Opinion statement
Purpose of review
Environmental surfaces in healthcare facilities, particularly in a patient room, are a critical pathway for healthcare-associated pathogen transmission. Despite well-established guides and recommendations regarding environmental surface cleaning and disinfection, there are several challenges in resource-limited settings. This viewpoint article will discuss the practice of environmental cleaning in resource-limited settings including challenges and relationship between environment and healthcare-associated infections in this setting and outlines pre-requisites to overcome these challenges.
Recent findings
Despite several barriers and challenges, environmental cleaning is a crucial component to help reduce transmission of healthcare-associated infections and multi-drug-resistant pathogens as well as emerging infectious disease-associated pathogens in resource-limited settings. However, there is a need to develop a multi-modal strategy together with a mechanism for monitor and feedback to improve the practices of environmental cleaning in resource-limited settings.
Summary
Additional researches on the barriers and implementation gaps and the role of collaborative network as well as how to apply technology would provide significant insights on the practices of environmental cleaning in resource-limited settings.
Similar content being viewed by others
Introduction
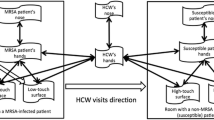
Contaminated environmental surfaces of patient rooms are a critical component in healthcare-associated infection (HAI) transmission [1] and a well-recognized cause of common source nosocomial outbreaks [2, 3]. It has also been well demonstrated that contact with the contaminated environment by healthcare personnel (HCP) is equally as likely as direct contact with a patient to lead to contamination of the healthcare provider’s hands or gloves that may result in patient-to-patient transmission of nosocomial pathogens [4•]. Thus, environmental surfaces and shared noncritical medical equipment may become contaminated with infectious agents and may contribute to cross-transmission by acquisition of transient hand carriage by HCP [5•]. Furthermore, evidence also suggested that a patient admitted to a room previously occupied by a patient with methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE), multi-drug-resistant Acinetobacter baumannii, or Clostridium difficile is at substantially increased risk for acquiring these pathogens [6,7,8,9, 10•]. Therefore, improved cleaning and disinfection of room surfaces will decrease the risk of HAIs in both resource-available and resource-limited settings.
Environmental cleaning practices and challenges in resource-limited settings
Standard methods to improve cleaning/disinfection of environmental surfaces in hospital rooms include improving cleaning/disinfection by environmental service workers through education and feedback on cleaning effectiveness (e.g., use of fluorescent dyes), “no-touch” methods (e.g., UV-C irradiation), and in the future possible use of self-disinfecting surfaces (e.g., impregnating or coating surfaces with heavy metal, such as silver or copper, or a germicide) [11, 12•, 13•]. In resource-limited settings, with the limitation of access to technology, it is important to adhere to basic concepts of environmental cleaning by the use of a risk stratification matrix to help determine the needed frequency of environmental cleaning/disinfection [14]. Table 1 provides an example of such risk stratification. Data on practice of environmental cleaning/disinfection in resource-limited settings is limited. Notable, a national survey in Thailand [15] revealed that > 90% of surveyed hospitals (n = 212) reported having implemented an environmental cleaning/disinfection (ECD) protocol. However, only 55.2% (117/212) had an ECD checklist available and 43.4% (92/212) had a mechanism to audit ECD practices. Among hospitals implementing an ECD protocol, high adherence to implemented ECD protocols and to ECD checklists was documented only in 57.1% (109/192) and 57.3% (67/117), respectively. Because routine and terminal environmental cleaning by environmental service workers are frequently inadequate [16•], these data emphasize the need to have mechanisms for monitoring adherence to ECD protocols as well as to ECD checklists. Interestingly, methods used to audit ECD practices include visual inspection of cleanliness (66%), microbiological monitoring (57%), fluorescence marking (14%), and ATP or bioluminescent testing (10%). It is recognized that barriers and challenges for hospitals to effectively implement ECD protocol include staff concerning, inadequate education provided to staff, lack of resource and funding, time constraints, and uncertainty regarding which cleaning and disinfecting products to use.
Simple suggestions to reduce environmental-related infection in a resource-limited setting, if implementable, include (1) all mattresses should be covered with plastic liner that completely encloses the mattress, which makes cleaning easier and eliminates possibility of mattress becoming contaminated with liquids and harboring bacteria; (2) all chairs should be cleanable (e.g., made of wood or plastic and not covered in fabric which can be lead to VRE transmission); (3) a combined cleaner/disinfectant solution should be used; (4) all touchable surfaces should be cleaned periodically (ideally daily but may vary depending on risk), when soiled and for terminal disinfection (patient discharged); (5) environmental service workers should be trained at the start of employment and yearly and should wear personal protective equipment (e.g., disposable gloves), if available; and (6) each nursing unit should have a checklist which defines which items are cleaned/disinfected by environmental services and which are cleaned/disinfected by nursing.
Environmental cleaning/disinfection and HAIs in resource-limited settings
It is estimated that contaminated environmental surfaces in patient room contributed up to 20% of several key healthcare-associated infection pathogens (e.g., MRSA, VRE, MDR-Gram-negative bacilli, and C. difficile) transmission in intensive care units [6,7,8,9, 10•, 16•]. All of these pathogens have been demonstrated to persist in the environmental from hours to days or, in some cases, up to months [17•]. Further, these pathogens have been demonstrated to frequently contaminate the surface environment and medical equipment in the rooms of colonized or infected patients, to transiently colonized hand of HCP, to be associated with person-to-person transmission, and to cause outbreaks in which environmental transmission deems to play a role [4•, 17•]. Throughout several regions of the world, hospital surfaces have also been demonstrated to be contaminated by several key emerging infectious diseases such as the coronavirus causing severe acute respiratory syndrome (SARS-CoV), novel influenza, and Middle East respiratory syndrome coronavirus (MERS-CoV) [18,19,20,21,22] and had been linked as a cause of person-to-person transmission of these pathogens [22].
In resource-limited settings, despite the limitations of research that has focused on the impact of environmental cleaning/disinfection on HAIs, a few works performed during major floods demonstrated that infection prevention measures featuring advance source control and environmental cleaning can significantly limit transmission of MDR-A. baumannii within a single institution and for the surveyed hospitals [23, 24]. Furthermore, the role of environmental cleaning has been highlighted in one national survey in Thailand that demonstrated the association of the presence of environmental cleaning service with the reduction in MDR-A. baumannii rates [25]. In a recent network meta-analysis [26] that included work from resource-available and resource-limited settings, the impact of environmental cleaning might have some differential effects on different MDR-Gram-negative bacilli. While a combination of infection prevention approaches inclusive of standard practice (e.g., adherence to hand hygiene and contact precaution recommendations), and antibiotic stewardship, environmental cleaning, and source control was the most effective intervention to prevent MDR-Gram-negative bacilli infection, environmental cleaning played a critical role of control of both MDR-A. baumannii and carbapenem-resistant Enterobacteriaceae (CRE) [26]. Together, these data highlight the need to enhance environmental cleaning to help effectively control MDR-Gram-negative pathogens particularly MDR-A. baumannii in resource-limited settings.
Pre-requisite of establishing effective environmental cleaning program in resource-limited settings
In a study that evaluated three-phase cleanliness in a surgical room in a Thai tertiary care center (unpublished data), the investigators tested the cleanliness before cleaning, after manual cleaning, and after a hydrogen peroxide system. Investigators used rodac plate cultures performed before cleaning, after manual cleaning, and after a hydrogen peroxide system. Despite the fact that the hydrogen peroxide system produced the best outcome in reducing bioburden of pathogens, the rodac plates revealed that the highest pathogens bioburden occurred after manual cleaning, which emphasizes the need for education and training for environmental staff in resource-limited settings. Additionally, in a two-stage observational study comparing manual bed cleaning in high- and low-resource settings [27], mattresses in low-resource settings were found to be highly contaminated prior to cleaning. Notable, cleaning significantly reduced biological contamination of mattresses in low-resource settings. After training, the contamination observed after cleaning in both the high- and low-resource settings seemed comparable and cleaning with appropriate types of cleaning materials reduced the contamination of mattresses adequately. Predictors for contaminated mattresses in a low-resource setting included the following: type of product used, type of ward, training, and the level of contamination prior to cleaning. Authors concluded that routine manual cleaning by trained staff can be as effective in a low-resource setting as in a high-resource setting [27]. Thus, a multi-modal strategy inclusive of training of domestic services staff, availability of adequate time to clean beds between patients, and application of the correct type of cleaning products is needed.
As routine and terminal cleaning of patient rooms is frequently inadequate, other key pre-requisites to help implement effective and sustainable environmental cleaning program include having good-to-excellent administration support and having hospital epidemiologist available in resource-limited settings as well as enhancement in hospital safety culture. These factors has been evident in a Thai national survey [15], which reveal a relationship between having good-to-excellent hospital administration support for the infection control program and association with high adherence to implemented ECD protocols and to ECD checklists, while having a hospital epidemiologist was associated with the presence of an ECD checklist as well as regular ECD auditing. National infection control curriculum should, therefore, be created to provide formal training for post-graduate physicians in infection prevention in order to become a hospital epidemiologist, as part of strategic infection prevention plans to improve national ECD practices. Lastly, strong organization safety culture and participating in collaborative network to reduce healthcare-associated infections may also play a significant role to help enhance the effort of HAIs and MDR pathogens control in resource-limited settings, as suggested by a national survey in the USA, Japan, and Thailand [28,29,30].
Future researches
Despite the critical role of environmental cleaning in reducing HAIs and MDR pathogens, there are some gaps on knowledge in this area in resource-limited settings. These gaps include the role of environmental cleaning on the transmission of HAIs and MDR pathogens in resource-limited settings where the magnitudes of problems may be more than resource-available settings. Researches on the barriers and implementation gaps should provide some insights to help improve the unmet needed pre-requisite for the environmental cleaning program as well as to help device appropriate intervention to improve environmental cleanings in resource-limited settings. The role of collaborative network emphasizing on basic environmental cleaning principle will also be needed to be explored. Lastly, because in most of the resource-limited setting, most patients are in multi-bed units making the use of UV devices and hydrogen peroxide systems become impractical, the study on the appropriate technology that is suitable for infrastructure, climate, and resource as well as their cost-effectiveness would provide more insights into the science of environmental cleaning in resource-limited settings.
Conclusions
Overwhelming evidences suggest the key role of environmental cleaning to help reduce transmission of HAIs and MDR pathogens as well as emerging infectious disease-associated pathogens. Despite several barriers and challenges, several data suggest a successful implementation of environmental cleaning program in resource-limited settings. With the limited resource, resource-limited settings should adhere to basic recommendation for environmental cleaning and selecting non-technology strategy first. Mechanisms to monitor, audit, and feedback should be available. Several pre-requisites inclusive of leadership support, availability of hospital epidemiologist, appropriate education, and training for staff as well as implementation of multi-modal strategy are essential for establishing an effective environmental cleaning program in resource-limited settings.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance
Datta R, Platt R, Yokoe DS, Huang SS. Environmental cleaning intervention and risk of acquiring multidrug-resistant organisms from prior room occupants. Arch Intern Med. 2011;171(6):491–4. https://doi.org/10.1001/archinternmed.2011.64.
Sehulster L, Chinn RY. Guidelines for environmental infection control in health-care facilities. Recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee (HICPAC). MMWR Recomm Rep. 2003;52(RR-10):1–42.
PIDAC - Best practices for environmental cleaning for prevention and control of infections (2009). Available from: http://www.oahpp.ca/resources/documents/pidac/Best%20Practices%20for%20Environmental%20Cleaning.pdf
• Weber DJ, Rutala WA. Understanding and preventing transmission of healthcare-associated pathogens due to contaminated hospital environment. Infect Control Hosp Epidemiol. 2013;34(05):449–52. This is a comprehensive review of the evidence demonstrating the importance of the contaminated surface environment in the transmission of healthcare-associated pathogens. https://doi.org/10.1086/670223.
• Huslage K, Rutala WA, Sickbert-Bennett E, Weber DJ. A quantitative approach to defining hi-touch surfaces in hospitals. Infect Control Hosp Epidemiol. 2010;31(08):850–3. This paper delineates which objects in both the ICU and non-ICU are frequently touched. https://doi.org/10.1086/655016.
Huang SS, Datta R, Piatt R. Risk of acquiring antibiotic-resistant bacteria from prior room occupants. Arch Intern Med. 2006;166(18):1945–51. https://doi.org/10.1001/archinte.166.18.1945.
Drees M, Snydman DR, Schmid CH, Barefoot L, Hansjosten K, Vue PM, et al. Prior environmental contamination increases the risk of acquisition of vancomycin resistant enterococci. Clin Infect Dis. 2008;46(5):678–85. https://doi.org/10.1086/527394.
Nseir S, Blazejewski C, Lubret R, Wallet F, Courcol R, Durocher A. Risk of acquiring multidrug-resistant gram-negative bacilli from prior room occupants in the intensive care unit. Clin Microbiol Infect. 2011;17(8):1201–8. https://doi.org/10.1111/j.1469-0691.2010.03420.x.
Shaughnessy MK, Micielli RL, DePestel DD, Arndt J, Strachan CL, Welch KB, et al. Evaluation of hospital room assignment and acquisition of Clostridium difficile infection. Infect Control Hosp Epidemiol. 2011;32(03):201–6. https://doi.org/10.1086/658669.
• Anderson DJ, Chen LF, Weber DJ, Moehring RW, Lewis SS, Triplett PF, et al. Enhanced terminal room disinfection and acquisition and infection caused by multidrug-resistant organisms and Clostridium difficile (the Benefits of Enhanced Terminal Room Disinfection study): a cluster-randomised, multicentre, crossover study. Lancet. 2017;389(10071):805–14. This is a complex study which demonstrated the benefits of “enhanced” terminal disinfection to decrease HAIs. https://doi.org/10.1016/S0140-6736(16)31588-4.
Hota B, Blom DW, Lyle EA, Weinstein RA, Hayden MK. Interventional evaluation of environmental contamination by vancomycin-resistant enterococci: failure of personnel, product, or procedure? J Hosp Infect. 2009;71(2):123–31. https://doi.org/10.1016/j.jhin.2008.10.030.
• Weber DJ, Rutala WA. Self-disinfecting surfaces: review of current methodologies and future prospects. Am J Infect Control. 2013;41(5 Suppl):S31–5. A comprehensive paper which reviewed possible methods to achieve “self-disinfecting” surfaces that could be used in a patient room. https://doi.org/10.1016/j.ajic.2012.12.005.
• Weber DJ, Kanamori H, Rutala WA. ‘No touch’ technologies for environmental decontamination: focus on ultraviolet devices and hydrogen peroxide systems. Curr Opin Infect Dis. 2016;29(4):424–31. This paper reviews the evidence that demonstrates that “no touch” methods for terminal room disinfection reduce HAIs. https://doi.org/10.1097/QCO.0000000000000284.
Ling ML, Apisarnthanarak A, Thu LT, Villanueva V, Pandjaitan C, Yusof MY. APSIC guidelines for environmental cleaning and decontamination. Antimicrob Resist Infect Control. 2015;4(1):58. https://doi.org/10.1186/s13756-015-0099-7.
Apisarnthanarak A, Weber DJ, Ratz D, Saint S, Khawcharoenporn T, Greene MT. National survey of environmental cleaning and disinfection in hospitals in Thailand. Infect Control Hosp Epidemiol. 2017;38(10):1250–3. https://doi.org/10.1017/ice.2017.171.
• Carling PC, Parry MF, von Beheren SM, Healthcare Environ Environmental Hygiene Study Group. Identifying opportunities to enhance environmental cleaning in 23 acute care hospitals. Infect Control Hosp Epidemiol. 2008;29(01):1–7. This paper demonstrates methods that improve terminal room disinfection. https://doi.org/10.1086/524329.
• Rutala WA, Weber DJ; Healthcare Infection Control Practices Ad Advisory Committee. Guideline for disinfection and sterilization in healthcare facilities. 2008. http://www.cdc.gov/hicpac/pubs.html. Accessed 25 Oct 2016. This paper is the official CDC Guideline on sterilization and disinfection of environmental surfaces and medical equipment/devices.
Dowell SF, Simmerman JM, Erdman DD, et al. Severe acute respiratory syndrome coronavirus on hospital surfaces. 2004;39:652–657.
Chen YC, Chang SC, Tsai KS, Lin FY. Certainties and uncertainties facing emerging respiratory infectious diseases: lessons from SARS. J Formos Med Assoc. 2008;107(6):432–42. https://doi.org/10.1016/S0929-6646(08)60150-3.
Simmerman JM, Suntarattiwong P, Levy J, Gibbons RV, Cruz C, Shaman J, et al. Influenza virus contamination of common household surfaces during the 2009 influenza A (H1N1) pandemic in Bangkok, Thailand: implications for contact transmission. Clin Infect Dis. 2010;51(9):1053–61. https://doi.org/10.1086/656581.
Shehata MM, Gomaa MR, Ali MA, Kayali G. Middle East respiratory syndrome coronavirus: a comprehensive review. Front Med. 2016;10(2):120–36. https://doi.org/10.1007/s11684-016-0430-6.
Kim SH, Chang SY, Sung M, Park JH, Bin Kim H, Lee H, et al. Extensive viable Middle East respiratory syndrome (MERS) coronavirus contamination in air and surrounding environment in MERS isolation wards. Clin Infect Dis. 2016;63(3):363–9. https://doi.org/10.1093/cid/ciw239.
Apisarnthanarak A, Li Yang H, Warren DK. Termination of an extreme-drug resistant-Acinetobacter baumannii outbreak in a hospital after flooding: lessons learned. Clin Infect Dis. 2012;55(11):1589–90. https://doi.org/10.1093/cid/cis726.
Apisarnthanarak A, Khawcharoenporn T, Mundy LM. Patterns of nosocomial infections, multidrug-resistant microorganisms, and mold detection after extensive black-water flooding: a survey from central Thailand. Infect Control Hosp Epidemiol. 2013;34(08):861–3. https://doi.org/10.1086/671277.
Apisarnthanarak A, Ratz D, Khawcharoenporn T, Patel PK, Weber DJ, Saint S, et al. National survey of practices to prevent methicillin-resistant Staphylococcus aureus and multidrug-resistant Acinetobacter baumannii in Thailand. Clin Infect Dis. 2017;64(Suppl 2):S161–6. https://doi.org/10.1093/cid/cix045.
Teerawattanapong N, Kengkla K, Dilokthornsakul P, Saokaew S, Apisarnthanarak A, Chaiyakunapruk N. Prevention and control of multidrug-resistant gram-negative bacteria in adult intensive care units: a systematic review and network meta-analysis. Clin Infect Dis. 2017;64(Suppl 2):S51–60. https://doi.org/10.1093/cid/cix112.
Hopman J, Hakizimana B, Meintjes WA, et al. Manual cleaning of hospital mattresses: an observational study comparing high- and low-resource settings. J Hosp Infect. 2016;92(1):14–8. https://doi.org/10.1016/j.jhin.2015.09.017.
Apisarnthanarak A, Greene MT, Kennedy EH, Khawcharoenporn T, Krein S, Saint S. National survey of practices to prevent healthcare-associated infections in Thailand: the role of safety culture and collaboratives. Infect Control Hosp Epidemiol. 2012;33(07):711–7. https://doi.org/10.1086/666330.
Krein SL, Fowler KE, Ratz D, Meddings J, Saint S. Preventing device-associated infections in US hospitals: national surveys from 2005 to 2013. BMJ Qual Saf. 2015;24(6):385–92. https://doi.org/10.1136/bmjqs-2014-003870.
Sakamoto F, Sakihama T, Saint S, Greene MT, Ratz D, Tokuda Y. Health care-associated infection prevention in Japan: the role of safety culture. Am J Infect Control. 2014;42(8):888–93. https://doi.org/10.1016/j.ajic.2014.05.018.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Apisarnthanarak declares that he has no conflicts of interest. Dr. Weber declares that he has no conflicts of interest.
Human and animal rights and informed consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Infection Prevention and Safety in Low and Middle Income Countries
Rights and permissions
About this article
Cite this article
Apisarnthanarak, A., Weber, D.J. Environmental Cleaning in Resource-Limited Settings. Curr Treat Options Infect Dis 10, 48–54 (2018). https://doi.org/10.1007/s40506-018-0149-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40506-018-0149-9