Abstract
Background
An unclassified variant (UV) in exon 1 of the MLH1 gene, c.112A > C, p.Asn38His, was found in six families who meet diagnostic criteria for Lynch syndrome. The pathogenicity of this variant was unknown. We aim to elucidate the pathogenicity of this MLH1 variant in order to counsel these families adequately and to enable predictive testing in healthy at-risk relatives.
Methods
We studied clinical data, microsatellite instability and immunohistochemical staining of MMR proteins, and performed genealogy, haplotype analysis and DNA testing of control samples.
Results
The UV showed co-segregation with the disease in all families. All investigated tumors showed a microsatellite instable pattern. Immunohistochemical data were variable among tested tumors. Three families had a common ancestor and all families originated from the same geographical area in The Netherlands. Haplotype analysis showed a common haplotype in all six families.
Conclusions
We conclude that the MLH1 variant is a pathogenic mutation and genealogy and haplotype analysis results strongly suggest that it is a Dutch founder mutation. Our findings imply that predictive testing can be offered to healthy family members. The immunohistochemical data of MMR protein expression show that interpreting these results in case of a missense mutation should be done with caution.
Similar content being viewed by others
Background
About 3% of all colorectal cancers is due to Lynch syndrome, an autosomal dominant condition caused by germline mutations in one of the DNA mismatch repair (MMR) genes, MLH1, MSH2, MSH6 and PMS2 [1]. Carriers of a mutation in one of these MMR genes have a high risk of developing colorectal cancer, endometrial cancer and also an increased risk of specific other malignancies including ovarian, upper urinary tract, gastric, small intestinal and biliary tract cancer and adenoma or carcinoma of the sebaceous gland [1].
In families who meet the Amsterdam and/or the revised Bethesda criteria [2], tumor examination is indicated including an assay for microsatellite instability (MSI) and immunohistochemistry (IHC) for mismatch repair (MMR) protein expression [2]. When a tumor shows MSI, with or without alterations in immunohistochemical staining of these proteins, mutation analysis of the MLH1, MSH2, MSH6 and PMS2 genes is offered.
Missense mutations comprise about 20% of all pathogenic mutations associated with Lynch syndrome [3, 4]. For most missense mutations, convincing evidence for pathogenicity is lacking, and these are called unclassified variants (UVs) or variants of uncertain clinical significance [3, 5, 6]. To gain more insight in the nature of such an UV, it is useful to study clinical, morphological and molecular features of affected patients and their families.
In this study, we describe six Dutch families with Lynch syndrome and a previously described UV in the MLH1 gene [7–10]. We have combined data from literature with several parameters studied in these families like co-segregation of the MSI/IHC results and the UV with disease, germline mutation analysis of MMR genes, haplotype analysis, geneaology, and germline mutation testing of healthy controls to gain more insight into the clinical significance of this UV.
Methods
Patients and families
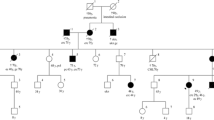
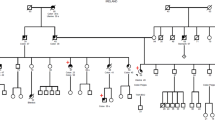
Probands of these six families were referred to a Family Cancer Clinic for genetic counselling because of a personal and/or family history of cancer. A detailed pedigree analysis was performed and, if possible, medical data of affected relatives were verified (Figure 1, Figure 2, Figure 3, Figure 4, Figure 5, Figure 6). For all affected relatives clinical data were recorded including sex, type of cancer, age at diagnosis or at death.
Pedigree from Family 2 (UMCU1). Co = colon cancer. Ut = endometrial cancer. Po = colonic polyp. Br = breast cancer. Cx = cervical cancer. # 'cancer of bones' according to family; no review of pathology. ## 'cancer of bones' according to family; consistent with neuroblastoma according to pathology report.
Microsatellite instability analysis and immunohistochemistry for MMR proteins
For MSI analysis, genomic DNA was isolated from formalin-fixed and paraffin-embedded tumor and normal tissue using standard procedures [11–13]. At least five markers from the Bethesda panel with some additional markers were used to assess MSI. According to international guidelines, a tumor was considered having a MSI-high phenotype when at least two out of five markers were instable [12].
IHC was performed on formalin-fixed, paraffin-embedded tumor and normal tissues. Slides were stained following routine diagnostic procedures [13] using appropriate antibodies against MLH1, MSH2, MSH6 and, when available, PMS2 proteins.
The cancer specimens were acquired from different hospitals where the patients were treated. These pathology departments used a variety of protocols for formalin-fixation and paraffin-embedding. MSI and IHC were conducted on 18 different specimens of 13 patients, including 13 colorectal tumors (two of which only IHC and no MSI), 1 sebaceous gland carcinoma, 1 sebaceous gland adenoma (only MSI, no IHC), 1 endometrial carcinoma, 1 duodenal carcinoma and 1 metastasis presumably of a primary tumor of the gastrointestinal tract or pancreas.
Mutation analysis
When a tumor was classified as MSI high, with or without alterations in immunohistochemical staining of one or more of the MMR proteins, germline mutation analysis was offered. DNA was isolated from peripheral blood lymphocytes of affected patients.
For some patients, a complete germline mutation analysis for not only MLH1 [GenBank: NM_000249] but also MSH2 [GenBank: NM_000251] and MSH6 [GenBank: NM_000179], and in some cases PMS2 [GenBank: NM_000535], was performed (see Additional file 1 - Table S1: Microsatellite instability- and immunohistochemistry results in patients with MLH1 missense mutation).
Genealogy
Extended pedigrees of 6 generations were studied to investigate possible common ancestry of the six families.
Shared haplotype analysis
Haplotype analysis was performed of all probands and affected relatives if available to investigate a possible common origin of the UV. The haplotype consists of 6 VNTR markers, with 3 markers flanking each side of the gene.
Control population
Germline mutation analysis for this UV was performed by DNA sequencing in 94 healthy, anonymous Dutch controls.
Results
Clinical phenotype of families
All six families fulfilled the revised Bethesda criteria, while Families 1, 2 and 4 also fulfilled the Amsterdam II criteria. Figure 1, Figure 2, Figure 3, Figure 4, Figure 5 and Figure 6 show phenotypic details of these families.
Family 1 has features of Muir-Torre syndrome, a phenotypic variant of Lynch syndrome [14].
MSI and IHC
All tested colorectal and other carcinomas showed a MSI-high phenotype. Of 17 tumors that were examined immunohistochemically for MMR protein expression, 6 stained negative for one or more MMR proteins. Only one of these was negative for MLH1. MSI and IHC results are summarized in Table S1 (see Additional file 1 - Table S1: Microsatellite instability- and immunohistochemistry results in patients with MLH1 missense mutation).
Germline mutation analysis
In 13 affected patients an UV in exon 1 of the MLH1 gene, c.112A > C, p.Asn38His, was found. All tested affected patients carried the UV, indicating complete co-segregation of the UV with the disease. In some patients additional germline mutation analysis of the MSH2, MSH6 and/or the PMS2 gene was performed (see Additional file 1 - Table S1: Microsatellite instability- and immunohistochemistry results in patients with MLH1 missense mutation) and no mutations were found in these genes.
Genealogy
All ancestors of the six families originated from the same region (Mid-East Netherlands) and certain family names were frequently found in the different pedigrees. Three families (Families 1, 2 and 6) were shown to have a common ancestor, who linked the independently referred individuals of these families together into a seven-generation family (Figure 7).
Haplotype analysis
All tested patients (n = 12) from six families were shown to share a common allele (see Additional file 2 - Table S2: Haplotype analysis results).
Controls
The MLH1 variant c.112A > C was not detected in DNA of 94 healthy anonymous controls.
Discussion
In this study, we show that the MLH1 alteration, c.112A > C (p.Asn38His), most likely represents a pathogenic missense mutation causing Lynch syndrome.
Criteria for pathogenicity of missense mutations include difference in the chemical properties of the amino acid (Grantham score), evolutionary conservation of the amino acid, absence in the normal population, co-segregation with the disease and association between MSI and/or absence of immunohistochemical staining for the MMR protein in the tumor [4]. From literature and available databases, it is known that this mutation lies in a functional domain of the MLH1 gene and the Asn-38 codon has a full evolutionary conservation in the MLH1 protein [3, 8, 15]. Furthermore, another missense mutation in this codon, p.Asn38Asp, c.112A > G, is proven to be pathogenic and affects the activity of the MLH1 protein: the protein cannot bind Mg++, does not have ATPase activity and cannot correct for a MLH1 deficiency [15]. Lack of mismatch repair activity has also been described recently for this p.Asn38Asp mutation [10]. The variant described in our study induces a greater shift in Grantham score than the variant described by Takahashi [15]. Also, according to SIFT and MAPP-MMR scores this UV should be considered pathogenic [9, 10].
The results of our study show a complete co-segregation between the cancer cases (colorectal, endometrial and other Lynch syndrome related cancers) and the mutation in six families. All tumors show an MSI high phenotype, mostly without alterations in immunohistochemical protein expression. Pathogenic mutations are known to induce MSI and absence of immunohistochemical staining of one or more MMR proteins. In general, immunohistochemical staining is considered to be more sensitive in detecting MMR gene mutations than MSI analysis [16–18]. However, missense mutations can have subtle effects on the protein products of the MMR genes, which can result in loss of function while maintaining intact protein that is able to bind antibodies [19]. Therefore, immunochemistry can show normal staining of the proteins, as seen in the majority of the tumors in this study. In accordance to our findings, other studies have also described tumors showing an MSI high phenotype but normal expression of the MMR proteins [7, 10]. We also observed loss of immunostaining of PMS2 in some tumors. Loss of PMS2 protein expression is often seen in tumors from MLH1 mutation carriers, since PMS2 and MLH1 form heterodimeric complexes in mismatch repair [19]. Surprisingly, we also observed loss of MSH2 and/or MSH6 protein expression. An artefact in the methods used in this study is unlikely since staining in normal tissue of the same patients was normal and others also have reported loss of MSH2 and MSH6 protein expression in a tumor of a MLH1 missense mutation carrier [19] and control tissue showed normal staining. Immunostaining can be dependent on a second hit which can differ for each tumor [20].
Taken together, in the case of missense mutations, which might lead to presence of abnormal protein products that still have binding capacity for antibodies, MSI might be more sensitive for detecting patients with MMR gene mutations than immunohistochemical staining [21, 22].
All tested family members share a common allele and their ancestors originate from the same region in The Netherlands. Genealogical studies showed that three families have a common ancestor. The variant in our families is reported earlier by Van Puijenbroek [7], who studied genome-wide copy neutral loss of heterozygosity (LOH) in a cohort of Dutch familial and sporadic colorectal carcinomas. To our knowledge, this mutation has not been reported in families outside The Netherlands [3, 10, 23, 24]. Together with the genealogy and haplotype analysis, this makes the mutation likely to be a Dutch founder mutation.
In the described families, a Lynch syndrome phenotype is clearly present. In addition to colorectal and endometrial cancer, other cancers were also present in mutation carriers: skin cancers (sebaceous gland carcinoma and squamous cell carcinoma), sarcoma, malignant histiocytoma, ureteral cell cancer, astrocytoma and bone tumors not otherwise specified. Most of these cancers fit within the tumor spectrum of Lynch syndrome. However, sarcoma and histiocytoma are not common tumor types seen in Lynch syndrome. There are some studies that suggest these tumors can be part of the tumor spectrum and a causal relationship with MSH2 has been shown [25–27]. In our study, we confirmed that the patient with the sarcoma is a carrier of the MLH1 mutation. We were not able to confirm that the patient affected with histiocytoma has been a carrier of the MLH1 gene mutation. In Family 1 (VUmc C198), the family history is consistent with Muir-Torre syndrome [14]. Sebaceous gland carcinomas are part of the Lynch syndrome tumor spectrum [28, 29]. Our patients affected with sebaceous gland carcinoma and sebaceous gland adenoma were proven to be carriers of the MLH1 gene mutation and tumor specimens showed a MSI-high phenotype, which confirms the diagnosis Lynch/Muir-Torre syndrome.
Conclusions
The results of our study show compelling evidence that the described missense mutation c.112A > C, p.Asn38His, affecting a strongly conserved position in the ATP-ase domain of the MLH1 gene, is indeed a pathogenic germline alteration which causes Lynch syndrome. Therefore, predictive testing can be offered to non-affected family members. In addition, the results of IHC are not unambiguous and apparently not reliable in diagnosing Lynch syndrome in at least a part of the families with missense mutations in MMR genes.
References
Lynch HT, Lynch PM, Lanspa SJ, Snyder CL, Lynch JF, Boland CR: Review of the Lynch syndrome: history, molecular genetics, screening, differential diagnosis, and medicolegal ramifications. Clin Genet 2009, 76: 1–18. 10.1111/j.1399-0004.2009.01230.x
Vasen HF, Moslein G, Alonso A, Bernstein I, Bertario L, Blanco I, Burn J, Capella G, Engel C, Frayling I, et al.: Guidelines for the clinical management of Lynch syndrome (hereditary non-polyposis cancer). J Med Genet 2007, 44: 353–362. 10.1136/jmg.2007.048991
Woods MO, Williams P, Careen A, Edwards L, Bartlett S, McLaughlin JR, Younghusband HB: A new variant database for mismatch repair genes associated with Lynch syndrome. Hum Mutat 2007, 28: 669–673. 10.1002/humu.20502
Peltomäki P, Vasen H: Mutations associated with HNPCC predisposition -- Update of ICG-HNPCC/INSiGHT mutation database. Dis Markers 2004, 20: 269–276.
Ou J, Niessen RC, Lutzen A, Sijmons RH, Kleibeuker JH, de Wind N, Rasmussen LJ, Hofstra RM: Functional analysis helps to clarify the clinical importance of unclassified variants in DNA mismatch repair genes. Hum Mutat 2007, 28: 1047–1054. 10.1002/humu.20580
Lucci-Cordisco E, Boccuto L, Neri G, Genuardi M: The use of microsatellite instability, immunohistochemistry and other variables in determining the clinical significance of MLH1 and MSH2 unclassified variants in Lynch syndrome. Cancer Biomark 2006, 2: 11–27.
van Puijenbroek M, Middeldorp A, Tops CM, van Eijk R, van der Klift HM, Vasen HF, Wijnen JT, Hes FJ, Oosting J, van Wezel T, Morreau H: Genome-wide copy neutral LOH is infrequent in familial and sporadic microsatellite unstable carcinomas. Fam Cancer 2008, 7: 319–330. 10.1007/s10689-008-9194-8
Gille JJ, Hogervorst FB, Pals G, Wijnen JT, van Schooten RJ, Dommering CJ, Meijer GA, Craanen ME, Nederlof PM, de Jong D, et al.: Genomic deletions of MSH2 and MLH1 in colorectal cancer families detected by a novel mutation detection approach. Br J Cancer 2002, 87: 892–897. 10.1038/sj.bjc.6600565
Chao EC, Velasquez JL, Witherspoon MS, Rozek LS, Peel D, Ng P, Gruber SB, Watson P, Rennert G, Anton-Culver H, et al.: Accurate classification of MLH1/MSH2 missense variants with multivariate analysis of protein polymorphisms-mismatch repair (MAPP-MMR). Hum Mutat 2008, 29: 852–860. 10.1002/humu.20735
Drost M, Zonneveld JB, van Dijk L, Morreau H, Tops CM, Vasen HF, Wijnen JT, de Wind N: A cell-free assay for the functional analysis of variants of the mismatch repair protein MLH1. Hum Mutat 2010, 31: 247–253. 10.1002/humu.21180
Umar A, Boland CR, Terdiman JP, Syngal S, de la Chapelle A, Ruschoff J, Fishel R, Lindor NM, Burgart LJ, Hamelin R, et al.: Revised Bethesda Guidelines for hereditary nonpolyposis colorectal cancer (Lynch syndrome) and microsatellite instability. J Natl Cancer Inst 2004, 96: 261–268. 10.1093/jnci/djh034
Boland CR, Thibodeau SN, Hamilton SR, Sidransky D, Eshleman JR, Burt RW, Meltzer SJ, Rodriguez-Bigas MA, Fodde R, Ranzani GN, Srivastava S: A National Cancer Institute Workshop on Microsatellite Instability for cancer detection and familial predisposition: development of international criteria for the determination of microsatellite instability in colorectal cancer. Cancer Res 1998, 58: 5248–5257.
Wagner A, Hendriks Y, Meijers-Heijboer EJ, de Leeuw WJ, Morreau H, Hofstra R, Tops C, Bik E, Brocker-Vriends AH, van Der Meer C, et al.: Atypical HNPCC owing to MSH6 germline mutations: analysis of a large Dutch pedigree. J Med Genet 2001, 38: 318–322. 10.1136/jmg.38.5.318
Eisen DB, Michael DJ: Sebaceous lesions and their associated syndromes: part II. J Am Acad Dermatol 2009, 61: 563–578. quiz 579–580 10.1016/j.jaad.2009.04.059
Takahashi M, Shimodaira H, Andreutti-Zaugg C, Iggo R, Kolodner RD, Ishioka C: Functional analysis of human MLH1 variants using yeast and in vitro mismatch repair assays. Cancer Res 2007, 67: 4595–4604. 10.1158/0008-5472.CAN-06-3509
Pinol V, Castells A, Andreu M, Castellvi-Bel S, Alenda C, Llor X, Xicola RM, Rodriguez-Moranta F, Paya A, Jover R, Bessa X: Accuracy of revised Bethesda guidelines, microsatellite instability, and immunohistochemistry for the identification of patients with hereditary nonpolyposis colorectal cancer. JAMA 2005, 293: 1986–1994. 10.1001/jama.293.16.1986
Southey MC, Jenkins MA, Mead L, Whitty J, Trivett M, Tesoriero AA, Smith LD, Jennings K, Grubb G, Royce SG, et al.: Use of molecular tumor characteristics to prioritize mismatch repair gene testing in early-onset colorectal cancer. J Clin Oncol 2005, 23: 6524–6532. 10.1200/JCO.2005.04.671
Hofstra RM, Spurdle AB, Eccles D, Foulkes WD, de Wind N, Hoogerbrugge N, Hogervorst FB: Tumor characteristics as an analytic tool for classifying genetic variants of uncertain clinical significance. Hum Mutat 2008, 29: 1292–1303. 10.1002/humu.20894
Raevaara TE, Korhonen MK, Lohi H, Hampel H, Lynch E, Lonnqvist KE, Holinski-Feder E, Sutter C, McKinnon W, Duraisamy S, et al.: Functional significance and clinical phenotype of nontruncating mismatch repair variants of MLH1. Gastroenterology 2005, 129: 537–549.
Goel A, Nguyen TP, Leung HC, Nagasaka T, Rhees J, Hotchkiss E, Arnold M, Banerji P, Koi M, Kwok CT, et al.: De novo constitutional MLH1 epimutations confer early-onset colorectal cancer in two new sporadic Lynch syndrome cases, with derivation of the epimutation on the paternal allele in one. Int J Cancer 2010, in press.
Niessen RC, Berends MJ, Wu Y, Sijmons RH, Hollema H, Ligtenberg MJ, de Walle HE, de Vries EG, Karrenbeld A, Buys CH, et al.: Identification of mismatch repair gene mutations in young patients with colorectal cancer and in patients with multiple tumours associated with hereditary non-polyposis colorectal cancer. Gut 2006, 55: 1781–1788. 10.1136/gut.2005.090159
Lagerstedt Robinson K, Liu T, Vandrovcova J, Halvarsson B, Clendenning M, Frebourg T, Papadopoulos N, Kinzler KW, Vogelstein B, Peltomaki P, et al.: Lynch syndrome (hereditary nonpolyposis colorectal cancer) diagnostics. J Natl Cancer Inst 2007, 99: 291–299. 10.1093/jnci/djk051
Ou J, Niessen RC, Vonk J, Westers H, Hofstra RM, Sijmons RH: A database to support the interpretation of human mismatch repair gene variants. Hum Mutat 2008, 29: 1337–1341. 10.1002/humu.20907
Fokkema IF, den Dunnen JT, Taschner PE: LOVD: easy creation of a locus-specific sequence variation database using an "LSDB-in-a-box" approach. Hum Mutat 2005, 26: 63–68. 10.1002/humu.20201
Hirata K, Kanemitsu S, Nakayama Y, Nagata N, Itoh H, Ohnishi H, Ishikawa H, Furukawa Y: A novel germline mutation of MSH2 in a hereditary nonpolyposis colorectal cancer patient with liposarcoma. Am J Gastroenterol 2006, 101: 193–196. 10.1111/j.1572-0241.2005.00308.x
Lynch HT, Deters CA, Hogg D, Lynch JF, Kinarsky Y, Gatalica Z: Familial sarcoma: challenging pedigrees. Cancer 2003, 98: 1947–1957. 10.1002/cncr.11743
Sijmons R, Hofstra R, Hollema H, Mensink R, van der Hout A, Hoekstra H, Kleibeuker J, Molenaar W, Wijnen J, Fodde R, et al.: Inclusion of malignant fibrous histiocytoma in the tumour spectrum associated with hereditary non-polyposis colorectal cancer. Genes Chromosomes Cancer 2000, 29: 353–355. 10.1002/1098-2264(2000)9999:9999<::AID-GCC1042>3.0.CO;2-T
South CD, Hampel H, Comeras I, Westman JA, Frankel WL, de la Chapelle A: The frequency of Muir-Torre syndrome among Lynch syndrome families. J Natl Cancer Inst 2008, 100: 277–281. 10.1093/jnci/djm291
Lachiewicz AM, Wilkinson TM, Groben P, Ollila DW, Thomas NE: Muir-Torre syndrome. Am J Clin Dermatol 2007, 8: 315–319. 10.2165/00128071-200708050-00008
Acknowledgements
We would like to thank prof. dr. H. te Riele, Netherlands Cancer Institute, Amsterdam, The Netherlands, for critically revising the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
EVR counseled part of the families and acted as first author on this manuscript. MGEMA has been involved in drafting the manuscript and revising it critically for important intellectual content. FBLH carried out part of the MSI/IHC analysis and initiated the first contact between different family cancer clinics to elucidate the nature of this UV. IK counseled part of the families and initiated the first contact between different family cancer clinics to elucidate the nature of this UV. MEVG carried out part of the germline mutation analysis and supervised part of the MSI/IHC analysis. JVE counseled part of these families and initiated revision of tumor material in some of the patients. KSJ carried out part of the MSI/IHC analysis. EFAMH carried out genealogic research on these families. RPS counseled part of the families and made it possible to include two families in this study. YJV carried out part of the germline mutation analysis and supervised part of the MSI/IHC analysis. GJAO carried out part of the MSI/IHC analysis and revised tumor material from some patients. FHM counselled part of the families, made a substantial contribution to conception and design of this study, and has been involved in revising the manuscript critically for important intellectual content. JJPG carried out part of the germline mutation analysis, haplotype analysis and germline mutation analysis of the Dutch controls and has been involved in drafting the manuscript and revising it critically for important intellectual content. All authors read, revised and approved the final manuscript.
Electronic supplementary material
13053_2010_152_MOESM1_ESM.DOC
Additional file 1: Table S1. Microsatellite instability- and immunohistochemistry results in patients with MLH1 missense mutation and affected family members. (DOC 60 KB)
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
This article is published under license to BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
van Riel, E., Ausems, M.G., Hogervorst, F.B. et al. A novel pathogenic MLH1 missense mutation, c.112A > C, p.Asn38His, in six families with Lynch syndrome. Hered Cancer Clin Pract 8, 7 (2010). https://doi.org/10.1186/1897-4287-8-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1897-4287-8-7