Abstract
Despite all the applications of nanotechnology, limited data are available on their environmental and health risks. Many nanomaterials have been developed; however, their safety evaluation is not performed at the same speed. Toxicity can occur at different trophic levels (microorganisms, invertebrates, and vertebrates), and because of that, alternative/complementary models have been successfully employed. This mini-review aims to report some studies that have successfully used different alternative models to assess nanotoxicology and to stimulate their use by other groups of this important field. This will provide more information about nanomaterials and also to evoke studies to improve formulations.

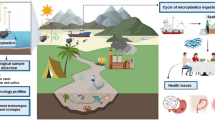
Adapted from Peralta-Videa et al. (2011)
Similar content being viewed by others
References
Agunbiade FO, Moodley B (2014) Pharmaceuticals as emerging organic contaminants in Umgeni River water system KwaZulu-Natal, South Africa. Environ Monit Assess 186:7273–7291. https://doi.org/10.1007/s10661-014-3926-z
Ahamed M, Posgai R, Gorey TJ, Nielsen M, Hussain SM, Rowe JJ (2010) Silver nanoparticles induced heat shock protein 70, oxidative stress and apoptosis in Drosophila melanogaster. Toxicol Appl Pharmacol 242:263–269. https://doi.org/10.1016/j.taap.2009.10.016
Araj Salah-Eddin A, Salem NM, Ghabeish IH, Awwad AM (2015) Toxicity of nanoparticles against Drosophila melanogaster (Diptera: Drosophilidae). J Nanomater. https://doi.org/10.1155/2015/758132
Armstrong N, Ramamoorthy M, Lyon D, Jones K, Duttaroy A (2013) Mechanism of silver nanoparticles action on insect pigmentation reveals intervention of copper homeostasis PloS one 8:e53186. https://doi.org/10.1371/journal.pone.0053186
Ates M, Daniels J, Arslan Z, Farah IO, Rivera HF (2013) Comparative evaluation of impact of Zn and ZnO nanoparticles on brine shrimp (Artemia salina) larvae: effects of particle size and solubility on toxicity. Environ Sci Process Impacts 2013:https://doi.org/10.1039/c1032em30540b https://doi.org/10.1039/c2em30540b
Ates M, Demir V, Arslan Z, Daniels J, Farah IO, Bogatu C (2015) Evaluation of alpha and gamma aluminum oxide nanoparticle accumulation toxicity and depuration in Artemia salina larvae. Environ Toxicol 30:109–118. https://doi.org/10.1002/tox.21917
Avanesian A, Semnani S, Jafari M (2009) Can Drosophila melanogaster represent a model system for the detection of reproductive adverse drug reactions? Drug Discov Today 14:761–766. https://doi.org/10.1016/j.drudis.2009.05.010
Avila D, Helmcke K, Aschner M (2012) The Caenorhabiditis elegans model as a reliable tool in neurotoxicology. Hum Exp Toxicol 31:236–243. https://doi.org/10.1177/0960327110392084
Barros SM et al (2016) A review of solute encapsulating nanoparticles used as delivery systems with emphasis on branched amphipathic peptide capsules. Arch Biochem Biophys 596:22–42. https://doi.org/10.1016/j.abb.2016.02.027
Baun A, Hartmann NB, Grieger K, Kusk KO (2008) Ecotoxicity of engineered nanoparticles to aquatic invertebrates: a brief review and recommendations for future toxicity testing. Ecotoxicology 17:387–395. https://doi.org/10.1007/s10646-008-0208-y
Bianchini MC et al (2016) Peumus boldus (Boldo) aqueous extract present better protective effect than boldine against manganese-induced toxicity in D. melanogaster. Neurochem Res 41:2699–2707. https://doi.org/10.1007/s11064-016-1984-z
Borm PJ et al (2006) The potential risks of nanomaterials: a review carried out for ECETOC Particle and fibre toxicology 3:11. https://doi.org/10.1186/1743-8977-3-11
Boverhof DR, David RM (2010) Nanomaterial characterization: considerations and needs for hazard assessment and safety evaluation. Anal Bioanal Chem 396:953–961. https://doi.org/10.1007/s00216-009-3103-3
Chakraborty C, Sharma AR, Sharma G, Lee S-S (2016) Zebrafish: A complete animal model to enumerate the nanoparticle toxicity. J Nanobiotechnol 14:65. https://doi.org/10.1186/s12951-016-0217-6
Charao MF et al (2015) Caenorhabditis elegans as an alternative in vivo model to determine oral uptake, nanotoxicity, and efficacy of melatonin-loaded lipid-core nanocapsules on paraquat damage. Int J Nanomed 10:5093–5106. https://doi.org/10.2147/IJN.S84909
Chen T-H, Lin C-Y, Tseng M-C (2011) Behavioral effects of titanium dioxide nanoparticles on larval zebrafish (Danio rerio). Mar Pollut Bull 63:303–308. https://doi.org/10.1016/j.marpolbul.2011.04.017
Chen H, Roco MC, Son J, Jiang S, Larson CA, Gao Q (2013) Global nanotechnology development from 1991 to 2012: patents, scientific publications, and effect of NSF funding. Journal of Nanoparticle Research 15:1951. https://doi.org/10.1007/s11051-013-1951-4
Chen H, Wang B, Feng W, Du W, Ouyang H, Chai Z, Bi X (2015) Oral magnetite nanoparticles disturb the development of Drosophila melanogaster from oogenesis to adult emergence. Nanotoxicology 9:302–312. https://doi.org/10.3109/17435390.2014.929189
Chifiriuc MC, Ratiu AC, Popa M, Ecovoiu AA (2016) Drosophotoxicology: an emerging research area for assessing nanoparticles interaction with living organisms. Int J Mol Sci 17:36. https://doi.org/10.3390/ijms17020036
Chi-Hsin H, Zhi-Hong W, Chan-Shing L, Chiranjib C (2007) The Zebrafish model: use in studying cellular mechanisms for a spectrum of clinical disease entities. Curr Neurovascular Res 4:111–120. https://doi.org/10.2174/156720207780637234
Clemente Z, Castro VLSS, Moura MAM, Jonsson CM, Fraceto LF (2014) Toxicity assessment of TiO2 nanoparticles in zebrafish embryos under different exposure conditions. Aquat Toxicol 147:129–139. https://doi.org/10.1016/j.aquatox.2013.12.024
Contado C (2015) Nanomaterials in consumer products: a challenging analytical problem Frontiers. Chemistry. https://doi.org/10.3389/fchem.2015.00048
Contreras EQ, Puppala HL, Escalera G, Zhong W, Colvin VL (2014) Size-dependent impacts of silver nanoparticles on the lifespan, fertility, growth, and locomotion of Caenorhabditis elegans. Environ Toxicol Chem 33:2716–2723. https://doi.org/10.1002/etc.2705
Cozzens S, Cortes R, Soumonni O, Woodson T (2013) Nanotechnology and the millennium development goals: water, energy, and agri-food. J Nanopart Res 15:2001. https://doi.org/10.1007/s11051-013-2001-y
Curtis J, Greenberg M, Kester J, Phillips S, Krieger G (2006) Nanotechnology and nanotoxicology: a primer for clinicians. Toxicol Rev 25:245–260
Das S, Debnath N, Patra P, Datta A, Goswami A (2012) Nanoparticles influence on expression of cell cycle related genes in Drosophila: a microarray-based toxicogenomics study. Toxicol Environ Chem 94:952–957
Elsaesser A, Howard CV (2012) Toxicology of nanoparticles. Adv Drug Deliv Rev 64:129–137. https://doi.org/10.1016/j.addr.2011.09.001
Fan W, Cui M, Liu H, Wang C, Shi Z, Tan C, Yang X (2011) Nano-TiO2 enhances the toxicity of copper in natural water to Daphnia magna. Environ Pollut 159:729–734. https://doi.org/10.1016/j.envpol.2010.11.030
Fangueiro JF, Gonzalez-Mira E, Martins-Lopes P, Egea MA, Garcia ML, Souto SB, Souto EB (2013) A novel lipid nanocarrier for insulin delivery: production, characterization and toxicity testing. Pharm Dev Technol 18:545–549. https://doi.org/10.3109/10837450.2011.591804
Farré M, Gajda-Schrantz K, Kantiani L, Barceló D (2009) Ecotoxicity and analysis of nanomaterials in the aquatic environment. Anal Bioanal Chem 393:81–95. https://doi.org/10.1007/s00216-008-2458-1
Galdiero E et al (2017) Daphnia magna and Xenopus laevis as in vivo models to probe toxicity and uptake of quantum dots functionalized with gH625. Int J Nanomed 12:2717–2731. https://doi.org/10.2147/IJN.S127226
Gambardella C et al (2014) Effects of selected metal oxide nanoparticles on Artemia salina larvae: evaluation of mortality and behavioural and biochemical responses. Environ Monit Assess 186:4249–4259. https://doi.org/10.1007/s10661-014-3695-8
Gorth DJ, Rand DM, Webster TJ (2011) Silver nanoparticle toxicity in Drosophila: size does matter. Int J Nanomed 6:343–350. https://doi.org/10.2147/IJN.S16881
Guarnieri DJ, Heberlein U (2003) Drosophila melanogaster, a genetic model system for alcohol research. Int Rev Neurobiol 54:199–228
Hadrup N, Sharma AK, Poulsen M, Nielsen E (2015) Toxicological risk assessment of elemental gold following oral exposure to sheets and nanoparticles—A review. Regul Toxicol Pharmacol RTP 72:216–221. https://doi.org/10.1016/j.yrtph.2015.04.017
Jacques MT, Oliveira JL, Campos EV, Fraceto LF, Avila DS (2017) Safety assessment of nanopesticides using the roundworm Caenorhabditis elegans. Ecotoxicol Environ Saf 139:245–253. https://doi.org/10.1016/j.ecoenv.2017.01.045
Jang S, Jang H, Lee Y, Suh D, Baik S, Hong BH, Ahn JH (2010) Flexible, transparent single-walled carbon nanotube transistors with graphene electrodes. Nanotechnology 21:425201. https://doi.org/10.1088/0957-4484/21/42/425201
Johnston H et al (2013) Engineered nanomaterial risk lessons learnt from completed nanotoxicology studies: potential solutions to current and future challenges. Crit Rev Toxicol 43:1–20. https://doi.org/10.3109/10408444.2012.738187
Jorgensen EM (2005) Gaba WormBook : the online review of C elegans biology:1-13 https://doi.org/10.1895/wormbook.1.14.1
Juch H, Nikitina L, Debbage P, Dohr G, Gauster M (2013) Nanomaterial interference with early human placenta: Sophisticated matter meets sophisticated tissues. Reprod Toxicol 41:73–79. https://doi.org/10.1016/j.reprotox.2013.05.011
Jung SK et al (2015) Multi-endpoint, high-throughput study of nanomaterial toxicity in Caenorhabditis elegans. Environ Sci Technol 49:2477–2485. https://doi.org/10.1021/es5056462
Kah M (2015) Nanopesticides and nanofertilizers: emerging contaminants or opportunities for risk mitigation? Front Chem. https://doi.org/10.3389/fchem.2015.00064
Lam CW, James JT, McCluskey R, Arepalli S, Hunter RL (2006) A review of carbon nanotube toxicity and assessment of potential occupational and environmental health risks. Crit Rev Toxicol 36:189–217
Libralato G (2014) The case of Artemia spp. in nanoecotoxicology. Mar Environ Res 101:38–43. https://doi.org/10.1016/j.marenvres.2014.08.002
Lin S, Zhao Y, Nel AE, Lin S (2013) Zebrafish: an in vivo model for nano EHS studies. Small (Weinheim an der Bergstrasse, Germany) 9:1608–1618. https://doi.org/10.1002/smll.201202115
Liu X, Vinson D, Abt D, Hurt RH, Rand DM (2009) Differential toxicity of carbon nanomaterials in Drosophila: larval dietary uptake is benign, but adult exposure causes locomotor impairment and mortality. Environ Sci Technol 43:6357–6363
Liu B, Campo EM, Bossing T (2014) Drosophila embryos as model to assess cellular and developmental toxicity of multi-walled carbon nanotubes (MWCNT) in living organisms. PLoS ONE 9:e88681. https://doi.org/10.1371/journal.pone.0088681
Mackay TF, Anholt RR (2006) Of flies and man: Drosophila as a model for human complex traits. Annu Rev Genomics Hum Genet 7:339–367. https://doi.org/10.1146/annurev.genom.7.080505.115758
Madani SY, Mandel A, Seifalian AM (2013) A concise review of carbon nanotube’s toxicology. Nano Rev. https://doi.org/10.3402/nano.v4i0.21521
Manfra L, Savorelli F, Pisapia M, Magaletti E, Cicero AM (2012) Long-term Lethal Toxicity Test with the Crustacean Artemia franciscana. J Vis Exp JoVE:3790 . https://doi.org/10.3791/3790
Miao W, Zhu B, Xiao X, Li Y, Dirbaba NB, Zhou B, Wu H (2015) Effects of titanium dioxide nanoparticles on lead bioconcentration and toxicity on thyroid endocrine system and neuronal development in zebrafish larvae. Aquat Toxicol 161:117–126. https://doi.org/10.1016/j.aquatox.2015.02.002
Misra JR, Horner MA, Lam G, Thummel CS (2011) Transcriptional regulation of xenobiotic detoxification in Drosophila Genes & development 25:1796–1806. https://doi.org/10.1101/gad.17280911
Mora M, Bonilla E, Medina-Leendertz S, Bravo Y, Arcaya JL (2014) Minocycline increases the activity of superoxide dismutase and reduces the concentration of nitric oxide, hydrogen peroxide and mitochondrial malondialdehyde in manganese treated Drosophila melanogaster. Neurochem Res 39:1270–1278. https://doi.org/10.1007/s11064-014-1309-z
Moraes BS, Vieira SM, Salgueiro WG, Michels LR, Colome LM, Avila DS, Haas SE (2016) Clozapine-loaded polysorbate-coated polymeric nanocapsules: physico-chemical characterization and toxicity evaluation in Caenorhabditis elegans model. J Nanosci Nanotechnol 16:1257–1264
Moreno-González R, Campillo JA, García V, León VM (2013) Seasonal input of regulated and emerging organic pollutants through surface watercourses to a Mediterranean coastal lagoon. Chemosphere 92:247–257. https://doi.org/10.1016/j.chemosphere.2012.12.022
Nel AE et al (2009) Understanding biophysicochemical interactions at the nano-bio interface. Nat Mater 8:543–557. https://doi.org/10.1038/nmat2442
Nowack B, Bucheli TD (2007) Occurrence, behavior and effects of nanoparticles in the environment. Environ Pollut 150:5–22. https://doi.org/10.1016/j.envpol.2007.06.006
Nunes BS, Carvalho FD, Guilhermino LM, Van Stappen G (2006) Use of the genus Artemia in ecotoxicity testing. Environ Pollut 144:453–462. https://doi.org/10.1016/j.envpol.2005.12.037
Oberdorster G et al (2005) Principles for characterizing the potential human health effects from exposure to nanomaterials: elements of a screening strategy. Part Fibre Toxicol 2:8. https://doi.org/10.1186/1743-8977-2-8
Oberdörster G, Stone V, Donaldson K (2007) Toxicology of nanoparticles: A historical perspective. Nanotoxicology 1:2–25. https://doi.org/10.1080/17435390701314761
Ong C, Lim JZ, Ng CT, Li JJ, Yung LY, Bay BH (2013) Silver nanoparticles in cancer: therapeutic efficacy and toxicity. Curr Med Chem 20:772–781
Ong C, Yung LY, Cai Y, Bay BH, Baeg GH (2015) Drosophila melanogaster as a model organism to study nanotoxicity. Nanotoxicology 9:396–403. https://doi.org/10.3109/17435390.2014.940405
Ozkan Y, Altinok I, Ilhan H, Sokmen M (2016) Determination of TiO2 and AgTiO2 nanoparticles in Artemia salina: toxicity morphological changes, uptake and depuration. Bull Environ Contam Toxicol 96:36–42. https://doi.org/10.1007/s00128-015-1634-1
Pal A, He Y, Jekel M, Reinhard M (2014) Gin KY-H. Emerging contaminants of public health significance as water quality indicator compounds in the urban water cycle Environment International 71:46–62. https://doi.org/10.1016/j.envint.2014.05.025
Panahifar A, Mahmoudi M, Doschak MR (2013) Synthesis and in vitro evaluation of bone-seeking superparamagnetic iron oxide nanoparticles as contrast agents for imaging bone metabolic activity. ACS Appl Mater Interfaces 5:5219–5226. https://doi.org/10.1021/am4010495
Pandey UB, Nichols CD (2011) Human disease models in Drosophila melanogaster and the role of the fly in therapeutic drug discovery. Pharmacol Rev 63:411–436. https://doi.org/10.1124/pr.110.003293
Parichy DM, Elizondo MR, Mills MG, Gordon TN, Engeszer RE (2009) Normal table of post-embryonic zebrafish development: staging by externally visible anatomy of the living fish. Dev Dyn Off Publ Am Assoc Anat 238:2975–3015. https://doi.org/10.1002/dvdy.22113
Peralta-Videa JR, Zhao L, Lopez-Moreno ML, de la Rosa G, Hong J, Gardea-Torresdey JL (2011) Nanomaterials and the environment: a review for the biennium 2008–2010. J Hazard Mater 186:1–15. https://doi.org/10.1016/j.jhazmat.2010.11.020
Posgai R, Ahamed M, Hussain SM, Rowe JJ, Nielsen MG (2009) Inhalation method for delivery of nanoparticles to the Drosophila respiratory system for toxicity testing. Sci Total Environ 408:439–443. https://doi.org/10.1016/j.scitotenv.2009.10.008
Posgai R, Cipolla-McCulloch CB, Murphy KR, Hussain SM, Rowe JJ, Nielsen MG (2011) Differential toxicity of silver and titanium dioxide nanoparticles on Drosophila melanogaster development, reproductive effort, and viability: size, coatings and antioxidants matter. Chemosphere 85:34–42. https://doi.org/10.1016/j.chemosphere.2011.06.040
Powell MC, Kanarek MS (2006) Nanomaterial health effects–part 1: background and current knowledge. WMJ Off Publ State Med SocWis 05:16–20
Rand MD, Montgomery SL, Prince L, Vorojeikina D (2014) Developmental toxicity assays using the Drosophila model. Curr Protocols Toxicol 59(1):12. https://doi.org/10.1002/0471140856.tx0112s59
Rui Q, Zhao Y, Wu Q, Tang M, Wang D (2013) Biosafety assessment of titanium dioxide nanoparticles in acutely exposed nematode Caenorhabditis elegans with mutations of genes required for oxidative stress or stress response. Chemosphere 93:2289–2296. https://doi.org/10.1016/j.chemosphere.2013.08.007
Samaee S-M, Rabbani S, Jovanović B, Mohajeri-Tehrani MR, Haghpanah V (2015) Efficacy of the hatching event in assessing the embryo toxicity of the nano-sized TiO2 particles in zebrafish: a comparison between two different classes of hatching-derived variables. Ecotoxicol Environ Saf 116:121–128. https://doi.org/10.1016/j.ecoenv.2015.03.012
Savolainen K et al (2013) Nanosafety in Europe 2015–2025: Towards Safe and Sustainable Nanomaterials and Nanotechnology Innovations. EDITA, Helsinki
Schindler DW (1987) Detecting Ecosystem Responses to Anthropogenic Stress. Can J Fish Aquat Sci 44:s6–s25. https://doi.org/10.1139/f87-276
Scholz S, Fischer S, Gündel U, Küster E, Luckenbach T, Voelker D (2008) The zebrafish embryo model in environmental risk assessment—applications beyond acute toxicity testing. Environ Sci Pollut Res 15:394–404. https://doi.org/10.1007/s11356-008-0018-z
Schuler RL, Hardin BD, Niemeier RW (1982) Drosophila as a tool for the rapid assessment of chemicals for teratogenicity. Teratog Carcinog Mutagen 2:293–301
Scown TM, van Aerle R, Tyler CR (2010) Review: Do engineered nanoparticles pose a significant threat to the aquatic environment? Crit Rev Toxicol 40:653–670. https://doi.org/10.3109/10408444.2010.494174
Stewart AM, Grossman L, Nguyen M, Maximino C, Rosemberg DB, Echevarria DJ, Kalueff AV (2014) Aquatic toxicology of fluoxetine: understanding the knowns and the unknowns. Aquat Toxicol 156:269–273. https://doi.org/10.1016/j.aquatox.2014.08.014
Strähle U et al (2012) Zebrafish embryos as an alternative to animal experiments—a commentary on the definition of the onset of protected life stages in animal welfare regulations. Reprod Toxicol 33:128–132. https://doi.org/10.1016/j.reprotox.2011.06.121
Sun TY, Gottschalk F, Hungerbühler K, Nowack B (2014) Comprehensive probabilistic modelling of environmental emissions of engineered nanomaterials. Environ Pollut 185:69–76. https://doi.org/10.1016/j.envpol.2013.10.004
Tan C, Wang W-X (2014) Modification of metal bioaccumulation and toxicity in Daphnia magna by titanium dioxide nanoparticles. Environ Pollut 186:36–42. https://doi.org/10.1016/j.envpol.2013.11.015
Tejeda-Benitez L, Olivero-Verbel J (2016) Caenorhabditis elegans, a biological model for research in toxicology. Rev Environ Contam Toxicol 237:1–35. https://doi.org/10.1007/978-3-319-23573-8_1
Thomaidi VS, Stasinakis AS, Borova VL, Thomaidis NS (2015) Is there a risk for the aquatic environment due to the existence of emerging organic contaminants in treated domestic wastewater? Greece as a case-study. J Hazard Mater 283:740–747. https://doi.org/10.1016/j.jhazmat.2014.10.023
Truong L, Saili KS, Miller JM, Hutchison JE, Tanguay RL (2012) Persistent adult zebrafish behavioral deficits results from acute embryonic exposure to gold nanoparticles. Comp Biochem Physiol 155:269–274. https://doi.org/10.1016/j.cbpc.2011.09.006
Tugulea AM, Bérubé D, Giddings M, Lemieux F, Hnatiw J, Priem J, Avramescu ML (2014) Nano-silver in drinking water and drinking water sources: stability and influences on disinfection by-product formation. Environ Sci Pollut Res Int 21:11823–11831. https://doi.org/10.1007/s11356-014-2508-5
Vanhaecke P, Persoone G, Claus C, Sorgeloos P (1981) Proposal for a short-term toxicity test with Artemia nauplii. Ecotoxicol Environ Saf 5:382–387. https://doi.org/10.1016/0147-6513(81)90012-9
Vecchio G (2015) A fruit fly in the nanoworld: once again Drosophila contributes to environment and human health. Nanotoxicology 9:135–137. https://doi.org/10.3109/17435390.2014.911985
Vega-Alvarez S, Herrera A, Rinaldi C, Carrero-Martinez FA (2014) Tissue-specific direct microtransfer of nanomaterials into Drosophila embryos as a versatile in vivo test bed for nanomaterial toxicity assessment. Int J Nanomed 9:2031–2041. https://doi.org/10.2147/IJN.S56459
Wang J et al (2011) Disruption of zebrafish (Danio rerio) reproduction upon chronic exposure to TiO2 nanoparticles. Chemosphere 83:461–467. https://doi.org/10.1016/j.chemosphere.2010.12.069
Wu Q et al (2013) Comparison of toxicities from three metal oxide nanoparticles at environmental relevant concentrations in nematode Caenorhabditis elegans. Chemosphere 90:1123–1131. https://doi.org/10.1016/j.chemosphere.2012.09.019
Xia G, Liu T, Wang Z, Hou Y, Dong L, Zhu J, Qi J (2016) The effect of silver nanoparticles on zebrafish embryonic development and toxicology. Artif Cells Nanomed Biotechnol 44:1116–1121. https://doi.org/10.3109/21691401.2015.1011803
Yang L et al (2007) Transcriptional profiling reveals barcode-like toxicogenomic responses in the zebrafish embryo. Genome Biol 8:R227–R227. https://doi.org/10.1186/gb-2007-8-10-r227
Yang Y, Zhang C, Hu Z (2013) Impact of metallic and metal oxide nanoparticles on wastewater treatment and anaerobic digestion. Environ Sci Processes Impacts 15:39–48. https://doi.org/10.1039/C2EM30655G
Zhu X, Zhu L, Duan Z, Qi R, Li Y, Lang Y (2008) Comparative toxicity of several metal oxide nanoparticle aqueous suspensions to Zebrafish (Danio rerio) early developmental stage. J Environ Sci Health Part A 43:278–284. https://doi.org/10.1080/10934520701792779
Acknowledgements
Authors would like to thank the funding agencies (CNPq, CAPES, and FAPERGS) for their support. Avila DS is recipient of PQ2 research fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that no conflict of interest is involved in the production of this manuscript.
Rights and permissions
About this article
Cite this article
Ávila, D.S., Roncato, J.F. & Jacques, M.T. Nanotoxicology assessment in complementary/alternative models. Energ. Ecol. Environ. 3, 72–80 (2018). https://doi.org/10.1007/s40974-018-0086-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40974-018-0086-y