Abstract
Purpose
Considering the re-emergence of poliomyelitis (PM) in non-endemic regions, it becomes apparent that vaccine preventable diseases can rapidly develop epi- or even pandemic potential. Evaluation of the current vaccination status is required to inform patients, health care providers and policy makers about vaccination gaps.
Methods
Between October 28 2022 and November 23 2022, 5,989 adults from the VACCELEREATE Volunteer Registry completed an electronic case report form on their previous PM vaccine doses including number, types/-valencies and the time of administration based on their vaccination records. A uni-/multivariable regression analysis was performed to assess associations in participant characteristics and immunization status.
Results
Among German volunteers (n = 5,449), complete PM immunization schedule was found in 1,981 (36%) participants. Uncertain immunization, due to unknown previous PM vaccination (n = 313, 6%), number of doses (n = 497, 9%), types/-valencies (n = 1,233, 23%) or incoherent immunization schedule (n = 149, 3%) was found in 40% (n = 2,192). Out of 1,276 (23%) participants who reported an incomplete immunization schedule, 62 (1%) never received any PM vaccine. A total of 5,074 (93%) volunteers reported having been vaccinated at least once and 2,087 (38%) indicated that they received vaccination within the last ten years. Female sex, younger age, as well as availability of first vaccination record were characteristics significantly associated with complete immunization (p < 0.001).
Conclusion
Full PM immunization schedule was low and status frequently classified as uncertain due to lack of details on administered doses. There is an obviousneed for improved recording to enable long-term access to detailed vaccination history in the absence of a centralized immunization register.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The coronavirus disease 2019 (COVID-19) pandemic has shown the global impact of emerging infectious diseases, including the burden on healthcare systems, socio-psychological implications, and economic damage [1, 2]. The global response to public health emergencies derives from a broad range of instruments implemented by means of pandemic preparedness [2]. Various response strategies, including masking, quarantining, testing and other containment efforts achieved to reduce burden and spread of COVID-19, but in particular the introduction of effective vaccines had a significant impact on reducing hospitalizations and mortality [2, 3]. Adequate and reliable documentation of administered doses is crucial not only with respect to vaccine surveillance and monitoring, but also for individual knowledge, and safety [4,5,6].
Poliomyelitis (PM) is a highly contagious infectious disease caused by poliovirus (PV), a non-enveloped ribonucleic acid (RNA) enterovirus, classified as member of the picornaviridae family [7]. It encompasses three distinct serotypes and is able to affect motor neurons of the spinal cord and brainstem, which may result in irreversible paralysis and even death [8].
Since the World Health Assembly launched the global PM eradication plan in 1988, when over 350.000 PM cases were diagnosed across 125 countries, considerable progress has been achieved [9, 10]. Most regions were certified free of wildtype poliovirus (WPV) as result of global immunization programs and mass vaccination campaigns [9, 11]. However, WPV type 1 is still circulating in Afghanistan and circulating vaccine-derived polioviruses (cVDPV), particularly type 1 and 2, are emerging in Pakistan and Ukraine, even causing outbreaks [9, 12, 13]. In July 2022, one case of PM with paralysis related to cVDPV type 2 was confirmed in an unvaccinated adult in the State of New York [14]. Moreover, cVDPV type 2 in London, United Kingdom (UK) sewage samples raised public health concerns [15]. Considering this re-emergence of PM in non-endemic regions, it becomes apparent that vaccine preventable diseases other than COVID-19, and even those assumed to be largely controlled, can rapidly develop epi- or even pandemic potential.
While PM vaccination rates in children are monitored by the RKI (German federal government agency and research institute for disease control and prevention) and ASHIP (Association of Statutory Health Insurance Physicians) in Germany, the last evaluation in adults occurred in 2013 as part of the German Health Interview and Examination Survey for Adults (DEGS1) [16, 17]. Current vaccination coverage among the German population may differ largely from these records due to vaccine hesitancy, increasing migration from Middle East and Ukraine and non-harmonized European vaccination schedules [5, 18,19,20].
We therefore performed this population-based survey to give insights into the vaccination coverage for PM in the German population and identify knowledge gaps within the monitoring and recording system.
Methods
To assess the PM vaccination status in adults, the VACCELERATE Volunteer Registry (VR) was consulted. VACCELERATE is a European Commission funded consortium dedicated to vaccine research, that runs a VR to promote clinical studies and citizen science initiatives. Volunteers can sign up via an electronic survey (https://vaccelerate.eu/volunteer-registry-2) [21]. With over 30,000 adult volunteers in 16 different European countries by October 2022, it provided a considerable number of potential participants, thus allowing a broad evaluation. The VR was approved by the Ethics Commission of Cologne University’s Faculty of Medicine (Germany) (identifier 20–1536).
All registered adults (n = 33,207) were invited by e-mail to complete an electronic case report form (eCRF) to collect information on past PM vaccinations including all dates, as well as administered vaccine types and valences. Children were not included as specific data regarding their vaccination rates are available from the RKI and ASHIP vaccination monitoring, assessing data from school entry examinations and vaccination services from the ASHIP in Germany [6, 22, 23]. The proportion of registered children originating from other European countries involved was comparatively small. The eCRF was made available in three different languages (English/German/Greek) as adult VACCELERATE volunteers originated mainly from Germany (n = 31,917), Ireland (n = 678), Austria (n = 194) and Cyprus (n = 156) at time of consultation. Basic demographic data, as well as medical history including underlying diseases were matched from their registration entries. Regional differences were analyzed by comparing geographical distribution of vaccination status by using postal codes.
To assess whether data obtained was complete, a question addressing the first entry in the oldest available vaccination certificate was included. To assess maintenance/completeness of vaccination records we compared year of birth and year of first entry in the oldest available vaccination card.
The eCRF provided a fill in aid, including label names of all PM vaccines that had been available in the four countries from 1955 onwards. Additionally, explanations on vaccine types/-valencies were included and participants were able to reply by e-mail to send inquiries in case of uncertainties. Incoming replies were processed on a daily basis during the study period.
To directly address the issue of potential vaccination gaps, automated evaluation of vaccination status was provided after eCRF was completed. “Complete PM immunization status” was defined as full primary series comprising three trivalent doses and at least one booster vaccination (Supplementary Fig. 1) [24, 25]. In addition to that specific populations with increased risk of exposure to poliovirus, e.g. healthcare workers are recommended to receive a booster dose of inactivated poliovirus (IPV) vaccine or an IPV-containing vaccine every 10 years [24]. Based on entries, participants received preprogrammed replies to primarily inform them in case of incomplete immunization, defined as three or less trivalent (or equivalent mono- or bivalent) vaccines. If classification was not possible, vaccination status was classified as uncertain.
Vaccination status and primary series (basic immunization) was classified as complete, uncertain or incomplete. To further assess the group with “uncertain vaccination status”, additional analyses regarding underlying causes for uncertainty and probability for completeness despite minor or major inconsistencies were performed. To assess the probability for completeness, three different criteria including number of doses, year of application of individual doses, as well as the coherence of recommended vaccination schedules were considered as detailed below. If one or more criteria were met, uncertain vaccination status was categorized as probably complete.
To our knowledge, formerly recommended PM vaccination schedules consisted of up to six vaccines including primary series (i.e., former German Democratic Republic (GDR) vaccination schedule: three monovalent oral attenuated poliomyelitis vaccine (OPV) doses against each serotype I-III, followed by two trivalent doses) and a booster dose [24]. Therefore, one criterion for probable completeness was defined as ≥ 6 doses regardless of otherwise lacking and/or incoherent data. The second criterion, addressing the year of vaccination, was based upon the Standing Committee on Vaccination (STIKO) of the Federal Republic of Germany (FRG) recommending the sole use of trivalent inactivated poliomyelitis vaccine (IPV) to prevent cVDPV from 1998 [24]. It was assumed that all vaccines received after 1998 were IPV. If four trivalent doses were achieved, uncertain immunization was categorized as probably complete. As vaccines of different types and valences were eventually combined depending on time and place of administration, we valued mono-/bivalent vaccines differently than trivalent doses (i.e., three monovalent doses equal one trivalent dose) to assess the coherence to recommended vaccination schedules [24]. Besides the GDR vaccination schedule, other former recommended vaccination schemes including the FRG vaccination scheme with three trivalent doses for primary series were considered according to archived vaccination schedule tables by the STIKO [24, 26]. For this sub analysis, the pattern of reported vaccines was holistically assessed. If an immunization schedule was correctly pursued, but minor specifications were lacking, it was assumed that the vaccination schedule was completed as recommended (e.g., complete primary series, followed by PM vaccine of unknown type/valency) and therefore categorized as probably complete.
To provide a scale projection specific for Germany, as well as assessment as to the representativity in terms of sex- and age-related, as well as regional distribution of the cohort as compared to the general population, the most recently available number of registered inhabitants by sex, age groups and federal states of registered inhabitants according to the federal Statistical Office from December 31 2021 was considered [27].Statistical analysis was performed using SPSS Statistics Version 25.0.0.0 (IBM Corp). A uni- and multivariable regression analysis was performed to assess significant associations in patient characteristics and incomplete and/or uncertain vaccination status as compared to complete immunization. Report of all the individual P values and confidence intervals was done, with no mathematical correction for multiple comparisons. The significance level was set at a P value < 0.05.
Results
Between October 28 2022 and November 23 2022, 5.630 of 33.207 (17%) invited volunteers completed the eCRF with most participants originating from Germany (n = 5.449, 96·8%), followed by Ireland (n = 87, 1·5%) and Austria (n = 49, 0·9%).
As the number of participants from countries other than Germany was comparatively low and not representative, respective data was not considered and included in the evaluation.
Within Germany, 3,193 (58.6%) women and 2,231 (40.9%) men provided information on their PM vaccinations. Median year of birth was 1975 (IQR 1964–1986). Detailed participant characteristics are given in Table 1, and geographical distribution in Fig. 1.Compared to the general population in Germany, the regional distribution of participants in our study was proportional to the number of inhabitants per federal state in the general population in Germany, except for overrepresentation of participants from North-Rhine-Westphalia and underrepresentation of participants from Lower Saxony. Nationwide, only the age group 40–60 years was overrepresented. Regarding sex, there was a higher proportion of women in our cohort compared to the general population in Germany (51% women vs. 49% men).
Details on demographic and regional representativeness of the cohort are given in the web-only supplement (Supplementary Tables 4 and 5).
Vaccination status
Although the majority of participants reported having received five (n = 1,009; 18.5%), followed by four (n = 805; 14.8%) PM vaccine doses, complete vaccination status was found in 1,981 (36.4%) individuals only.
In 1,382 (25.4%) participants, vaccination status was uncertain despite receipt of ≥ 4 doses, as vaccine types and valency were either insufficiently specified (n = 1,233; 22.6%) or the immunization schedule reported was incoherent (n = 149; 2.7%). A total 497 (9.1%) volunteers stated they had been vaccinated, but number and valency were unknown, and 313 (5.7%) volunteers did not know whether they had been vaccinated. This resulted in 2,192 (40.2%) of participants being classified with uncertain vaccination status.
Of 1,276 (23.4%) volunteers with incomplete immunization, 62 (4.9%) volunteers declared never having received a PM vaccine, 14.7% of volunteers had incomplete primary series, and 5,074 (93.1%) volunteers reported having been vaccinated at least once.
A scaled projection for Germany (n = 84,270,625) based on our data estimated that 19,719,326 (23.4%) German inhabitants may have incomplete vaccination.
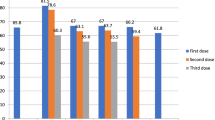
For details on age, sex, reported doses for primary series and vaccination status see Fig. 2.
Regression analysis
Certificate of vaccination
Volunteers with complete vaccination status significantly more often had access to their first vaccination certificate (78.5%), when compared to those with uncertain (58.6%) or incomplete vaccination history (28.6%; p < 0.001). Results were similar for documented complete primary series. Median year of birth and first entry in a vaccination card was close, i.e. 1975 and 1978, in those reporting complete vaccination status. That interval was significantly larger in those with incomplete vaccination status (1968 and 1992; p < 0.001).
Demographics
Year of oldest available vaccination card equaled year of birth in 582 (84.2%) participants between 18 and 29 years, and in 45 (14.5%) participants ≥ 70 years (Supplementary Table 2). Increasing age was associated with higher likelihood of uncertain/incomplete vaccination status and primary series in uni-, as well as multivariable analysis (Table 2). Correspondence of year of birth and first entry in a vaccination card was significantly more frequent in female when compared to male participants (63.0% vs. 52.8%; p < 0.001). Female participants were more likely to have a documented complete vaccination than male participants (39.3% vs. 32.2%, p < 0.001). Male sex was associated with incomplete and uncertain vaccination status, as well as primary series (Table 2). Highest proportion of complete vaccination was found in female participants between 18 and 29 years (48.5%), and 50 and 59 years (45.7%). The highest proportion of incomplete vaccination status was found in male participants > 70 years (47.0%) (Fig. 1C). The same applied for primary series, but with a wider range (64.9% vs. 36.6%) compared to vaccination status (Fig. 1D).
Underlying diseases
Within participants with incomplete vaccination status, 1,017 (80.0%) had underlying chronic disease (Table 1), of whom 38 (3.0%) reported an immunodeficiency including human immunodeficiency virus (HIV) (n = 19; 1.5%) or active cancer within recent two years (n = 19; 1.5%). Furthermore, hypertension and diabetes mellitus were significantly associated with incomplete or uncertain vaccination and primary series (Table 2).
Regional differences
No significant regional differences were observed for vaccination status or primary series (Supplementary Table 1). The proportion of volunteers with chronic kidney disease (1.8% vs. 0.5%; p < 0.001), as well as psychiatric disorders (7.9% vs. 5.4%; p < 0.02) were significantly higher in volunteers from eastern as compared to western German states. Also, maintenance of first vaccination certificate was more likely in volunteers from eastern states (64.3% vs. 58.2%; p < 0.06).
Timing of vaccination
A total of 2,087 (38.3%) participants indicated that they received a PM vaccine within the last 10 years. Most vaccines were obtained in 2020 (n = 218; 5.1%), closely followed by 2019 (n = 216; 5.0%). No increase was observed for 2022 as compared to prior four years (Fig. 2). Longer interval since last vaccination (> 1 year) was associated with incomplete primary series and vaccination status (Table 2). While between 1955 and 1991, vaccination rates were overall higher in men compared to women, vaccination rates were consistently higher in women from 1991 (Fig. 3). In 1958 and 1966 only men reported on receipt of PM vaccination.
Sub-analysis of cohort with uncertain vaccination status
Sub-analysis of vaccines reported by volunteers with uncertain vaccination status revealed that out of 2,192 participants, 423 (19.3%) were probably complete (Supplementary Fig. 2). Added to those classified as complete (n = 1,981), a total of 2,404 (44.1%) volunteers would have complete vaccination status. Underlying causes for uncertain vaccination status were identified as unknown vaccine types and/or valences (56.3%), unknown number of doses (22.7%), unknown PM vaccination (14.3%) and incoherent vaccination scheme (6.8%). Most frequently applied criterion was number of doses (n = 365; 85.1%), followed by coherence of vaccination schedule (n = 58; 13.5%) and year of application (n = 6; 1.4%).
Discussion
Using poliomyelitis as a precedent, this investigation shows that current immunization recording in Germany is not fit for the next pandemic. While 23% of volunteers had an incomplete vaccination status according to the recommended schedule besides fully available individual vaccination records, more than 40% of participants were classified with uncertain vaccination status, mostly due to the lack of specific details on previously administered doses.
For vaccines less established than those against PM, or less often part of a combination vaccine, rates must be assumed worse [9, 24]. The rates found in our study appear even direr when seen against a background of successful global mass PM vaccination campaigning, and considering that adult volunteers in the VACCELERATE registry may likely more interested in the topic [9, 21].
Pandemic preparedness comprises knowledge of vaccination gaps at a population-level [2]. The (re-)emergence of highly contagious and vaccine preventable pathogens demands for broad, rapid and specific knowledge of previously administered vaccine doses to target those with incomplete vaccination schedules [2, 3]. Sufficient vaccination coverage is required to induce protective population immunity and prevent outbreaks [28]. In the absence of a centralized vaccination registry data on individual previous immunization can only be derived from personal vaccination certificates, medical records or self-reports [5, 18, 20].
With an increased level of migration on the one hand, and growing vaccine hesitancy on the other hand, recent isolation of PV appears even more alarming behind the background that the greatest risk for re-establishment arises from the juxtaposition of un-/under immunized- and large populations vaccinated with IPV-only schemes [14, 18, 20, 29]. Since 1998 the Standing Committee on Vaccination (STIKO) of the Federal Republic of Germany (FRG) recommends the sole use of IPV to prevent cVDPV [24]. According to the risk assessment by the European Centre for Disease Prevention and Control (ECDC), there is a moderate risk of infection and low risk for disease in IPV-only vaccinees, while OPV vaccinees have a very low risk for both, infection and disease [29]. Low- and unvaccinated groups on the contrary have a high probability of infection and moderate risk for disease [29]. The calculated herd immunity threshold of 80 to 86% would not be reached according to our study data [28].
The last evaluation of the PM vaccination status in German adults was accomplished over ten years ago as part of the first wave of German Health Interview and Examination Survey for Adults (DEGS1) [16]. Data collected from vaccination cards and self-reports thereby revealed a lifetime prevalence for at least one PM vaccination of 85.6% [16]. In the present study, we found a slightly higher lifetime vaccination prevalence with 93.1% among registered volunteers. Recent public warnings after PV detection in non-endemic regions may have motivated people to check their PM immunization status [30]. However, our results showed no significant increase in vaccination rates in 2022 compared to the four preceding years, while most doses were recorded in 2020 at the beginning of the COVID-19 pandemic.
A 2002 PM seroprevalence study on PM in Germany including sera from 2.564 adults indicated high levels of population immunity showing neutralizing antibodies against PV type 1 in 96.8% of subjects, as well as 96.8% against type 2 and 89.6% type 3 respectively [31]. This study also evaluated regional differences in immunization status. Higher rates of sufficient PM immunization were assessed in sera from individuals from Western German states [31]. In our study, no significant differences in reported PM vaccination status were found in volunteers from Western versus Eastern states. These geographic analyses warrant caution as volunteers may have moved between regions.
Similar to our findings, there was an association between demographics and vaccination status in DEGS1, showing higher vaccination rates in women compared to men and in younger compared to older participants [16]. Highest sex related difference in vaccination rates was found in both, DEGS1 and our study, in the 50–59 years age group. Higher vaccination rates in women were also reported for other vaccine preventable diseases and geographic locations for example influenza in the US and COVID-19 in Canada [32, 33]. As these are not childhood vaccinations like PM, sex-specific differences in health care adherence and decision-making that might have played a role there are less plausible causes regarding PM vaccination rates [32, 34].
In DEGS1, women were able to present their vaccination certificate more often [16]. Highest rates of complete vaccination records were found in women between 18 and 29 years and lowest rates in men between 60 and 69 years [16]. Our study showed similar findings. Year of birth and oldest available certificate entry was higher in female and younger participants. Not surprisingly, we found that the closer the oldest available vaccination certificate was to birth year, the more likely immunization records were complete. We assume incomplete vaccination records and increasing recall bias over time are main causes for reporting incomplete vaccination status. Each vaccine obtained in Germany is documented in a personal paper-based vaccine certificate [16]. Seasonal vaccines, e.g. influenza, are not necessarily documented here. If space for documentation is used up, individuals may obtain a new certificate. As there is no integral harmonized vaccination recording in Germany, data on previous vaccines can be lacking if a particular certificate is lost [5].Our study has several limitations. It is questionable whether our sample is representative as participants of the VACCELERATE Volunteer Registry (VR) may be more interested in vaccination, suggesting that the dark figure of incomplete vaccination might be even higher. While the VR was promoted in refugee reception- and health department centres, the immigrant status was not assessed in the present investigation [21]. Nevertheless, a projection for Germany based upon our data would still result in a considerable number of at least 19.719.326 (23.4%) individuals with incomplete vaccination status potentially at risk when exposed to PV [27]. Hypothetically, seropositivity might be higher taking the high number of volunteers with any vaccination against PM into account. It is important to note that this study is limited by the fact that serological analyses were not included. The study was not meant to assess seroprevalence of protection against PV but collect available data on prior administered vaccine doses to identify potential vaccination gaps at a population-level and inform participants in case of incomplete vaccination schedule.
Further limitations consist of recall and/or reporting bias as vaccines were often administered many years ago, and data transfer from a paper-based vaccination certificate into an eCRF can be erroneous. Future PM vaccination status evaluations may use technical assistance for automated reading of vaccination certificates, such as the Vacuna® app employing image recognition [35].
Ideally, vaccinations were recorded electronically in a centralized registry to allow for permanent and online-availability, and for international harmonization as individually introduced for COVID-19 vaccination during the pandemic [4]. In daily routine, missing vaccination records cause uncertainty among treating physicians as well as time-consuming investigations by medical staff [25]. The involvement of electronic tools would allow verification of self-reported data [35]. Social structures between individual participants and/or common households were not assessed within this investigation to assess the potential impact on reported data with special regard to gender-specific differences.
In conclusion, PM vaccination status was difficult to assess due to absent or incomplete records. The current paper-based vaccination recording system in Germany fails to inform the individual citizen and public health authorities. Further investigations including serological analyses adding to the data collected in this study are needed to elucidate the population PM immunization status, especially considering vulnerable patient groups with uncertain or incomplete vaccination history.
Data availability
Data available on request from the authors. The data that support the findings of this study are available from the corresponding author, OAC, upon reasonable request. Basic, Share upon Request.
References
Bayham J, Fenichel EP. Impact of school closures for COVID-19 on the US health-care workforce and net mortality: a modelling study. Lancet Public Health. 2020;5:e271–8. https://doi.org/10.1016/S2468-2667(20)30082-7.
Bourrier MS, Deml MJ. The legacy of the pandemic preparedness regime: an integrative review. Int J Public Health. 2022;67:1604961. https://doi.org/10.3389/ijph.2022.1604961.
Chen YT. Effect of vaccination patterns and vaccination rates on the spread and mortality of the COVID-19 pandemic. Health Policy Technol. 2023;12:100699. https://doi.org/10.1016/j.hlpt.2022.100699.
Mbunge E, Dzinamarira T, Fashoto SG, Batani J. Emerging technologies and COVID-19 digital vaccination certificates and passports. Public Health Pract (Oxf). 2021;2:100136. https://doi.org/10.1016/j.puhip.2021.100136.
Wiot F, Shirley J, Prugnola A, Di Pasquale A, Philip R. Challenges facing vaccinators in the 21(st) century: results from a focus group qualitative study. Hum Vaccin Immunother. 2019;15:2806–15. https://doi.org/10.1080/21645515.2019.1621147.
Rieck T, Feig M, Siedler A. Impfquoten von Kinderschutzimpfungen in Deutschland –aktuelle Ergebnisse aus der RKI-Impfsurveillance. Epidemiologisches Bulletin. 2022;48:3–25. https://doi.org/10.25646/10838.
Viktorova EG, Khattar S, Samal S, Belov GA. Poliovirus Replicon RNA generation, transfection, packaging, and quantitation of replication. Curr Protoc Microbiol. 2018. https://doi.org/10.1002/cpmc.47.
Pait CF. Virology of poliomyelitis. Calif Med. 1950;73:391–3.
Bigouette JP, Wilkinson AL, Tallis G, Burns CC, Wassilak SGF, Vertefeuille JF. Progress toward polio eradication - worldwide, January 2019-June 2021. MMWR Morb Mortal Wkly Rep. 2021;70:1129–35. https://doi.org/10.15585/mmwr.mm7034a1.
World Health A. Global eradication of poliomyelitis by the year 2000. Geneva: World Health Organization; 1988.
Kew OM, Cochi SL, Jafari HS, Wassilak SG, Mast EE, Diop OM, et al. Possible eradication of wild poliovirus type 3–worldwide, 2012. MMWR Morb Mortal Wkly Rep. 2014;63:1031–3.
Alleman MM, Jorba J, Henderson E, Diop OM, Shaukat S, Traoré MA, et al. Update on vaccine-derived poliovirus outbreaks - worldwide, January 2020-June 2021. MMWR Morb Mortal Wkly Rep. 2021;70:1691–9. https://doi.org/10.15585/mmwr.mm7049a1.
Khetsuriani N, Perehinets I, Nitzan D, Popovic D, Moran T, Allahverdiyeva V, et al. Responding to a cVDPV1 outbreak in Ukraine: Implications, challenges and opportunities. Vaccine. 2017;35:4769–76. https://doi.org/10.1016/j.vaccine.2017.04.036.
Leggiadro RJ. Public health response to a case of paralytic poliomyelitis in an unvaccinated person and detection of poliovirus in wastewater—New York, June-August 2022. Pediatr Infect Dis J. 2022;41:898. https://doi.org/10.1097/inf.0000000000003696.
Klapsa D, Wilton T, Zealand A, Bujaki E, Saxentoff E, Troman C, et al. Sustained detection of type 2 poliovirus in London sewage between February and July, 2022, by enhanced environmental surveillance. Lancet. 2022. https://doi.org/10.1016/s0140-6736(22)01804-9.
Poethko-Müller C, Schmitz R. Vaccination coverage in German adults: results of the German health interview and examination survey for adults (DEGS1). Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz. 2013;56:845–57. https://doi.org/10.1007/s00103-013-1693-6.
Robert-Koch-Institut. 2022 Impfquoten von Kinderschutzimpfungen in Deutschland. Epidemiologisches Bulletin 48/2022.
Feinstein S, Poleacovschi C, Drake R, Winters LA. States and refugee integration: a comparative analysis of France, Germany, and Switzerland. J Int Migr Integr. 2022;23:2167–94. https://doi.org/10.1007/s12134-021-00929-8.
Cojocaru E, Cojocaru C, Cojocaru E, Oancea CI. Health risks during ukrainian humanitarian crisis. Risk Manag Healthc Policy. 2022;15:1775–81. https://doi.org/10.2147/rmhp.S375021.
Stoeckel F, Carter C, Lyons BA, Reifler J. The politics of vaccine hesitancy in Europe. Eur J Public Health. 2022;32:636–42. https://doi.org/10.1093/eurpub/ckac041.
Salmanton-García J, Stewart FA, Heringer S, Koniordou M, Álvarez-Barco E, Argyropoulos CD, et al. VACCELERATE volunteer registry: a European study participant database to facilitate clinical trial enrolment. Vaccine. 2022;40:4090–7. https://doi.org/10.1016/j.vaccine.2022.05.022. Accessed 5 May 2023
Robert-Koch-Institut. Tasks and Aims. https://www.rki.de/EN/Content/Institute/institute_node.html.
Robert-Koch-Institut. KV-Impfsurveillance: Auswertung von Abrechnungsdaten der Kassenärztlichen Vereinigungen. (2023). https://www.rki.de/DE/Content/Infekt/Impfen/Impfstatus/kv-impfsurveillance/kvis_node.html#doc2391060bodyText2. Accessed 05 May 2023.
Robert-Koch-Institut. Schutzimpfung gegen Poliomyelitis: Häufig gestellte Fragen und Antworten. (2022). https://www.rki.de/SharedDocs/FAQ/Impfen/Poliomyelitis/FAQ-Liste_Poliomyelitis_Impfen.html. Accessed 02 May 2023.
Reiter S. Ausgewählte Daten zum Impf- und Immunstatus in Deutschland. Bundesgesundheitsblatt - Gesundheitsforschung -Gesundheitsschutz. 2004;47:1144–50. https://doi.org/10.1007/s00103-004-0952-y.
Robert-Koch-Institut. Archiv der STIKO-Empfehlungen. (2023). https://www.rki.de/DE/Content/Kommissionen/STIKO/Empfehlungen/Archiv/seit_1972/archiv_tab.html. Accessed 05 May 2023.
Bundesamt S. Bevölkerung - Anzahl der Einwohner in den Bundesländern in Deutschland am 31. Dezember 2021. (2022). https://de.statista.com/statistik/daten/studie/71085/umfrage/verteilung-der-einwohnerzahl-nach-bundeslaendern/. Accessed 05 May 2023.
Plans-Rubió P. Evaluation of the establishment of herd immunity in the population by means of serological surveys and vaccination coverage. Hum Vaccin Immunother. 2012;8:184–8. https://doi.org/10.4161/hv.18444.
European Centre for Disease Prevention and Control. Wild-type poliovirus 1 transmission in Israel – what is the risk to the EU/EEA? Stockholm: ECDC. 2013. polio-risk-assessment-transmission-in-Israel.pdf (europa.eu). Accessed 05 May 2024.
Mercader-Barceló J, Otu A, Townley TA, Adepoju P, Walley J, Okoibhole LO, et al. Rare recurrences of poliomyelitis in non-endemic countries after eradication: a call for global action. Lancet Microbe. 2022. https://doi.org/10.1016/S2666-5247(22)00253-1.
Diedrich S, Claus H, Schreier E. Immunity status against poliomyelitis in Germany: determination of cut-off values in International Units. BMC Infect Dis. 2002;2:2. https://doi.org/10.1186/1471-2334-2-2.
Applewhite A, Stancampiano FF, Harris DM, Manaois A, Dimuna J, Glenn J, et al. A retrospective analysis of gender-based difference in adherence to influenza vaccination during the 2018–2019 season. J Prim Care Community Health. 2020;11:2150132720958532. https://doi.org/10.1177/2150132720958532.
Ray JG, Park AL. SARS-CoV-2 vaccination, ABO blood group and risk of COVID-19: population-based cohort study. BMJ Open. 2022;12:e059944. https://doi.org/10.1136/bmjopen-2021-059944.
Matoff-Stepp S, Applebaum B, Pooler J, Kavanagh E. Women as health care decision-makers: implications for health care coverage in the United States. J Health Care Poor Underserved. 2014;25:1507–13. https://doi.org/10.1353/hpu.2014.0154.
Benedikt M, Tobias H. vacuna. (2019–2023). https://vacuna.io/. Accessed 04 May 2023.
Funding
Open Access funding enabled and organized by Projekt DEAL. VACCELERATE has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement 101037867.
Author information
Authors and Affiliations
Consortia
Contributions
JN, JS, HJS and OAC conceived the study idea. JN, JSG and LMC collected data. JN, JSG, HJS and JS interpreted data. JSG performed statistical analysis. JN wrote the first draft of the manuscript. All authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
HJS has received consultancy or speaker honoraria from Pfizer, CSL Seqirus, Sanofi, Cologne University, Global Health Press, Minervax, VacciZONE and GOPA/KfW. JAN has received speaker honoraria by Hikma. JSG has received speaker honoraria from Gilead and Pfizer. JS has received research grants by the Ministry of Education and Research (BMBF) and Basilea Pharmaceuticals Inc.; has received speaker honoraria by Pfizer Inc., Gilead, Hikma and AbbVie; has been a consultant to Gilead, Alvea Vax. and Micron Research. PWGM has received honoraria and/or travel grants from Astrazeneza, Gilead Sciences, ViiV, Janssen and Pfizer. OAC reports grants or contracts from Amplyx, Basilea, BMBF, Cidara, DZIF, EU-DG RTD (101037867), F2G, Gilead, Matinas, MedPace, MSD, Mundipharma, Octapharma, Pfizer, Scynexis; Consulting fees from Abbvie, Amplyx, Biocon, Biosys, Cidara, Da Volterra, Gilead, IQVIA, Janssen, Matinas, MedPace, Menarini, Molecular Partners, MSG-ERC, Noxxon, Octapharma, Pardes, Pfizer, PSI, Scynexis, Seres; Honoraria for lectures from Abbott, Abbvie, Al-Jazeera Pharmaceuticals, Astellas, Gilead, Grupo Biotoscana/United Medical/Knight, Hikma, MedScape, MedUpdate, Merck/MSD, Mylan, Noscendo, Pfizer, Shionogi; Payment for expert testimony from Cidara; Participation on a Data Safety Monitoring Board or Advisory Board from Actelion, Allecra, Cidara, Entasis, IQVIA, Janssen, MedPace, Paratek, PSI, Pulmocide, Shionogi, The Prime Meridian Group; A patent at the German Patent and Trade Mark Office (DE 10 2021 113 007.7); Stocks from CoRe Consulting, and EasyRadiology. LMC, MZ and ZDP have nothing to declare.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nacov, J.A., Stemler, J., Salmanton-García, J. et al. Challenges in assessing the immunization status of adults in Germany—lessons from a population-based VACCELERATE survey on polio vaccination. Infection 52, 1563–1574 (2024). https://doi.org/10.1007/s15010-024-02296-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s15010-024-02296-9