Abstract
Purpose
Blood cultures (BCs) are key for pathogen detection in septic patients. We investigated the extent to which sampling was performed and what factors were associated with the absence of general or inadequate BC sampling.
Methods
We conducted a retrospective cohort study of hospitalized patients with sepsis admitted to one of three EDs in 2018. Primary outcome was the extent of general BC collection of at least 1 set. Secondary outcome was the extent of adequate BC sampling, defined as ≥ 2 sets before antibiotic therapy (AT). Multivariable logistic regression analysis was performed to identify factors associated with deficits in both outcomes.
Results
1143 patients were analyzed. BCs were collected from 946 patients. Single BCs were taken from 520 patients, ≥ 2 sets from 426 patients. Overall, ≥ 2 BCs were taken from 349 patients before AT. BC sampling before AT occurred significantly more frequently when ≥ 2 BC sets were taken rather than a single one (81.9%, versus 68.4%, p < 0.001) and this also led to the highest pathogen detection rate in our cohort (65.6%). A body temperature of ≥ 38 °C was the a supporting factor for general and adequate BC collection in all three EDs. Retrospective analysis of 533 patients showed that the qSOFA score had no influence on general or adequate BC collection.
Conclusion
Data on everyday clinical practice in the pre-analytical phase of microbiological diagnostics shows considerable deficits and indicates the need for more implementation of best practice. The variations identified in BC sampling between EDs should be further investigated.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There is a significant association of sepsis with morbidity and mortality [1]. Numerous deaths resulting from sepsis could be prevented by targeted and adequate antibiotic therapy (AT) [2]. Identifying the causative pathogen is key to ensuring adequate AT. Blood cultures (BCs) continue to be the gold standard for detecting causative pathogens in patients with sepsis [3]. In 2017, the Surviving Sepsis Campaign (SSC) published guidelines to assist clinicians in identifying and treating septic patients. One of the cornerstones of these guidelines is appropriate diagnostics, in particular BC diagnostics [4]. Once sepsis is suspected, a minimum of two sets of BCs should be obtained immediately. Although recently updated international guidelines do not refer explicitly to the necessity of BCs [5], data shows that it is nonetheless essential that at least two sets of BCs be taken from patients before the administration of AT [6, 7]. This recommendation continues to be part of current German guidelines [8]. Indeed, Collazos-Blanco et al. suggest that three sets should be obtained per patient to ensure a greater probability of identifying the causative pathogen [9].
Numerous studies have investigated the implementation of sepsis guidelines and recommendations [10,11,12,13]. However, although these studies have examined whether BCs were obtained, they did not take into consideration the timing in relation to AT administration or the number of sets taken per patient.
Because emergency departments (EDs) are most frequently involved in the early diagnosis and treatment of patients with community-onset sepsis [10], we describe the results of a retrospective analysis of BC sampling in patients with suspected sepsis in three German EDs with a focus on (1) BC sampling in general and (2) an adequate BC collection of two BC sets before AT administration. Furthermore, we analyze factors associated with gaps in general and adequate BC collection.
Methods
Study design and setting
In this retrospective cohort study, we analyzed the BC sampling in EDs of hospitalized patients with a hospital discharge diagnosis of “sepsis” based on ICD-10 (International Statistical Classification of Diseases and Related Health Conditions) System A40-A41 [14]. Data was obtained in three EDs (A, B and C) of acute care hospitals: Hospital A is a primary care hospital with 443 beds and 15,809 ED patient visits in 2018; hospital B is a secondary care hospital with 692 beds and 34,368 ED patient visits; hospital C is a tertiary care hospital with 830 beds and 44,782 ED patient visits.
Study population
Included were all inpatients ≥ 18 years of age admitted to hospitals A–C via the ED and who were discharged between 01.01.2018 and 31.12.2018 with a diagnosis of “sepsis” [14]. Only patients who had received emergency care in EDs A–C with standardized documentation were included.
Exclusion criteria
Patients were excluded from the analysis (1) if patients’ admission had been planned prior to their visit to the ED; (2) if they had been transferred from another hospital; (3) if they were admitted directly to the intensive care unit; or (4) if they were diagnosed with hospital-acquired sepsis. Hospital-acquired sepsis was diagnosed by identifying signs and symptoms of sepsis that first appeared on day 3 or later.
Outcome
The primary outcome was the extent to which a BC consisting of at least one set was generally taken during the initial treatment of a patient in the ED. The secondary outcome was the extent of adequate BC collection. Adequate BC sampling was defined as a BC consisting of at least two sets per patient prior to the administration of AT, each set containing one anaerobe and one aerobe sample. Gaps in primary and secondary outcomes were further analyzed in relation to their association with patient-based and organizational factors.
In addition, pathogen detection rates were analyzed for patients receiving 1 or more BC sets before or after AT administration.
Data source and collection
Eligible patients were reviewed and analyzed for inclusion and exclusion criteria. The following data was obtained from the patient charts: patient characteristics and routine clinical data such as information on vital signs, possible immunosuppression, relevant comorbidities and medication, the presumed (source of) infection, and the chronological course of the taking of BCs and the administration of AT. Furthermore, data from the microbiological laboratory was evaluated in order to obtain as much information on BC sampling and the exact number of sets taken per patient. Data was entered into the study database using the online survey tool Lime survey, version 2.0.
Data analysis
In the descriptive analysis we specified number and percent for categorical parameters and median and interquartile range (IQR) for continuous parameters. Differences were tested using the chi-square test or the Wilcoxon rank-sum test. A multivariable logistic regression analysis was performed to examine factors associated with deficits in primary and secondary outcomes. All patient characteristics as well as clinical and laboratory parameters documented in the ED were taken into consideration in the analysis. Parameters not documented were categorized as “no information” and considered as a separate category. The multivariable model was calculated for (a) all three EDs and (b) individual EDs with stepwise forward variable selection where the significance level was set at < = 0.05 to include a parameter in the model. As part of a sensitivity analysis for data of all three EDs, we also considered the parameter based on the physicians’ estimate of the suspected focus of the infection in the model. All analyses were exploratory in nature and were performed with SAS (version 9.4) and SPSS (version 25).
Results
Overall, data from 1143 patients was included in the analysis. As shown in Table 1, BCs were taken from 946 patients (83%), most often one set (n = 520; 46%). Two or more sets were taken from 426 patients (37%). The overall rate of BCs taken did not differ among EDs A, B, and C (82, 82. and 83%, respectively), while the number of sets per patient varied significantly between the EDs with ≥ 2 BC sets for 30% of patients in ED C to 78% in ED A (p < 0.001). Significant differences were also seen in the AT administration documented. With 801 patients (70%) documented overall, the highest rate of documentation of AT administration occurred in ED C (75%), followed by ED A (71%) and ED B (62%). Overall, 349 patients (31%) had a documented adequate sampling of ≥ 2 BC sets prior to the administration of AT. Patients received antibiotics significantly more frequently after BC collection when two or more sets were taken rather than a single BC (81.9%, versus 68.4%, p < 0.001).
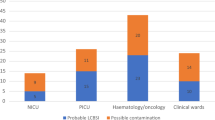
(6) The most frequent pathogens identified are shown in Fig. 1.
The median age of the patients was 75 (interquartile range (IQR) 65–81); they were predominantly male (59%). Forty-seven percent of patients were immunocompromised, whereby age and percentage of immunocompromised patients differed significantly between the 3 Eds (Supplement Table 1). All patients’ characteristics are stratified in accordance to general BC sampling and adequate BC collection in Table 2.
As summarized in Table 3, 46% of patients had a body temperature above 38 °C and 18% had systolic blood pressure < 100 mm Hg. A respiratory rate of > 20 breaths per minute was documented in 28% of patients.
The analysis of 553 patients for whom all criteria of the qSOFA score were available showed that patients with a retrospectively determined qSOFA score > = 2 received general or no BC collection to the same extent (49.8% versus 51.9%). This distribution was also evident in patients with a retrospectively determined qSOFA score of < 2 who did or did not receive adequate BC sampling (49.7% versus 50.3%).
Multivariable logistic regression and sensitivity analysis
Multivariable logistic regression analysis for data from all three EDs showed a decreased likelihood of general BC collection in patients with a systolic blood pressure ≥ 140 mmHg and for whom data on mental alteration or systolic blood pressure was missing. Medically induced immunosuppression or a body temperature ≥ 38 °C were supporting factors for receiving BCs (Table 4).
The likelihood of adequate BC collection with ≥ 2 BC sets before AT was lower for female patients, for patients with missing data on mental alteration or blood pressure, and in certain EDs.
Patients with a temperature ≥ 38 °C, systolic blood pressure ≤ 100 mm Hg, or documented immunosuppression had a higher chance that adequate BC collection would take place (Table 5).
Sensitivity analysis
For both outcomes, factors identified in the multivariate analysis were confirmed in the sensitivity analysis, which took into account the respectively presumed focus of the infection.
Multivariable logistic regression for individual EDs
Additional multivariable logistic regression analyses for each individual ED identified a body temperature ≥ 38 °C as supporting factor for receiving general and adequate BC sampling in all 3 EDs. Factors, such as medically induced immunosuppression or missing data on systolic blood pressure, were only significant in individual EDs (Table 6).
Discussion
In our retrospective study, we analyzed data from 1143 patients prior to their inpatient admission for suspected sepsis. This is a rather small proportion of a total 95,000 patient visits to the 3 participating EDs per year. However, the importance of adequate BC diagnostics in early treatment of sepsis warrants a precise consideration of BC sampling as an important part of AMS in healthcare [13].
Interestingly, at least one BC was taken from an equally high proportion of patients during initial care. This shows that the risk of sepsis was perceived as often in all three EDs. Only single BCs were obtained from the majority of patients, which indicates that a suspicion of sepsis was followed up with a BC during these patients’ initial care, although sampling was not in accordance with best practice. As described by Fabre et al. in a survey in the US, HCWs are often of the opinion that a single set of BCs is adequate for detecting bacteremia [15].
No BC at all was taken from almost 20% of patients. Whether this reflects an effort to avoid excessive BC diagnostics as described in the literature, remains unclear [16]. One common argument for a more limited use of BC diagnostics is to avoid unnecessary treatment if contamination of a culture has occurred. In emergency care, however, it can be assumed that the clinical picture takes precedence and that initial AT given is independent of the results of BC diagnostics, which are only available later. Therefore, we cannot fully support the approach of restricting the use of BC diagnostics, particularly in patients who show signs of infection and who are likely to require hospital admission. Of course, contamination of samples is a problem, and all healthcare departments—EDs included—should check the contamination rates of microbiological samples regularly and implement appropriate interventions when necessary. Rather than restricting the number of BCs taken, we believe the optimal means of preventing contamination is the strict aseptic sampling of at least two BC sets, each obtained from different peripheral sites [3]. The recommended number of at least two BC sets per patient was obtained only for a good third of all patients. This indicates that a sampling of two sets is possible during patient care in an ED. In addition, the high proportion of patients from whom at least two sets were taken prior to AT administration suggests that following this recommended time sequence is also feasible. Interestingly, the chronological order of BC collection and AT administration was significantly more in line with recommendations when ≥ 2 sets were taken rather than only a single BC. In addition, the recommended chronological order was associated with a higher pathogen detection rate, which was highest in patients receiving ≥ 2 sets before AT administration in our cohort.
More education and training on identification of patients for BC diagnostic and adequate BC sampling are needed [17] and obstacles and facilitators should be addressed during implementation. Our analyses of data from all three EDs and data from individual EDs show that BCs in general were more likely to be obtained generally and adequately if patients had an elevated body temperature. This result is in line with the findings of the survey by Fabre et al. in which HCWs acknowledged that febrile patients were more likely to yield positive BCs [15]. In addition, a German nationwide survey found that fever above 38.5 °C is a very strong clinical criterion for BC sampling [18]. But increased body temperature is not a specific sign of sepsis, especially in the case of elderly patients, who represent the largest segment of patients in our study [19]. Therefore, close attention to elevated temperature should be balanced by an awareness that a temperature below the cut-off level does not rule out infection. According to our data an immunocompromised state seems also to be a strong stimulus for initiating microbiological blood analysis in the ED. Although the clinical presentation of sepsis might differ depending on immune status [20], healthcare workers’ (HCW) awareness of adequate microbiological diagnostics may be increased by the fact that immunosuppressive medications increase the risk of sepsis [21, 22].
With the exception of elevated body temperature obstacles and facilitators identified in the analysis of all three EDs were not confirmed for individual EDs. This may be due to individual analyses’ smaller numbers and to potential differences between the three cohorts, which were selected by hospitals’ coding of sepsis [23]. Another factor could be the variations in the pre-analytical phase of BC diagnostics among the EDs.
In one ED a gender bias was associated with the outcome of no adequate BC sampling since adequate BC collection was less likely to be performed for female patients than male patients. This result is somewhat consistent with an analysis by Henning et al. which showed that the volume of blood collected for BCs was significantly greater for male patients than for female patients [24]. Furthermore, gender differences in sepsis management have been described. For example, Shallcross et al. found that male patients were more likely to receive BCs and antibiotics than female patients [13]. As has been consistently reported, men have a higher frequency of sepsis than women [25,26,27]. This distribution by gender may influence the decision of HCWs to administer adequate BC sampling more frequently to men.
Sensitivity analysis showed that specific infection sites had no influence on adequate BC sampling. We found that an unknown focus of infection increased the likelihood of adequate BC diagnostics. This could be due to the intense sufficient microbiological diagnostics needed to identify a pathogen. Furthermore, our analysis showed that a lack of documentation, especially of blood pressure, mental status, and infection foci, was an obstacle to general and adequate BC sampling. One explanation could be that these patients were in poor clinical condition and therefore required intensive treatment, which did not allow the documentation of clinical findings and BC sampling. However, it is also possible that organizational factors played a role. In critical care, as pointed out by Soto et al., disparities are most likely multifactorial, involving individual, community, and hospital-level factors [28]. Raupach-Rosin et al. found that the department in which a HCW worked was a factor associated with good BC practice, particularly for BC sampling [18]. Because we focused predominantly on patient factors that were associated with BC practice, we did not collect structural data from EDs that may influence BC practice. As shown by Pin et al., diagnostic stewardship training is not offered in every ED and should therefore be intensified [29].
Because we did not collect information on the treatment teams in the EDs, we were not able to describe the potential influence of gender-specific variations in practice on the part of physicians—there may, for example, have been greater adherence to guidelines among female physicians—that could also have resulted in differences in adequate blood culturing [30].
As mentioned above, a major limitation of our study is that our retrospective cohort was based on hospitals’ coding data of sepsis and using the ICD-10 code algorithm may underestimate the true incidence of sepsis [31]. Furthermore, as described by Schwarzkopf et al., accuracy of diagnosing and coding of sepsis varies between hospitals, which may have affected our analysis and biased the results [23]. Finally, all data was obtained retrospectively from patient charts and laboratory analysis of microbiological BCs. This method showed that relevant clinical information was not always documented. Therefore, we cannot precisely analyze clinical signs or the timing of AT administration in these cases. It could not be determined under which conditions BCs were initiated and collected, by whom, or the volume of blood cultured. Furthermore, data from only three EDs was analyzed and hence the generalizability of results is limited.
Conclusion
Nevertheless, our analysis provides insight into current practices related to the pre-analytical phase of BC diagnostics for ED patients suspected of sepsis. More teaching and implementation of best practice in diagnostic stewardship should be undertaken to close the gaps in initiating and undertaking adequate BC sampling. The variations in BC sampling identified among EDs should be further investigated.
Availability of data and material
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- AB:
-
Antibiotics
- AMS:
-
Antimicrobial stewardship
- AT:
-
Antibiotic therapy
- BC:
-
Blood culture
- CI:
-
Confidence interval
- ED:
-
Emergency department
- GCS:
-
Glasgow Coma Scale
- HCW:
-
Healthcare worker
- ICD:
-
International Statistical Classification of Diseases and Related Health Conditions
- IQR:
-
Interquartile range
- OR:
-
Odds ratio
- SOFA:
-
Sequential Organ Failure Assessment
- SSC:
-
Surviving Sepsis Campaign
References
Rudd KE, Johnson SC, Agesa KM, Shackelford KA, Tsoi D, Kievlan DR, et al. Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the global burden of disease study. Lancet. 2020;395:200–11.
Retamar P, Portillo MM, López-Prieto MD, Rodríguez-López F, de Cueto M, García MV, et al. Impact of inadequate empirical therapy on the mortality of patients with bloodstream infections: a propensity score-based analysis. Antimicrob Agents Chemother. 2012;56:472–8.
Lamy B, Dargère S, Arendrup MC, Parienti J-J, Tattevin P. How to optimize the use of blood cultures for the diagnosis of bloodstream infections? A state-of-the art. Front Microbiol. 2016;7:697.
Rhodes A, Evans LE, Alhazzani W, Levy MM, Antonelli M, Ferrer R, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock: 2016. Crit Care Med. 2017;45:486–552.
Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Crit Care Med. 2021;49:e1063–143.
Scheer CS, Fuchs C, Gründling M, Vollmer M, Bast J, Bohnert JA, et al. Impact of antibiotic administration on blood culture positivity at the beginning of sepsis: a prospective clinical cohort study. Clin Microbiol Infect. 2019;25:326–31.
Cheng MP, Stenstrom R, Paquette K, Stabler SN, Akhter M, Davidson AC, et al. Blood culture results before and after antimicrobial administration in patients with severe manifestations of sepsis: a diagnostic study. Ann Intern Med. 2019;171:547–54.
Brunkhorst FM, Weigand M, Pletz M, Gastmeier P, Lemmen SW, Meier-Hellmann A et al. S3-leitlinie sepsis prävention, diagnose, therapie und nachsorge. AWMF-registernummer: 079-001 langversion 3.1-2018 deutsche sepsis gesellschaft e. V. 2018
Collazos-Blanco A, Pérez-García F, Sánchez-Carrillo C, de Egea V, Muñoz P, Bouza E. Estimation of missed bloodstream infections without the third blood culture set: a retrospective observational single—centre study. Clin Microbiol Infect. 2019;25(4):469–73.
Jeon K, Na SJ, Oh DK, Park S, Choi EY, Kim SC, et al. Characteristics, management and clinical outcomes of patients with sepsis: a multicenter cohort study in Korea. Acute Crit Care. 2019;34:179–91.
Levy MM, Artigas A, Phillips GS, Rhodes A, Beale R, Osborn T, et al. Outcomes of the surviving sepsis campaign in intensive care units in the USA and Europe: a prospective cohort study. Lancet Infect Dis. 2012;12:919–24.
Seymour CW, Gesten F, Prescott HC, Friedrich ME, Iwashyna TJ, Phillips GS, et al. Time to treatment and mortality during mandated emergency care for sepsis. N Engl J Med. 2017;376:2235–44.
Shallcross LJ, Freemantle N, Nisar S, Ray D. A cross–sectional study of blood cultures and antibiotic use in patients admitted from the emergency department: missed opportunities for antimicrobial stewardship. BMC Infect Dis. 2016;16:166.
Deutsches institut für medizinische dokumentation und information (DIMDI) internationale statistische klassifikation der krankheiten und verwandter gesundheitsprobleme 10. revision german modification version 2018. https://www.dimdi.de/static/de/klassifikationen/icd/icd-10-gm/kode-suche/htmlgm2018/. Accessed 10 March 2023
Fabre V, Milstone AM, Keller SC, Carroll KC, Cosgrove SE. prescribers‘ knowledge, attitudes and perceptions about blood culturing practices for adult hospitalized patients: a call for action. Infect Control Hosp Epidiomol. 2018;39:1394–6.
Pawlowicz A, Holland C, Zou B, Payton T, Tyndall JA, Allen B. Implementation of an evidence-based algorithm reduces blood culture overuse in an adult emergency department. Gen Int Med Clin Innov. 2016;1:26–9.
Rothe K, Spinner CD, Ott A, Querbach C, Dommasch M, Aldrich C, et al. Strategies for increasing diagnostic yield of community-onset bacteraemia within the emergency department: a retrospective study. PLoS ONE. 2019;14:e0222545.
Raupach-Rosin H, Duddeck A, Gehrlich M, Helmke C, Huebner J, Pletz MW, Mikolajczyk R, Karch A. Deficites in knowledge, attitude, and practice towards blood culture sampling: results of a nationswide mixed-methods study among inpatient care physicians in Germany. Infection. 2017;45:433–41.
Singler K, Bertsch T, Heppner HJ, Kob R, Hammer K, Biber R, et al. Diagnostic accuracy of three different methods of temperature measurement in acutely ill geriatric patients. Age Ageing. 2013;42:740–6.
Jamme M, Daviaud F, Charpentier J, Marin N, Thy M, Hourmant Y, et al. Time course of septic shock in immunocompromised and nonimmunocompromised patients. Crit Care Med. 2017;45:2031–9.
Wang HE, Shapiro NI, Griffin R, Safford MM, Judd S, Howard G. Chronic medical conditions and risk of sepsis. PLoS ONE. 2012;7:e48307.
Poutsiaka DD, Davidson LE, Kahn KL, Bates DW, Snydman DR, Hibberd PL. Risk factors for death after sepsis in patients immunosuppressed before the onset of sepsis. Scand J Infect Dis. 2009;41:469–79.
Schwarzkopf D, Rose N, Fleischmann-Struzek C, Boden B, Dorow H, Edel A, et al. Understanding the biases to sepsis surveillance and quality assurance caused by inaccurate coding in administrative health data. Infection. 2023. https://doi.org/10.1007/s15010-023-02091-y.
Henning C, Aygul N, Dinnetz P, Wallgren K, Ozenci V. Detailed analysis of the characteristics of sample volume in blood culture bottles. J Clin Microbiol. 2019;2019:e00268-e319.
Fleischmann C, Thomas-Rueddel DO, Hartmann M, Hartog CS, Welte T, Heublein S, et al. Hospital incidence and mortality rates of sepsis. Dtsch Arztebl Int. 2016;113:159–66.
Campanelli F, Landoni G, Cabrini L, Zangrillo A. Gender differences in septic intensive care unit patients. Minerva Anestesiol. 2018;84:504–8.
Chou EH, Mann S, Hsu T-C, Hsu W-T, Liu CC-Y, Bhakta T, et al. Incidence, trends, and outcomes of infection sites among hospitalizations of sepsis: a nationwide study. PLoS ONE. 2020. https://doi.org/10.1371/journal.pone.0227752.
Soto GJ, Martin GS, Gong MN. Healthcare disparities in critical illness. Crit Care Med. 2013;41:2784–93.
Pin M, Somasundaram R, Wrede C, Schwab F, Gastmeier P, Hansen S. Antimicrobial resistance control in the emergency department: a need for concrete improvement. Antimicrob Resist Infect Control. 2022;11:94.
Baumhäkel M, Müller U, Böhm M. Influence of gender of physicians and patients on guideline-recommended treatment of chronic heart failure in a cross-sectional study. Eur J Heart Fail. 2009;11:299–303.
Fleischmann-Struzek C, Thomas-Rüddel DO, Schettler A, Schwarzkopf D, Stacke A, Seymour CW, et al. Comparing the validity of different ICD coding abstraction strategies for sepsis case identification in German claims data. PLoS ONE. 2018;13:e0198847.
Acknowledgements
The authors acknowledge Gerald Brennan for editorial assistance.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
PG, SH and CB formulated the research question of this manuscript. CB handled data collection, data analysis and drafting of the manuscript. AG handled database processing and management. FS supervised the data analysis. BAL, RS and LH gave valuable input for optimizing the data collection and critically reviewed the article for important intellectual content. SH coordinated the study and was involved in the data analysis. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Approval from Charité-Universitätsmedizin Berlin ethics committee was obtained in December 2019, (EA4/211/19). Due to the retrospective use of anonymized data, patient consent was not required.
Consent for publication
Not applicable.
Conflict interests
The authors declare that they have no competing interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Berninghausen, C., Schwab, F., Gropmann, A. et al. Deficits in blood culture collection in the emergency department if sepsis is suspected: results of a retrospective cohort study. Infection (2024). https://doi.org/10.1007/s15010-024-02197-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s15010-024-02197-x