review of the literature, we found that both an impaired immune system and contact with farm animals might be risk factors. Growth of T. pyogenes in blood cultures is unlikely to be missed during routine analysis, as it shows marked beta-hemolysis on blood agar culture plates, which generally leads to further characterization of the bacteria. Susceptibility to penicillin, ceftriaxone, and macrolides seems to be retained and the reported mortality in the few patients with T. pyogenes endocarditis is high.
Similar content being viewed by others
Background
Trueperella pyogenes (T. pyogenes) is a Gram-positive, non-motile, non-spore-forming coccobacillus or short rod that occurs singly, in pairs, or in clusters [1, 2]. Over the years, various taxonomic revisions have been made, changing its classification from Bacillus pyogenes, to Corynebacterium pyogenes, Actinomyces pyogenes, Arcanobacterium pyogenes, and finally Trueperella pyogenes [2, 3]. It is known to colonize the skin and mucosal surfaces of many domestic animals, such as cattle, swine, sheep and goats, causing a variety of infections, including mastitis, wound infections, pneumonia and liver abscess [4, 5]. Infections in humans occur only sporadically and mostly in immunocompromised patients with contact to farm animals [4], leading to sepsis [6], endocarditis [7], pneumonia [8], and skin ulcers [9], among others. We report a case of sepsis and endocarditis caused by T. pyogenes in a Swiss farmer and summarize eight similar cases from the literature.
Case presentation
A 71-year-old man presented to the emergency department of a Swiss university hospital with a three-day history of general weakness, malaise and fever. Symptoms occurred three days after administration of a first dose of cemiplimab for treatment of a recurring locally advanced basal cell carcinoma of the left cheek. Due to a pancreatic neuroendocrine tumor with liver metastasis, the patient was currently under palliative treatment with everolimus and somatostatin receptor-targeted radionuclide therapy. A routine transthoracic echocardiogram (TTE) prior to the start of immunotherapy had shown signs compatible with cardiac amyloidosis and coronary artery disease (CAD). The patient was a retired farmer, still living on a cattle farm in Switzerland.
On presentation ear temperature was 38.6 °C, blood pressure 79/44 mm Hg, heart rate 89 beats per minute, respiratory rate 27 breaths per minute, and oxygen saturation 88% on ambient air. He was somnolent, but no focal neurologic deficits were observed. The heart sounds were regular, with a systolic murmur at the third intercostal space, left lower sternal border. Auscultation of the lungs was unremarkable. The ulcerating basal cell carcinoma, measuring 3 × 5 cm, was visible on the left cheek, without signs of inflammation. Examination of the skin and joints was otherwise unremarkable. Laboratory workup showed a C-reactive protein (CRP) of 96 mg/L (normal < 5 mg/L), with normal leucocytes of 5,5 × 109/L (normal range 3 to 10,5 × 109/L), and thrombocytopenia of 107 × 109/L (normal range 150 to 450 × 109/L). Chest X-ray demonstrated signs of mild pulmonary venous congestion. A computed tomography (CT) of the head showed no evidence of soft tissue abscess in the area surrounding the basal cell carcinoma. After collection of blood cultures, empiric treatment with piperacillin/tazobactam (4.5 g every eight hours) was initiated for suspected sepsis of unknown origin in an immunocompromised patient. The patient initially required hemodynamic stabilization with norepinephrine in the intermediate care unit.
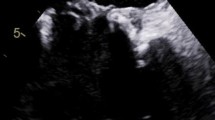
In both pairs of blood cultures growth of T. pyogenes was detected. The species was identified using matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI Biotyper® system, Bruker-Daltonics, Germany). Time to positivity (TTP) was 29 h for the aerobic culture, and 26 h for the anaerobic culture. Blood cultures collected 72 h after initiating antibiotic treatment remained positive. In the wound swab of the basal cell carcinoma growth of Staphylococcus aureus and Candida albicans were noted, but not of T. pyogenes. TTE showed new evidence of mild mitral valve stenosis and tricuspid valve insufficiency. A transesophageal echocardiography (TEE) revealed two vegetations at the posterior leaflet of the mitral valve with a size of 9 × 8 and 15 mm, respectively (Fig. 1). Additionally, mild mitral valve insufficiency and moderate tricuspid valve insufficiency were documented.
The persistently positive blood cultures with isolation of T. pyogenes in consecutive samples, the echocardiographic evidence of mitral valve vegetations, and the presence of fever were consistent with two major and one minor modified Duke Criteria, respectively [10]. Furthermore, somnolence, thrombocytopenia, hypoxemia, and arterial hypotension corresponded to a Sequential Organ Failure Assessment (SOFA) Score of 6 points [11]. A diagnosis of sepsis due to mitral valve endocarditis with T. pyogenes was made.
The antibiotic therapy with piperacillin/tazobactam was deescalated to amoxicillin/clavulanic acid intravenously (2.2 g every four hours) and was switched to targeted treatment with penicillin G (4 million units every four hours) upon receipt of antibiotic sensitivity testing results (minimum inhibitory concentration (MIC) for penicillin G 0,016 mg/L). The case was discussed at the multidisciplinary endocarditis board, and despite the size of > 10 millimeters of one of the vegetations and increased risk of embolism, a decision against surgery was made [12]. This was based on the rapid clinical stabilization, the mild degree of mitral valve insufficiency, and the patient’s comorbidities. Additionally, it took into account the patients’ aversion against surgery and a prolonged hospital stay. Under antibiotic treatment with penicillin G, the patient’s condition gradually improved. Blood cultures drawn ten days after start of the antibiotic treatment remained negative. Clinical signs of septic emboli did not occur at any time.
The patient was discharged after 13 days, receiving an outpatient parenteral antibiotic therapy (OPAT). As he declined treatment with continuous infusion therapy, the antibiotics were switched from penicillin G to ceftriaxone (2 g every 24 h). A follow-up TEE three weeks after diagnosis showed a reduction in size for both vegetations and an unchanged mild insufficiency of the mitral valve. The antibiotic therapy was terminated after a total of six weeks. Blood cultures drawn two weeks after stop of the antibiotic treatment remained negative.
Discussion and conclusions
T. pyogenes is an uncommon cause of infections in humans and the current literature is restricted to case reports. Patients with intraabdominal infection, skin ulcer, sepsis, arthritis, pneumonia, and pyelitis have been described [6, 8, 9, 13,14,15,16,17]. A literature search revealed only eight other reported cases of T. pyogenes endocarditis (Table 1), which occurred in Europe, Asia, and Northern America. To our knowledge, no literature review focusing on endocarditis has been published so far. Patients with T. pyogenes endocarditis were predominantly male (7/9 patients), and their age ranged from 20 to 77 years (median 57 years). The left-sided valves, i.e., aortic and mitral valve were affected in all nine patients.
The reservoirs and routes of transmission of T. pyogenes are still poorly understood [4]. It is thought to be a common colonizer of the skin and mucosal membranes of various domestic and wild animals, leading to endogenous infections by mechanical injuries [5]. The animal-to-animal transmission may occur by contaminated utensils [18], by natural environment [19], or by biting flies (Hydrotaea irritans) [18,19,20]. In humans, T. pyogenes is not part of the normal flora and infections are often associated with animal contact [5]. Interestingly, only in 3/9 patients in our literature review, confirmed exposure to farm animals was reported, which raises the question for an alternative route of transmission. Another patient lived close to a cattle farm but had no direct contact to the animals or their products.
Infections caused by T. pyogenes in humans seem to occur primarily in immunocompromised populations [4], and 5/9 of the patients in our literature review had a functionally impaired immune system. In our patient, history of two active malignancies, ongoing chemo- and immunotherapy, and residency on a cattle farm are predisposing factors for developing a T. pyogenes infection. The open facial wound of the basal cell carcinoma could have been a possible entry site. Since the wound swab did not show growth of T. pyogenes, the site of inoculation could not be determined with certainty.
T. pyogenes shows marked beta-hemolysis on blood agar culture plates and is therefore unlikely to be missed during routine diagnostics in the clinical microbiology laboratory: beta-hemolytic bacteria are regularly further characterized, as this feature can indicate pathogens such as Staphylococcus aureus, Streptococcus pyogenes, and Streptococcus agalactiae. In the present case, the diagnosis was not made upon specific suspicion or instruction; the bacteria were identified during routine analysis.
The increase of antimicrobial resistance of T. pyogenes in animals due to the frequent use of antibiotics in agriculture is an emerging problem [4, 21]. Resistance to tetracyclines, aminoglycosides and trimethoprim-sulfamethoxazole are common [21, 22]. According to susceptibility tests in cattle, penicillin and cephalosporin may retain activity [23]. In our literature review of human patients with T. pyogenes endocarditis, resistance to trimethoprim-sulfamethoxazole was reported for 3/4 patients [7, 24]. Susceptibility to penicillin, ceftriaxone, and macrolides was noted in all cases reporting results of antibiotic resistance testing (6/6). Beta-lactam antibiotics were the predominant antibiotic class used for treatment. In one patient additional surgical treatment was performed. The mortality of endocarditis caused by T. pyogenes was high, with 6/9 patients not surviving the disease. Due to little knowledge about this bacterial species, considering the size of valvular vegetation, we decided to treat our patient for 6 weeks instead of the generally recommended 4 weeks of treatment for native valve endocarditis [12]. Historically, the treatment of Arcanobacterium endocarditis has involved a combination of beta-lactam and aminoglycoside antibiotics, but the clinical benefits of this approach remain uncertain. At the time of endocarditis diagnosis, the patient was clinically stable, afebrile, and willing to finish the antibiotic treatment in an outpatient setting. Adding an aminoglycoside to the prescribed beta-lactam regimen at this stage would have complicated treatment logistics, such as the need for additional timed infusions and therapeutic drug monitoring, potentially leading to additional drug toxicity. In view of the scarce data supporting combination treatment in this situation, we decided to refrain from this approach.
In conclusion, T. pyogenes is a rare causative organism of infectious endocarditis in humans and descriptions are mainly restricted to case reports. In our case report and review of the literature, we found that both an impaired immune system and contact with farm animals might be risk factors for the infection. Growth of T. pyogenes in blood cultures is unlikely to be missed during routine analysis, as it shows marked beta-hemolysis on blood agar culture plates, which generally leads to further characterization of the bacteria. Susceptibility to penicillin, ceftriaxone, and macrolides seems to be retained and the reported mortality in the few patients with T. pyogenes endocarditis is high.
Data Availability
The dataset supporting the conclusion of this article are available on request from the corresponding author JS.
Abbreviations
- CAD:
-
Coronary artery disease
- CRP:
-
C-reactive protein
- CT:
-
Computed tomography
- MIC:
-
Minimum inhibitory concentration
- OPAT:
-
Outpatient parenteral antibiotic therapy
- SOFA:
-
Sequential Organ Failure Assessment
- TEE:
-
Transesophageal echocardiography
- TTE:
-
Transthoracic echocardiogram
- TTP:
-
Time to positivity
References
Reddy C, Cornell C, Fraga A. Transfer of Corynebacterium pyogenes (glage) Eberson to the genus Actinomyces as Actinomyces pyogenes (glage) comb. Nov. Int J Syst Evol MicroBiol. 1982;32(4):419–29.
Yassin A, Hupfer H, Siering C, Schumann P. Comparative chemotaxonomic and phylogenetic studies on the genus Arcanobacterium Collins 1982 emend. Lehnen 2006: proposal for Trueperella gen. nov. and emended description of the genus Arcanobacterium. International Journal of Systematic and Evolutionary Microbiology. 2011;61(6):1265-74.
Deliwala S, Beere T, Samji V, McDonald PJ, Bachuwa G. When zoonotic Organisms Cross Over-trueperella pyogenes Endocarditis presenting as a septic embolic Stroke. Cureus. 2020;12(4):e7740.
Rzewuska M, Kwiecień E, Chrobak-Chmiel D, Kizerwetter-Świda M, Stefańska I, Gieryńska M. Pathogenicity and virulence of Trueperella pyogenes: a review. Int J Mol Sci. 2019;20(11):2737.
Jost BH, Billington SJ. Arcanobacterium pyogenes: molecular pathogenesis of an animal opportunist. Antonie Van Leeuwenhoek. 2005;88:87–102.
Levy CE, Pedro RJ, Von Nowakonski A, Holanda LM, Brocchi M, Ramos MC. Arcanobacterium pyogenes Sepsis in farmer, Brazil. Emerg Infect Dis. 2009;15(7):1131.
Plamondon M, Martinez G, Raynal L, Touchette M, Valiquette L. A fatal case of Arcanobacterium pyogenes endocarditis in a man with no identified animal contact: case report and review of the literature. Eur J Clin Microbiol Infect Dis. 2007;26(9):663–6.
Hermida Ameijeiras A, Romero Jung P, Cabarcos Ortiz de Barron A, Treviño Castallo M, editors. One case of Pneumonia with Arcanobacterium pyogenes. Anales de Medicina Interna; 2004.
Kavitha K, Latha R, Udayashankar C, Jayanthi K, Oudeacoumar P. Three cases of Arcanobacterium pyogenes-associated soft tissue Infection. J Med Microbiol. 2010;59(6):736–9.
Fowler VG Jr., Durack DT, Selton-Suty C, Athan E, Bayer AS, Chamis AL, et al. The 2023 Duke-International Society for Cardiovascular Infectious Diseases Criteria for Infective endocarditis: updating the modified Duke Criteria. Clinical Infectious Diseases; 2023.
Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The Third International Consensus definitions for Sepsis and septic shock (Sepsis-3). JAMA. 2016;315(8):801–10.
Habib G, Lancellotti P, Antunes MJ, Bongiorni MG, Casalta J-P, Del Zotti F, et al. 2015 ESC guidelines for the management of infective endocarditis. Eur Heart J. 2015;36(44):3075–128.
Drancourt M, Oulès O, Bouche V, Peloux Y. Two cases of Actinomyces pyogenes Infection in humans. Eur J Clin Microbiol Infect Dis. 1993;12(1):55–7.
Kotrajaras R, Tagami H. Corynebacterium pyogenes: its pathogenic mechanism in epidemic leg ulcers in Thailand. Int J Dermatol. 1987;26(1):45–50.
Lynch M, O’Leary J, Murnaghan D, Cryan B. Actinomyces pyogenes septic arthritis in a diabetic farmer. J Infect. 1998;37(1):71–3.
Nicholson P, Kiely P, Stret J, Mahalingum K. Septic arthritis due to Actinomyces pyogenes. Injury. 1998;29(8):640–2.
Semaan A, Tayeh GA, Chebel JA, Hallit R, Matta M, Hajj P. Arcanobacterium pyogenes and encrusted pyelitis. Future Sci OA. 2019;6(1):Fso430.
Risseti R, Zastempowska E, Twarużek M, Lassa H, Pantoja J, De Vargas A, et al. Virulence markers associated with Trueperella pyogenes Infections in livestock and companion animals. Lett Appl Microbiol. 2017;65(2):125–32.
Ribeiro MG, Risseti RM, Bolaños CA, Caffaro KA, de Morais AC, Lara GH, et al. Trueperella pyogenes multispecies Infections in domestic animals: a retrospective study of 144 cases (2002 to 2012). Vet Q. 2015;35(2):82–7.
Chirico J, Jonsson P, Kjellberg S, Thomas G. Summer mastitis experimentally induced by Hydrotaea irritans exposed to bacteria. Med Vet Entomol. 1997;11(2):187–92.
Trinh HT, Billington SJ, Field AC, Songer JG, Jost BH. Susceptibility of Arcanobacterium pyogenes from different sources to tetracycline, macrolide and lincosamide antimicrobial agents. Vet Microbiol. 2002;85(4):353–9.
Feßler AT, Schwarz S. Antimicrobial Resistance in Corynebacterium spp., Arcanobacterium spp., and Trueperella pyogenes. Microbiol Spectr. 2017;5(6):5603.
Ashrafi Tamai I, Mohammadzadeh A, Zahraei Salehi T, Mahmoodi P. Genomic characterisation, detection of genes encoding virulence factors and evaluation of antibiotic resistance of Trueperella pyogenes isolated from cattle with clinical metritis. Antonie Van Leeuwenhoek. 2018;111:2441–53.
Zhang H, Shi Z, Yang Q, Chen Y, Xu Y. Endocarditis caused by Arcanobacterium pyogenes. Chin Med J (Engl). 2014;127(19):3510–1.
Jootar P, Gherunpong V, Saitanu K. Corynebacterium pyogenes endocarditis. Report of a case with necropsy and review of the literature. J Med Assoc Thai. 1978;61(10):596–601.
Reddy I, Ferguson DA Jr, Sarubbi FA. Endocarditis due to Actinomyces pyogenes. Clin Infect Dis. 1997;25(6):1476–7.
Gómez-Mateos J, Ubeda A, Florez C, León E. Endocarditis due to Arcanobacterium pyogenes: the first case in Europe. Enferm Infecc Microbiol Clin. 2009;27(4):251–2.
Chesdachai S, Larbcharoensub N, Chansoon T, Chalermsanyakorn P, Santanirand P, Chotiprasitsakul D, et al. Arcanobacterium pyogenes endocarditis: a case report and literature review. Southeast Asian J Trop Med Public Health. 2014;45(1):142–8.
Tang ASO, Leong TS, Tan R, Chua HH, Chew LP. Thrombotic thrombocytopenic purpura-like syndrome associated with Arcanobacterium pyogenes endocarditis in a post-transplant patient: a case report. Med J Malaysia. 2018;73(5):345.
Acknowledgements
Not applicable.
Funding
The authors received no specific funding for this work.
Author information
Authors and Affiliations
Contributions
Authors’ contributions: JS, PL, CMT and ANF cared for the patient and managed the case. JS wrote the paper. All authors were involved in editing the manuscript.
Corresponding author
Ethics declarations
Conflicts of Interest/Competing interests
The authors declare no conflicts of interest.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Written informed consent for publication was obtained from the patient.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Stuby, J., Lardelli, P., Thurnheer, C.M. et al. Trueperella pyogenes endocarditis in a Swiss farmer: a case report and review of the literature. BMC Infect Dis 23, 821 (2023). https://doi.org/10.1186/s12879-023-08810-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12879-023-08810-y