Abstract
The rate of HIV-1 gene expression is a key step that determines the kinetics of virus spread and AIDS progression. Viral entry and gene expression were described to be the key determinants for cell permissiveness to HIV. Recent reports highlighted the involvement of miRNA in regulating HIV-1 replication post-transcriptionally. In this study we explored the role of cellular factors required for miRNA-mediated mRNA translational inhibition in regulating HIV-1 gene expression. Here we show that HIV-1 mRNAs associate and co-localize with components of the RNA Induced Silencing Complex (RISC), and we characterize some of the proteins required for miRNA-mediated silencing (miRNA effectors). RCK/p54, GW182, LSm-1 and XRN1 negatively regulate HIV-1 gene expression by preventing viral mRNA association with polysomes. Interestingly, knockdown of RCK/p54 or DGCR8 resulted in virus reactivation in PBMCs isolated from HIV infected patients treated with suppressive HAART.
Similar content being viewed by others
Background
RNA silencing (RNAi) is a new gene regulatory mechanism conserved from plants to humans. RNAi mediators are small non-coding RNAs (sncRNAs) that function through sequence specific mRNA targeting to either induce their degradation and/or inhibit translation [1, 2]. In mammals, RNAi is mediated by different classes of small non-coding RNAs including piRNAs, microRNAs and siRNAs [3–5]. MicroRNAs are produced from a primary transcript (pri-miRNA) which is processed in the nucleus by the microprocessor complex containing RNase Drosha and DGCR8. The resulting product or pre-miRNA is exported to the cytoplasm through the exportin-5 pathway. Cytoplasmic pre-miRNA is processed by typeIII RNase Dicer to miRNA/miRNA* duplex of 19 to 25 nucleotides. miRNA/miRNA* is incorporated into the RNA-Induced Silencing Complex (RISC) where miRNA* is degraded while miRNA serves as a guide for mRNA targeting [2]. Key components of miRISC are proteins of the Argonaute family (Ago1 to Ago4) that are required for miRNA-mediated silencing [6, 7]. To ensure mRNA translational inhibition and decay, miRISC, loaded with miRNA and its mRNA targets, associate with proteins involved in mRNA processing [2]. A key factor in this process is the GW182 protein that interacts directly with Argonaute1 (Ago1) [8], and the human homologs of GW182 that interact with Ago1–4 [9]. GW182 orchestrates both mRNA decapping, through the recruitment of p54/RCK that regulates the activity of the decapping enzymes DCP1/DCP2 [10], and mRNA deadenylation by recruiting the CCR4-NOT1 complex [11]. mRNA decapping and deadenylation leads to mRNA decay through the action of XRN1, a 5'-3' exonuclease [10]. Interestingly, RNAi effectors, including miRNAs and their target mRNAs, Ago proteins, GW182, RCK/p54, LSm-1 and DCP proteins co-localize in cytoplasmic structures called GW-bodies or P-bodies suggesting that miRNA-mediated silencing occurs at these sites [11–15]. Emerging evidence suggests that miRNA-mediated gene regulation serves as a defence mechanism against both RNA and DNA viruses in mammals [16–20]. The present study was designed to explore physical and functional interaction between effectors of miRNA-mediated silencing and HIV-1 replication.
Results and discussion
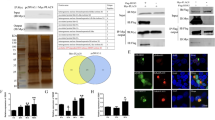
To investigate whether RNAi effectors regulate HIV-1 replication, we analyzed virus replication in cells where expression of RNAi effectors was reduced using specific siRNA. HeLa cells were transfected with siRNA specific to RCK/p54, GW182, LSm-1 or XRN1. As controls, HeLa cells were transfected with scrambled siRNA (Scr) or CDK9 specific siRNA and subsequently infected with HIV-1 (Figure 1a). Knockdown of RCK/p54, GW182, LSm-1 and XRN1 enhanced virus replication by up to 10 fold (Figure 1b). As we have previously shown, knockdown of Drosha [21] and DGCR8 (Figure 1b), the two subunits of the microprocessor complex, increased virus production while knockdown of the CDK9 subunit of the PTEFb complex that is required for viral gene expression, reduced HIV-1 production (Figure 1b). Interestingly, analysis of HIV-1 cytoplasmic mRNA distribution on glycerol gradient showed that knockdown of RCK/p54 shifted HIV-1 mRNA from the non-polysomal fraction to polysomes as compared to control siRNA transfected cells (Figure 2, upper panel). As control, we analyzed the distribution of endogenous mRNA expressed from a gene encoding Hdm2. Knockdown of RCK/p54 did not affect Hdm2 mRNA distribution (Figure 2, lower panel). These experiments show that GW182, RCK/p54, LSm-1 and XRN1, factors required for RNAi, are repressors of HIV-1 gene expression that act by preventing HIV-1 mRNA translation.
miRNA effectors are repressors of HIV-1 replication. HeLa cells were transfected with siRNA as indicated. 48 hours post transfection, cells were analyzed for RCK/p54, LSm-1, GW182, XRN1, DGCR8, DROSHA and CDK9 expression by Western blotting (a), or infected with a single round infectious virus (HIV-1-VSV-luc 200 ng/ml) and cell extracts were measured for luciferase activity 48 hours after infection (b). Results are presented as fold HIV production relative to Scr transfected cells, and data are representative of three independent experiments.
RCK/p54 restricts HIV-1 mRNA association with polysomes. Cytoplasmic extracts from HeLa cells that were transfected with the indicated siRNA and infected with HIV-1-VSVG-luc were run on glycerol gradient (7% to 47%). Fractions were collected and their RNA contents were monitored by measuring absorbance at 254 nm. HIV-1 mRNA (top panel) and Hdm2 mRNA (lower panel) were quantified in all the fractions by Q-RT-PCR using specific oligonucleotides.
We next investigated the physical interaction between RNAi effectors and HIV-1 mRNA. 293 cells were mock transfected or transfected with combinations of pNL4-3, Myc-Ago2, a central component of the RISC complex, or its RNA-binding mutant Myc-Ago2PAZ9 constructs as indicated in figures 3 and 4. First, we verified that Myc-Ago2 and Myc-Ago2PAZ9 were equally expressed (Figure 3a). Second, cytoplasmic extracts were prepared, and a fraction was used for total RNA extraction while the rest was subjected to immunoprecipitation using anti-Myc antibody to purify Myc-Ago2 associated mRNP. Both total RNA (Figure 3b, left panels) and Myc-Ago2 associated RNA (Figure 3b, right panels) were reverse transcribed and subjected to PCR amplification using oligonucleotides specific for HIV-1 TAR RNA (a structured motif associated with all HIV-1 mRNAs) or HIV-1 unspliced mRNA, Hdm2 mRNA or GAPDH mRNA. PCR analysis of total RNA showed that equal amounts of HIV-1, Hdm2 and GAPDH mRNAs were present in all samples (Figure 3b, left panels). HIV-1 mRNAs (both TAR and unspliced) were associated with Myc-Ago2, but not with Myc-Ago2PAZ9 mutant (Figure 3b, right panels). In agreement with the results shown in figure 2, Hdm2 mRNA was not detected in Myc-Ago2 mRNPs, suggesting that under these conditions Hdm2 is not regulated by RNAi. A similar experiment was performed to analyze the association of HIV-1 multispliced mRNA with Myc-Ago2 mRNPs. The RT-PCR reactions were performed in the presence of 32P-α ATP and were analyzed by autoradiography (Figure 3c). HIV-1 multispliced mRNAs associated with Myc-Ago2 (compare lane 3 to 2) and weakly with Myc-Ago2PAZ9 (compare lane 4 to lanes 3 and 2). Co-localization of HIV-1 mRNA and effectors of RNAi such as Ago2 and RCK/p54 within the P-bodies was also observed by immunofluorescence using HIV-1 containing MS2 binding sites and MS2-GFP constructs (Figure 4). Indeed, HIV-1 mRNAs visualized through their binding to MS2-GFP colocalized with endogenous RCK/p54 and ectopically expressed Myc-Ago2 (Figure 4). Our results show that HIV-1 mRNAs physically associate with Ago2, a central component of RISC, and co-localize with cellular proteins required for miRNA-mediated silencing such as RCK/p54 and Ago2 in P-bodies. We observed that all HIV-1 mRNA species associated with RISC. Accordingly, Huang et al. had identified 5 cellular miRNAs able to target the 3'UTR sequence present in all HIV-1 mRNAs [22]. Additionally, other cellular miRNAs able to target regions out side the 3'UTR may also participate [23].
HIV-1 mRNAs associate with Argonaute 2. 293 cells were transfected with HIV-1 molecular clone pNL4-3, Myc-Ago2 or Myc-AgoPAZ9 as indicated. 48 hours later cells were harvested and cytoplasmic extracts were prepared. Total RNA was purified from a fraction of harvested cells while the rest was subjected to immunoprecipitation using anti-Myc antibody. After washing, a fraction was used to analyze the amount of Myc-Ago2 and Myc-Ago2PAZ9 immunoprecipitated by Western blotting (a), and the rest of the Myc-IPs was used for RNA extraction. HIV-1 mRNAs (TAR and unspliced), Hdm2 and GAPDH mRNA were quantified from total RNA (b, left panel) or from Myc immunoprecipitated mRNPs (b, right panel) by RT-PCR using specific oligonucleotides. c) Experiment was performed as in fig 3 except that 293 cells were transfected with HIV-1 ΔPSP which contains a partial gag/pol deletion but retains all the mRNA splicing sites [66], and 32P-labelled nucleotides were used in the PCR reaction. PCR products were visualized by autoradiography.
HIV-1 mRNA co-localizes with RCK/p54 and Ago2. HeLa cells were transfected with Myc-Ago2 expression vector either alone (top panels) or co-transfected with HIV-1 vector containing 24 repeats of MS2 binding sites and MS2-GFP expression vectors [64, 65] (lower panels). Endogenous RCK/p54 and transfected Myc-Ago2 were visualized using specific primary antibodies and appropriate secondary antibodies coupled with Cy5 (shown in blue) and Cy3 (red) respectively. HIV-1 RNA bound to MS2-GFP is shown in green. Green, blue and red merged images are shown.
Emerging evidence suggests the physical and functional interactions between P-bodies and the viral life cycles [24]. Viral mRNA trafficking through P-bodies may represent a pool of translationally repressed viral transcripts otherwise used for efficient packaging or formation of viral-replication complexes. Indeed, yeast retrotransposons Ty1 and Ty3 mRNA associate with P-bodies, and this association is required for efficient retrotransposition [25–27]. In the case of BMV (Brome Mosaic Virus), formation of the virus replication complex occurs in P-bodies [28]. In addition, P-bodies may also function in host defences against viruses and transposable elements. Indeed, the cellular factors APOBEC 3G (A3G) and 3F (A3F), which are viral restriction factors, are found to accumulate in P-bodies [29, 30]. It has been suggested that A3G and A3F mediated HIV-1 restriction may involve viral mRNA targeting to P-bodies leading to their translational inhibition [30]. We, therefore, asked whether P-bodies are positive or negative regulators of HIV-1 replication. Thus, we analyzed HIV-1 replication in cells where P-bodies were disrupted by knocking down RCK/p54 or LSm-1 [31]. HeLa CD4+ cells were transfected with RCK/p54 or LSm-1 specific siRNA or control siRNA. Forty eight hours later, cells were infected with equal amounts of HIV-1 viral particles (as measured by p24 assay). HIV-1 p24 antigen was measured in cell culture supernatant 48 hours post-infection. As shown in figure 5b, knockdown of RCK/p54 or LSm-1 results in enhanced virus production as compared to infection of control siRNA transfected cells. To assess the infectivity of the produced viruses, an equal volume of supernatant from Scr, RCK/p54 and LSm-1 siRNAtransfected cells was used to infect HeLa CD4+ cells, and p24 release in the culture supernatant was measured 48 hours later (Figure 5c). Virus infectivity correlated with the amount of p24 produced (Figure 5b) showing that virions produced in RCK/p54 and LSm-1 knocked down cells are fully competent for replication and have no defect in steps such as RNA packaging. Since the knockdown of RCK/p54 and LSm-1 was shown to result in the disruption of P-bodies, we concluded from these experiments that accumulation of HIV-1 mRNA in P-bodies limits virus replication.
Disruption of P-bodies through knockdown of RCK/p54 and LSm-1 leads to enhanced production of infectious HIV-1 virions. HeLa CD4+ cells were transfected with siRNA as indicated. 48 hours post transfection cells were analyzed for RCK/p54 and LSm-1 expression by Western blotting (a) and infected with equal amounts of HIV-1 (200 ng/ml). b) Virus production was monitored 48 hours post infection by measuring p24 antigen in culture supernatant. c) To analyze the infectivity of new progeny virions, equal volumes of supernatant from siRNA transfected Hela CD4+cells were used to re-infect HeLa CD4+ cells. P24 antigen was measured in culture supernatant 48 hours post infection.
Next, we asked whether A3G-mediated HIV-1 restriction requires effectors of miRNA-mediated mRNA translational inhibition. Thus, we compared A3G-mediated HIV-1 restriction in cells where RCK/p54 or LSm-1 expression was reduced compared to control cells. HeLa cells were transfected with control siRNA or siRNA specific for RCK/p54 or LSm-1 (Figure 6, right panel). Forty-eight hours later, cells were transfected with an HIV-1 molecular clone lacking the vif gene (pNL4-3Δvif) either alone or with wild-type A3G or A3G mutant lacking antiviral activity (A3Gdm). HIV-1 p24 antigen was measured in culture supernatant 48 hours post-transfection. Interestingly, knock down of RCK/p54 or LSm-1 enhanced HIV-1 production regardless of A3G (Figure 6, left upper panel). Similarly, A3G but not A3Gdm reduced virus production regardless of RCK/p54 or LSm-1 expression (Figure 6, left upper panel). These results suggested that RCK/p54 or LSm-1 and A3G -mediated HIV-1 repression involves different mechanisms. We then analyzed the infectivity of HIV-1 produced from siRNA transfected cells. Equal amounts of p24 were used to infected HeLa CD4+ cells, and HIV-1 p24 antigen was measured in culture supernatant 48 hours post-infection. As shown in figure 6 (lower panel), virus produced in Scr siRNA transfected cells in the presence of A3G showed lower infectivity than those produced in its absence or in the presence of A3Gdm. Similar HIV-1-restriction activity of A3G was observed when the virus was produced in RCK/p54 or LSm-1 knocked down cells. This experiment showed that A3G-mediated HIV-1 restriction is independent of RNAi effectors RCK/p54 and LSm-1 and does not require P-bodies.
RNAi effectors and APOBEC 3G-mediated HIV-1 repression involve different pathways. HeLa CD4+ cells were transfected with the indicated siRNA. 48 hours later cells were analyzed for RCK/p54 and LSm-1 expression (right panel) or co-transfected with 1 μg of pNL4-3Δvif (lacking vif gene) and pcDNA or expression vectors for wild-type APOBEC3G or APOBEC3G double mutant lacking both deaminase and antiviral activity, A3G H65R/H257R [63]. HIV-1 production was measured 24 hours post-transfection in culture supernatant by quantifying p24 antigen (top left panel). Numbers on the top of the columns are fold increase relative to the respective Scr. Numbers on the top of Scr samples in A3Gwt and A3Gdm represent fold increase relative to Scr in pCDNA transfected cells. Infectivity assay was performed using equal amounts of p24 antigen to infect HeLa CD4+ cells. HIV-1 p24 antigen was measured 48 hours post infection (lower left panel). A representative experiment out of five is shown.
Taken together, our results show a physically repressive interaction between RNAi effectors and HIV-1 mRNA. Since cellular miRNAs were shown to play a role in HIV-1 latency [22], we asked whether RCK/p54, which is required for miRNA-mediated mRNA translational inhibition, contributes to HIV-1 silencing in vivo. Thus, PBMCs isolated from 3 HAART-treated HIV-1-infected patients with undetectable viremia were transfected with control siRNA or with siRNA specific for Drosha, DGCR8 or RCK/p54. Transfected cells were co-cultured with PHA/IL2-activated PBMCs isolated from healthy donors. Virus production was monitored every 3 days by measuring p24 antigen in the culture supernatant (Figure 7). As we have previously shown, knockdown of Drosha resulted in virus reactivation in PBMCs isolated from 3 HAART-treated HIV-1-infected patients [21]. Remarkably, viral replication from its natural reservoir resumed also when DGCR8 or RCK/p54 was silenced. No virus was isolated from control siRNA transfected PBMCs suggesting that virus production observed in Drosha, DGCR8 and RCK/p54 knock down was not due to actively infected PBMCs relieved from drug pressure. These results show that endogenous levels of Drosha, DGCR8 and RCK/p54 contribute to HIV-1 latency and/or its maintenance in infected patients.
Implication of RNAi in HIV-1 latency. PBMCs were isolated from three patients undergoing active HAART. Isolated PBMCs were transfected with the indicated siRNA and either analyzed for RCK/p54, DGCR8 and DROSHA expression by Western blotting 48 hours after transfection (right panel) or co-cultured with activated PBMCs obtained from healthy donors. Virus replication was monitored every 3 to 4 days post co-culture by measuring p24 antigen in culture supernatant. Shown is the amount of p24 antigen at day 15 post co-culture. No virus was isolated from Scr transfected-PBMCs for up to 27 days.
Conclusion
The outcome of HIV-1 infection results from complex interactions between viral components and host cell factors [32–35]. In most cases, HIV-1 successfully hijacks cellular pathways and bypasses restriction factors for optimal replication leading to continuous rounds of infection, replication, and cell death. Continuous viral replication causes the loss of CD4+T cells and progression to immunodeficiency in infected individuals. HAART treatment revealed the existence of a pool of resting memory CD4+ T cells harbouring integrated, but silent HIV-1 provirus [36, 37]. This latent reservoir is believed to be the major obstacle for virus eradication by HAART. Therefore, it is critical to understand how HIV-1 latency is established and maintained [38]. Post-integration latency takes place at both transcriptional and post-transcriptional levels [39]. Transcriptional latency involves different mechanisms ranging from integration position effect [40–42], limitation in transcription factors [43–46], establishment of chromatin repressive marks and recruitment of chromatin silencers [47–51]. Post-transcriptional silencing involves defects in mRNA export and translation [52–54]. All together, these studies show that HIV-1 post-integration latency is a multi-factorial process. In the present study, we show that HIV-1 gene expression is additionally regulated by the miRNA pathway. HIV-1 mRNA associates with components of the RISC complex by a mechanism that does not involve APOBEC3G, but does need sncRNAs. Accordingly, it has been recently shown that the suppressor of RNAi P19 from tomato bushy stunt virus, known to bind and sequester sncRNAs including miRNA, enhances HIV-1 replication [55]. Additionally, the RNAi suppressor function of HIV-1 Tat [56] could be complemented by VP35 from Ebola virus [57] and the NS3 protein of rice hoja blanca virus through sequesteration of small non-coding RNAs [58]. HIV-1 mRNAs associated with RISC are sequestered in the non-polysomal fraction, thereby preventing translation. In agreement with two previous reports [19, 21, 22], we show that knockdown of RCK/p54, a protein required for miRNA-mediated silencing, led to virus reactivation from PBMCs isolated from HIV-1 infected patients who were undergoing suppressive HAART.
A challenge in AIDS treatment is the need to activate latent viral reservoirs in order to eradicate these viruses through HAART. In this respect, targeting the miRNA processing pathway could offer a strategy that could be exploited to activate latent viral reservoirs, for instance, during HAART. Several molecules have been used to reactivate viral reservoirs [59]. However, none of these approaches provides the sequence specific targeting that can be achieved using siRNA. Recent data suggest that siRNA can be used therapeutically in vivo in certain mouse disease models [60] and more recently in non-human primates [61, 62]. It remains to be explored whether, as suggested here, the in vivo targeting of miRNA-effectors using siRNA can assist in activating latent HIV-1 reservoirs for eradication by HAART.
Methods
Constructs
HIV-1 molecular clone pNL4-3Δvif and expression plasmids for APOBEC3G were gift from Olivier Schwartz (Pasteur, France). APOBEC3G H65R/H257R mutant was previously described [63]. HIV-1 vector containing MS2 binding sites and MS2-GFP expression plasmids [64, 65] were gift from Alessandro Marcello (ICGEB. Trieste, Italy) and Edouard Bertrand (IGMM. Montpellier, France)
Transfections
PBMCs were transfected with siRNA or miRNA using the Nucleofector II Device with the appropriate Nucleofection solution according to the manufacturer's instructions (Amaxa). siRNA corresponding to DGCR8 (5'-CAUCGGACAAGAGUGUGAU(dTdT)-3'), Drosha (5'-CGAGUAGGCUUCGUGACUU(dTdT)-3'), RCK/p54 (5'-GCAGAAACCCUAUGAGAUUUU(dTdT)-3'), LSm-1 (5'-GUGACAUCCUGCCACCUCACUU(dTdT)-3'), GW182 (5'-UAGCGGACCAGACAUUUCU(dTdT)-3'), XRN1 (5'-AGA UGA ACU UAC CGU AGA A(dTdT)-3') and CDK9 (5'-CCAAAGCUUCCCCCUAUAATT(dTdT)-3') were synthesized (MWG). Expression level of knock down proteins was analyzed by Western blotting 48 hours after transfection. Briefly, cell-extracts were resolved on SDS-PAGE gels. Proteins were transferred to PVDF membrane by semi-dry electroblotting and probed overnight at 4°C with the primary antibody (anti-Drosha, LSm1, GW182 (Abcam), DGCR8 (Proteintech Group), anti-RCK/p54 (Bethyl)or anti-CDK9 (Santa Cruz), washed and incubated with the appropriate secondary antibody (Amersham) for 1 hour. Proteins were visualized by chemiluminescence according to the manufacturer's protocol (Pierce).
PBMC isolation and co-culture assay for virus production
Peripheral blood mononuclear cells of HIV-1 infected patients were isolated by lymphocyte separation medium density centrifugation (Lonza). PBMCs from healthy donors were pre-activated using 5 μg/ml PHA (phytohemagglutinin-P, DIFCO)/10 U/ml IL-2 (interleukin-2, Roche) for 72 hours. They were then washed once with PBS and once with RPMI medium before co-culture assay. siRNA transfected HIV-infected PBMC (106 cells/ml) were co-cultured with pre-activated PBMC (106 cells/ml) from the same healthy donor in the presence of 10 U/ml IL-2. The culture medium was collected every 3 or 4 days. Fresh pre-activated healthy PBMCs were added to the culture every 7 days. Viral production was measured by quantifying the amounts of p24 in the culture medium using an ELISA kit (Ingen).
Pseudotyped virion production and single-round infections
The plasmid pNL4-3-env-Luc+ harboring a luciferase gene (obtained from the NIAID AIDS Reagent Program) was co-transfected with the envelope plasmid pMD.2G encoding the G protein of vesicular stomatitis virus (VSV.G) into human embryonic kidney cells-293T. The virions, named HIV-1VSV-Luc, were collected and filtered using 0.45 μm filters 48 hours post-transfection. HeLa or HeLa CD4+ cells were infected over-night at 37°C, washed and resuspended in DMEM containing 10% FCS. Virus production was monitored in culture supernatant by measuring p24 antigen (Ingen) and by following luciferase activity according to the manufacturer's instructions (Promega).
Cytoplasmic extracts analysis on sucrose gradients
To isolate cytoplasmic extracts, cells were lysed for 10 minutes in buffer B (5 mM Tris-HCl pH 7.4, 1.5 mM KCl, 2.5 mM MgCl2, 0.5% NP40 and protease inhibitor). Nuclei were pelleted by centrifugation for 10 minutes at 10,000 rpm. 2 mg of cytoplasmic extracts were loaded on a 7–47% sucrose gradient. Briefly, 5 layers of 7 to 47% sucrose were prepared in sucrose buffer (20 mM Tris-HCl pH7.4, 80 mM NaCl, 5 mM MgCl2, 1 mM DTT and protease inhibitors) and diffused at 4°C for 16 hours to obtain a linear sucrose gradient. 2 mg of cytoplasmic extracts were loaded on the top of the column, and centrifuged for 3 hours at 36,000 rpm in a SW41Ti rotor. After ultracentrifugation, 28 fractions were collected and OD at 254 nm was measured in each fraction using a Nanodrop apparatus (Labtech).
RNA immunoprecipitation
293 cells were grown in 60 mm dishes and transfected with the indicated plasmids using calcium-phosphate. Cells were harvested 48 hours after transfection, lysed for 15 minutes in RIP buffer (20 mM Hepes, pH 7.5, 150 mM NaCl, 2.5 mM MgCl2 × 6H2O, 250 mM sucrose, 0.05% NP40, 0.5% Triton X-100) containing RNASIN (Promega) and 1 mM DTT, and centrifuged to pellet debris. Supernatants were incubated overnight with mouse anti-Myc mAb 9E10 (Amersham) at 4°C followed by 2 hours incubation with protein G-Sepharose. Immunoprecipitates were washed with RIP buffer, and nucleic acids were extracted with phenol/chloroform/isoamyl alcohol, isopropanol-precipitated, ethanol-washed and resuspended in RNase-free water. Total RNA was DNase I treated and reverse-transcribed using SuperScript First-Strand Synthesis System for RT-PCR (Invitrogen). RT products were PCR-amplified using either GAPDH (GAPDH forward: GTA TTG GGC GCC TGG TCA CC; reverse: CGC TCC TGG AAG ATG GTG ATG G), HIV-1 (HIV-1 forward: TAG TGT GTG CCC GTC TGT T; reverse: CTC TGG TTT CCC TTT CGC TTT C or Gag-reverse: GAT GGT TGT AGC TGT CCC AG for unspliced HIV RNA), or HDM2 specific oligonucleotides (HDM2 forward: GTA CCT GAG TCC GAT GAT TCC; reverse: ACC TAC TGA TGG TGC TGT AAC). PCR products were resolved on 1.5% agarose/TAE gels containing ethidium bromide. In vivo splicing assay and oligonucleotides BSS and SJ4.7A have been described [66]
References
Brodersen P, Sakvarelidze-Achard L, Bruun-Rasmussen M, Dunoyer P, Yamamoto YY, Sieburth L, Voinnet O: Widespread translational inhibition by plant miRNAs and siRNAs. Science. 2008, 320: 1185-1190. 10.1126/science.1159151.
Filipowicz W, Bhattacharyya SN, Sonenberg N: Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight?. Nat Rev Genet. 2008, 9: 102-114. 10.1038/nrg2290.
Tam OH, Aravin AA, Stein P, Girard A, Murchison EP, Cheloufi S, Hodges E, Anger M, Sachidanandam R, Schultz RM, Hannon GJ: Pseudogene-derived small interfering RNAs regulate gene expression in mouse oocytes. Nature. 2008, 453: 534-538. 10.1038/nature06904.
Stefani G, Slack FJ: Small non-coding RNAs in animal development. Nat Rev Mol Cell Biol. 2008, 9: 219-230. 10.1038/nrm2347.
Okamura K, Lai EC: Endogenous small interfering RNAs in animals. Nat Rev Mol Cell Biol. 2008, 9: 673-678. 10.1038/nrm2479.
Peters L, Meister G: Argonaute proteins: mediators of RNA silencing. Mol Cell. 2007, 26: 611-623. 10.1016/j.molcel.2007.05.001.
Liu J, Carmell MA, Rivas FV, Marsden CG, Thomson JM, Song JJ, Hammond SM, Joshua-Tor L, Hannon GJ: Argonaute2 is the catalytic engine of mammalian RNAi. Science. 2004, 305: 1437-1441. 10.1126/science.1102513.
Eulalio A, Huntzinger E, Izaurralde E: GW182 interaction with Argonaute is essential for miRNA-mediated translational repression and mRNA decay. Nat Struct Mol Biol. 2008, 15: 346-353. 10.1038/nsmb.1405.
Landthaler M, Gaidatzis D, Rothballer A, Chen PY, Soll SJ, Dinic L, Ojo T, Hafner M, Zavolan M, Tuschl T: Molecular characterization of human Argonaute-containing ribonucleoprotein complexes and their bound target mRNAs. Rna. 2008, 14: 2580-96. 10.1261/rna.1351608.
Rehwinkel J, Behm-Ansmant I, Gatfield D, Izaurralde E: A crucial role for GW182 and the DCP1:DCP2 decapping complex in miRNA-mediated gene silencing. Rna. 2005, 11: 1640-1647. 10.1261/rna.2191905.
Behm-Ansmant I, Rehwinkel J, Doerks T, Stark A, Bork P, Izaurralde E: mRNA degradation by miRNAs and GW182 requires both CCR4:NOT deadenylase and DCP1:DCP2 decapping complexes. Genes Dev. 2006, 20: 1885-1898. 10.1101/gad.1424106.
Pillai RS, Bhattacharyya SN, Artus CG, Zoller T, Cougot N, Basyuk E, Bertrand E, Filipowicz W: Inhibition of translational initiation by Let-7 MicroRNA in human cells. Science. 2005, 309: 1573-1576. 10.1126/science.1115079.
Bhattacharyya SN, Habermacher R, Martine U, Closs EI, Filipowicz W: Relief of microRNA-mediated translational repression in human cells subjected to stress. Cell. 2006, 125: 1111-1124. 10.1016/j.cell.2006.04.031.
Jakymiw A, Lian S, Eystathioy T, Li S, Satoh M, Hamel JC, Fritzler MJ, Chan EK: Disruption of GW bodies impairs mammalian RNA interference. Nat Cell Biol. 2005, 7: 1267-1274. 10.1038/ncb1334.
Liu J, Rivas FV, Wohlschlegel J, Yates JR, Parker R, Hannon GJ: A role for the P-body component GW182 in microRNA function. Nat Cell Biol. 2005, 7: 1261-1266.
Ding SW, Voinnet O: Antiviral immunity directed by small RNAs. Cell. 2007, 130: 413-426. 10.1016/j.cell.2007.07.039.
Gottwein E, Cullen BR: Viral and cellular microRNAs as determinants of viral pathogenesis and immunity. Cell Host Microbe. 2008, 3: 375-387. 10.1016/j.chom.2008.05.002.
Yeung ML, Benkirane M, Jeang KT: Small non-coding RNAs, mammalian cells, and viruses: regulatory interactions?. Retrovirology. 2007, 4: 74-10.1186/1742-4690-4-74.
Han Y, Siliciano RF: Keeping quiet: microRNAs in HIV-1 latency. Nat Med. 2007, 13: 1138-1140. 10.1038/nm1007-1138.
Weinberg MS, Morris KV: Are viral-encoded microRNAs mediating latent HIV-1 infection?. DNA Cell Biol. 2006, 25: 223-231. 10.1089/dna.2006.25.223.
Triboulet R, Mari B, Lin YL, Chable-Bessia C, Bennasser Y, Lebrigand K, Cardinaud B, Maurin T, Barbry P, Baillat V, Reynes J, Corbeau P, Jeang KT, Benkirane M: Suppression of microRNA-silencing pathway by HIV-1 during virus replication. Science. 2007, 315: 1579-1582. 10.1126/science.1136319.
Huang J, Wang F, Argyris E, Chen K, Liang Z, Tian H, Huang W, Squires K, Verlinghieri G, Zhang H: Cellular microRNAs contribute to HIV-1 latency in resting primary CD4+ T lymphocytes. Nat Med. 2007, 13: 1241-1247. 10.1038/nm1639.
Ahluwalia JK, Khan SZ, Soni K, Rawat P, Gupta A, Hariharan M, Scaria V, Lalwani M, Pillai B, Mitra D, Brahmachari SK: Human cellular microRNA hsa-miR-29a interferes with viral nef protein expression and HIV-1 replication. Retrovirology. 2008, 5: 117-10.1186/1742-4690-5-117.
Beckham CJ, Parker R: P bodies, stress granules, and viral life cycles. Cell Host Microbe. 2008, 3: 206-212. 10.1016/j.chom.2008.03.004.
Griffith JL, Coleman LE, Raymond AS, Goodson SG, Pittard WS, Tsui C, Devine SE: Functional genomics reveals relationships between the retrovirus-like Ty1 element and its host Saccharomyces cerevisiae. Genetics. 2003, 164: 867-879.
Devine SE, Boeke JD: Integration of the yeast retrotransposon Ty1 is targeted to regions upstream of genes transcribed by RNA polymerase III. Genes Dev. 1996, 10: 620-633. 10.1101/gad.10.5.620.
Beliakova-Bethell N, Beckham C, Giddings TH, Winey M, Parker R, Sandmeyer S: Virus-like particles of the Ty3 retrotransposon assemble in association with P-body components. Rna. 2006, 12: 94-101. 10.1261/rna.2264806.
Beckham CJ, Light HR, Nissan TA, Ahlquist P, Parker R, Noueiry A: Interactions between brome mosaic virus RNAs and cytoplasmic processing bodies. J Virol. 2007, 81: 9759-9768. 10.1128/JVI.00844-07.
Gallois-Montbrun S, Kramer B, Swanson CM, Byers H, Lynham S, Ward M, Malim MH: Antiviral protein APOBEC3G localizes to ribonucleoprotein complexes found in P bodies and stress granules. J Virol. 2007, 81: 2165-2178. 10.1128/JVI.02287-06.
Wichroski MJ, Robb GB, Rana TM: Human retroviral host restriction factors APOBEC3G and APOBEC3F localize to mRNA processing bodies. PLoS Pathog. 2006, 2: e41-10.1371/journal.ppat.0020041.
Chu CY, Rana TM: Translation repression in human cells by microRNA-induced gene silencing requires RCK/p54. PLoS Biol. 2006, 4: e210-10.1371/journal.pbio.0040210.
Goff SP: Host factors exploited by retroviruses. Nat Rev Microbiol. 2007, 5: 253-263. 10.1038/nrmicro1541.
Brass AL, Dykxhoorn DM, Benita Y, Yan N, Engelman A, Xavier RJ, Lieberman J, Elledge SJ: Identification of host proteins required for HIV infection through a functional genomic screen. Science. 2008, 319: 921-926. 10.1126/science.1152725.
König R, Zhou Y, Elleder D, Diamond TL, Bonamy GM, Irelan JT, Chiang CY, Tu BP, De Jesus PD, Lilley CE, Seidel S, Opaluch AM, Caldwell JS, Weitzman MD, Kuhen KL, Bandyopadhyay S, Ideker T, Orth AP, Miraglia LJ, Bushman FD, Young JA, Chanda SK: Global analysis of host-pathogen interactions that regulate early-stage HIV-1 replication. Cell. 2008, 135: 49-60. 10.1016/j.cell.2008.07.032.
Lama J, Planelles V: Host factors influencing susceptibility to HIV infection and AIDS progression. Retrovirology. 2007, 4: 52-10.1186/1742-4690-4-52.
Chun TW, Engel D, Berrey MM, Shea T, Corey L, Fauci AS: Early establishment of a pool of latently infected, resting CD4(+) T cells during primary HIV-1 infection. Proc Natl Acad Sci USA. 1998, 95: 8869-8873. 10.1073/pnas.95.15.8869.
Finzi D, Hermankova M, Pierson T, Carruth LM, Buck C, Chaisson RE, Quinn TC, Chadwick K, Margolick J, Brookmeyer R, Gallant J, Markowitz M, Ho DD, Richman DD, Siliciano RF: Identification of a reservoir for HIV-1 in patients on highly active antiretroviral therapy. Science. 1997, 278: 1295-1300. 10.1126/science.278.5341.1295.
Marcello A: Latency: the hidden HIV-1 challenge. Retrovirology. 2006, 3: 7-10.1186/1742-4690-3-7.
Lassen K, Han Y, Zhou Y, Siliciano J, Siliciano RF: The multifactorial nature of HIV-1 latency. Trends Mol Med. 2004, 10: 525-531. 10.1016/j.molmed.2004.09.006.
Jordan A, Bisgrove D, Verdin E: HIV reproducibly establishes a latent infection after acute infection of T cells in vitro. Embo J. 2003, 22: 1868-1877. 10.1093/emboj/cdg188.
Han Y, Lin YB, An W, Xu J, Yang HC, O'Connell K, Dordai D, Boeke JD, Siliciano JD, Siliciano RF: Orientation-dependent regulation of integrated HIV-1 expression by host gene transcriptional readthrough. Cell Host Microbe. 2008, 4: 134-146. 10.1016/j.chom.2008.06.008.
De Marco A, Biancotto C, Knezevich A, Maiuri P, Vardabasso C, Marcello A: Intragenic transcriptional cis-activation of the human immunodeficiency virus 1 does not result in allele-specific inhibition of the endogenous gene. Retrovirology. 2008, 5: 98-10.1186/1742-4690-5-98.
Weinberger LS, Burnett JC, Toettcher JE, Arkin AP, Schaffer DV: Stochastic gene expression in a lentiviral positive-feedback loop: HIV-1 Tat fluctuations drive phenotypic diversity. Cell. 2005, 122: 169-182. 10.1016/j.cell.2005.06.006.
Williams SA, Chen LF, Kwon H, Ruiz-Jarabo CM, Verdin E, Greene WC: NF-kappaB p50 promotes HIV latency through HDAC recruitment and repression of transcriptional initiation. Embo J. 2006, 25: 139-149. 10.1038/sj.emboj.7600900.
Kim YK, Bourgeois CF, Pearson R, Tyagi M, West MJ, Wong J, Wu SY, Chiang CM, Karn J: Recruitment of TFIIH to the HIV LTR is a rate-limiting step in the emergence of HIV from latency. Embo J. 2006, 25: 3596-3604. 10.1038/sj.emboj.7601248.
Tyagi M, Karn J: CBF-1 promotes transcriptional silencing during the establishment of HIV-1 latency. Embo J. 2007, 26: 4985-4995. 10.1038/sj.emboj.7601928.
Treand C, du Chene I, Bres V, Kiernan R, Benarous R, Benkirane M, Emiliani S: Requirement for SWI/SNF chromatin-remodeling complex in Tat-mediated activation of the HIV-1 promoter. Embo J. 2006, 25: 1690-1699. 10.1038/sj.emboj.7601074.
du Chéné I, Basyuk E, Lin YL, Triboulet R, Knezevich A, Chable-Bessia C, Mettling C, Baillat V, Reynes J, Corbeau P, Bertrand E, Marcello A, Emiliani S, Kiernan R, Benkirane M: Suv39H1 and HP1gamma are responsible for chromatin-mediated HIV-1 transcriptional silencing and post-integration latency. Embo J. 2007, 26: 424-435. 10.1038/sj.emboj.7601517.
Marban C, Suzanne S, Dequiedt F, de Walque S, Redel L, Van Lint C, Aunis D, Rohr O: Recruitment of chromatin-modifying enzymes by CTIP2 promotes HIV-1 transcriptional silencing. Embo J. 2007, 26: 412-423. 10.1038/sj.emboj.7601516.
Pearson R, Kim YK, Hokello J, Lassen K, Friedman J, Tyagi M, Karn J: Epigenetic silencing of human immunodeficiency virus (HIV) transcription by formation of restrictive chromatin structures at the viral long terminal repeat drives the progressive entry of HIV into latency. J Virol. 2008, 82: 12291-12303. 10.1128/JVI.01383-08.
Sadowski I, Lourenco P, Malcolm T: Factors controlling chromatin organization and nucleosome positioning for establishment and maintenance of HIV latency. Curr HIV Res. 2008, 6: 286-295. 10.2174/157016208785132563.
Pomerantz RJ, Seshamma T, Trono D: Efficient replication of human immunodeficiency virus type 1 requires a threshold level of Rev: potential implications for latency. J Virol. 1992, 66: 1809-1813.
Hermankova M, Siliciano JD, Zhou Y, Monie D, Chadwick K, Margolick JB, Quinn TC, Siliciano RF: Analysis of human immunodeficiency virus type 1 gene expression in latently infected resting CD4+ T lymphocytes in vivo. J Virol. 2003, 77: 7383-7392. 10.1128/JVI.77.13.7383-7392.2003.
Ciuffi A, Bleiber G, Muñoz M, Martinez R, Loeuillet C, Rehr M, Fischer M, Günthard HF, Oxenius A, Meylan P, Bonhoeffer S, Trono D, Telenti A: Entry and transcription as key determinants of differences in CD4 T-cell permissiveness to human immunodeficiency virus type 1 infection. J Virol. 2004, 78: 10747-10754. 10.1128/JVI.78.19.10747-10754.2004.
Qian S, Zhong X, Yu L, Ding B, de Haan P, Boris-Lawrie K: HIV-1 Tat RNA silencing suppressor activity is conserved across kingdoms and counteracts translational repression of HIV-1. Proc Natl Acad Sci USA. 2009, 106: 605-610. 10.1073/pnas.0806822106.
Bennasser Y, Le SY, Benkirane M, Jeang KT: Evidence that HIV-1 encodes an siRNA and a suppressor of RNA silencing. Immunity. 2005, 22: 607-619. 10.1016/j.immuni.2005.03.010.
Haasnoot J, de Vries W, Geutjes EJ, Prins M, de Haan P, Berkhout B: The Ebola virus VP35 protein is a suppressor of RNA silencing. PLoS Pathog. 2007, 3: e86-10.1371/journal.ppat.0030086.
Schnettler E, de Vries W, Hemmes H, Haasnoot J, Kormelink R, Goldbach R, Berkhout B: The NS3 protein of rice hoja blanca virus complements the RNAi suppressor function of HIV-1 Tat. EMBO Rep. 2009, 10: 258-63. 10.1038/embor.2009.6.
Alexaki A, Liu Y, Wigdahl B: Cellular reservoirs of HIV-1 and their role in viral persistence. Curr HIV Res. 2008, 6: 388-400. 10.2174/157016208785861195.
Palliser D, Chowdhury D, Wang QY, Lee SJ, Bronson RT, Knipe DM, Lieberman J: An siRNA-based microbicide protects mice from lethal herpes simplex virus 2 infection. Nature. 2006, 439: 89-94. 10.1038/nature04263.
Krutzfeldt J, Rajewsky N, Braich R, Rajeev KG, Tuschl T, Manoharan M, Stoffel M: Silencing of microRNAs in vivo with 'antagomirs'. Nature. 2005, 438: 685-689. 10.1038/nature04303.
Zimmermann TS, Lee AC, Akinc A, Bramlage B, Bumcrot D, Fedoruk MN, Harborth J, Heyes JA, Jeffs LB, John M, Judge AD, Lam K, McClintock K, Nechev LV, Palmer LR, Racie T, Röhl I, Seiffert S, Shanmugam S, Sood V, Soutschek J, Toudjarska I, Wheat AJ, Yaworski E, Zedalis W, Koteliansky V, Manoharan M, Vornlocher HP, MacLachlan I: RNAi-mediated gene silencing in non-human primates. Nature. 2006, 441: 111-114. 10.1038/nature04688.
Newman EN, Holmes RK, Craig HM, Klein KC, Lingappa JR, Malim MH, Sheehy AM: Antiviral function of APOBEC3G can be dissociated from cytidine deaminase activity. Curr Biol. 2005, 15: 166-170. 10.1016/j.cub.2004.12.068.
Molle D, Maiuri P, Boireau S, Bertrand E, Knezevich A, Marcello A, Basyuk E: A real-time view of the TAR:Tat:P-TEFb complex at HIV-1 transcription sites. Retrovirology. 2007, 4: 36-10.1186/1742-4690-4-36.
Boireau S, Maiuri P, Basyuk E, de la Mata M, Knezevich A, Pradet-Balade B, Backer V, Kornblihtt A, Marcello A, Bertrand E: The transcriptional cycle of HIV-1 in real-time and live cells. J Cell Biol. 2007, 179: 291-304. 10.1083/jcb.200706018.
Jacquenet S, Mereau A, Bilodeau PS, Damier L, Stoltzfus CM, Branlant C: A second exon splicing silencer within human immunodeficiency virus type 1 tat exon 2 represses splicing of Tat mRNA and binds protein hnRNP H. J Biol Chem. 2001, 276: 40464-40475. 10.1074/jbc.M104070200.
Acknowledgements
We are grateful to Kiernan R, Jeang KT, Emiliani S, and Voinnet O for helpful discussions and for critically reading the manuscript. Work in MB's laboratory was supported by ANRS, SIDACTION, ANR and FRM. OM was supported by fellowship from "infectiopôle grand sud". DL was supported by ANRS scholarship. AZ was supported by SIDACTION fellowship.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Competing interests
The authors declare that they have no competing interest.
Authors' contributions
CBC, OL and LD carried out most experiments. TR initiated and performed the experiments shown in figures 3 and 4. AZ participated in experiments analyzing the involvement of APOBEC3. WA, JJM, RJ, LY, and SA participated in some of the experiments such as isolation of PBMCs from HIV-1 infected patients. BY and BM directed, supervised and wrote the manuscript.
Christine Chable-Bessia, Oussama Meziane, Daniel Latreille contributed equally to this work.
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
Open Access This article is published under license to BioMed Central Ltd. This is an Open Access article is distributed under the terms of the Creative Commons Attribution License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Chable-Bessia, C., Meziane, O., Latreille, D. et al. Suppression of HIV-1 replication by microRNA effectors. Retrovirology 6, 26 (2009). https://doi.org/10.1186/1742-4690-6-26
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1742-4690-6-26