Abstract
Herbicide resistance is a major weed control issue that threatens the sustainability of rice cropping systems. Its epidemiology at large scale is largely unknown. Several rice weed species have evolved resistant populations in Italy, including multiple resistant ones. The study objectives were to analyze the impact in Italian rice fields of major agronomic factors on the epidemiology of herbicide resistance and to generate a large-scale resistance risk map. The Italian Herbicide Resistance Working Group database was used to generate herbicide resistance maps. The distribution of resistant weed populations resulted as not homogeneous in the area studied, with two pockets where resistance had not been detected. To verify the situation, random sampling was done in the pockets where resistance had never been reported. Based on data from 230 Italian municipalities, three different statistics, stepwise discriminant analysis, stepwise logistic regression, and neural network, were used to correlate resistance distribution in the main Italian rice growing area with seeding type, rotation rate, and soil texture. Through the integration of complaint monitoring, mapping, and neural network analyses, we prove that a high risk of resistance evolution is associated with traditional rice cropping systems with intense monoculture rates and where water-seeding is widespread. This is the first study that determines the degree of association between herbicide resistance and a few important predictors at large scale. It also demonstrates that resistance is present in areas where it had never been reported through extensive complaint monitoring. However, these resistant populations cause medium-low density infestations, likely not alarming rice farmers. This highlights the importance of integrated agronomic techniques at cropping system level to prevent the diffusion and impact of herbicide resistance or limit it to an acceptable level. The identification of concise, yet informative, agronomic predictors of herbicide resistance diffusion can significantly facilitate effective management and improve sustainability.
Similar content being viewed by others
1 Introduction
Weeds’ resistance to herbicides has been spreading quickly and is threatening the sustainability of crop production and therefore food security worldwide while increasing management costs (Mortensen et al. 2012; Hicks et al. 2018). In order to fulfill this challenge, large-scale data are needed to properly understand what occurs in the field (Editorial 2018). In several cropping systems, where diversity in space and time is low, resistance has reached epidemic level, destabilizing weed management in many agricultural areas (Orson 1999; Heap 2014; Busi et al. 2013; Evans et al. 2016). Efforts to prevent or slow down the selection of herbicide-resistant populations converge on reducing monoculture, diversifying herbicide strategies, promoting a judicious and correct use of herbicides, deepening the knowledge of weed biology and using a variety of agronomic techniques (Norsworthy et al. 2012; Evans et al. 2016). However, Hicks et al. (2018) found no evidence that using herbicides with different sites of action (SoA) reduces the evolution of resistance.
Italy is the largest rice producer in Europe with about 230,000 ha (2017 growing season). The main rice cultivation area is concentrated in the north-western regions of Piedmont and Lombardy where continuous paddy rice system is widespread. Weed management is one of the key aspects of rice cultivation because pedo-climatic conditions are favorable to weeds that are generally competitive, generate a persistent seed bank, and the weed flora is often dominated by difficult-to-control species, such as red rice and Echinochloa spp. Herbicide use is intense, with an average treatment frequency index higher than 2.5 (Ferrero and Vidotto 2010; Scarabel et al. 2013). In the last 15–20 years, the no. of available herbicide SoA has significantly decreased due to strict EU legislation, which led to several effective substances being withdrawn from the registration, and no new SoA is expected in the near future. Acetolactate synthase (ALS) inhibitors are by far the most used herbicides with more than 90% of rice fields treated at least once per year (Scarabel et al. 2012; Loddo et al. 2018).
Since the mid-1990s, several rice weed species have evolved resistant populations in Italy, including multiple resistant ones: Alisma plantago-aquatica, Schoenoplectus mucronatus, Cyperus difformis, Echinochloa spp. (Fig. 1), Oryza sativa f. spontanea (weedy red rice), and recently Cyperus esculentus L. (GIRE 2018; Loddo et al. 2018; Heap 2018; Sattin 2005; Scarabel et al. 2011, 2012, 2013; Panozzo et al. 2013). The Italian Herbicide Resistance Working Group (GIRE 2018) estimates that at least 40% of the rice cultivated area is affected by resistant populations.
The genus Echinochloa spp. includes the most troublesome species (Tabacchi et al. 2006) with some populations that have evolved multiple resistance (Panozzo et al. 2013). Difficulties in controlling Echinochloa spp. are not only related to their resistance status, but also their biological characteristics: very high seed production, long seed dormancy (Norris 1992; Holm et al. 1997), adaptation to water environment, C4 photosynthetic cycle, and high morphological variability (Tabacchi et al. 2006). This is why Echinochloa spp. is used as case study in this work.
Many publications describe the economic and agronomic impact of resistance, and many others suggest ways to prevent, slow down, or manage it (e.g., Norsworthy et al. 2012; Délye et al. 2013; Juraimi et al. 2013; Orson 1999). However, very few contribute to elucidating the impact of interactions between major agronomic and environmental factors on resistance epidemiology at a scale as large as the main rice production area in Italy (Evans et al. 2016). Epidemiology is concerned with the distribution and determinants of evolution in defined populations. It can provide relevant evidence-based information for preventing or reducing the spread of resistant populations by identifying the major risk factors.
Weed infestations and herbicide selection pressure in rice crops vary widely in relation to many agronomic and pedo-climatic conditions, so herbicide resistance evolves in a context of a series of interacting factors. However, detailed field-by-field data on these factors are rarely available at a large scale and therefore the identification of concise, yet informative, agronomic predictors of herbicide resistance distribution/diffusion would significantly facilitate effective management. The use of different statistical approaches, namely discriminant analysis, logistic regression, and artificial neural network, enables the quantification of the effect of pedo-climatic and management drivers on agro-ecological systems at large scale. For example, discriminant analysis was used to investigate the effect of rainfall-related variables on the occurrence of drought stress in maize (Zhang et al. 2013) and the effect of fertilizer regimes on the structure of the soil microbial community and its functions (Lazcano et al. 2013). Recently, a comparison of artificial neural networks and logistic regression was used to predict weed populations in chickpea and winter wheat (Mansourian et al. 2017) and to investigate the contribution of topographic and soil-related traits, as well as land use and maximum rainfall intensity as landslide drivers in landslide susceptibility mapping (Gong et al. 2018). The use of different approaches ensures a reliable depiction of the examined system as each approach relies on different assumptions and analytical solutions.
The objective of this study is to analyze the impact in Italian rice fields of two major agronomic techniques, seeding type (water- or dry-seeded), and crop rotation rate, as well as soil texture, on the diffusion of herbicide resistance. These factors are known to have a strong influence on rice weed infestations and crop cultivation more in general (Ferrero and Vidotto 2006). Data collected in 230 Italian municipalities distributed in the main rice growing area were processed with different statistical tests in order to detect the main drivers for the diffusion of resistance. We also aimed to estimate the risk of resistance evolution in the various municipalities and generate a resistance risk map.
2 Materials and methods
2.1 Data collection and mapping
Since 1997, the Italian Herbicide Resistance Working Group (GIRE, www.resistenzaerbicidi.it) has been monitoring, collecting, and testing putative herbicide-resistant populations nationwide, based on farmers and farmers’ advisor complaints of poor herbicide efficacy. All other available data on herbicide resistance in Italy were also collected, and all populations that were confirmed resistant to at least one herbicide through a standardized testing procedure (Panozzo et al. 2015a) were included in a national herbicide resistance database. The part of the database relative to the populations collected in the main rice producing area in Italy (approximately 200,000 ha, i.e., about 85% of the total rice growing area) was used as input to produce maps of herbicide resistance diffusion using iMAR application (Panozzo et al. 2015b; GIRE 2018). The maps were obtained by changing the color of the territory of the municipalities where at least one confirmed resistant population had been recorded. Therefore, municipalities with different numbers of resistant populations appear with the same color (Panozzo et al. 2015b). This, together with the nature of the monitoring done by GIRE, which is based on (a) end users complaints about herbicide failure and (b) priority given to samples collected in municipalities where herbicide resistance had not previously been reported, makes the output maps “qualitative” because they do not provide reliable information on the spread of resistance within each municipality. That is to say they indicate the areas at higher risk of resistance evolution.
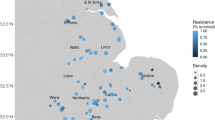
Two descriptive resistance maps were generated, one pooling all cases of resistance recorded since 1997 for the five rice weeds affected by herbicide resistance (A. plantago-aquatica, 66 cases; C. difformis, 29 cases; S. mucronatus, 81 cases; O. sativa f. spontanea—weedy red rice, 57 cases; and Echinochloa spp., 192 cases; Fig. 2a) and another for Echinochloa spp. only (Fig. 2b), which is the most diffused weed genus in Italian rice crops. A total of 584 populations were tested with three herbicides and 425 proved to be resistant to at least one of them. Most populations are resistant to ALS inhibitors, with a few Echinochloa spp. resistant to ACCase inhibitors or to both ALS and ACCase inhibitors.
Map of all resistant cases (a) and Echinochloa spp. only resistant cases (b) Recorded in the rice area: two “resistance-free” areas are evident: one (L) in Pavia province and a second (S) along the Sesia river. A municipality changes color when at least one population has been confirmed resistant in its territory. Different colors refer to resistance to herbicides with different site of action. Available online: www.resistenzaerbicidi.it (accessed on: 15 May 2018)
A database including all municipalities in the study area where rice fields represent more than 10% of the utilized agricultural area was created. An Italian municipality is a territory with autonomous administration that generally ranges in size from 15 to 40 km2. A single value for resistance presence/absence (regardless of weed species) was associated to each municipality: 0 where no resistance had been recorded and 1 where at least one case was confirmed. The percentage of rice cropping area under crop rotation (RR), percentage of water-seeded (WS) area, average percentage of sand (PS), and clay in soil (PC) were also added for each municipality. Given the very high correlation between the percentage of sand and silt (r = − 0.96), the latter was not considered in the analyses. A similar second database was created including only Echinochloa spp. Data were not available for four municipalities, which were therefore excluded from the analyses. Herbicide strategy at municipality level was not considered among the predictors because data are scant, not homogeneous, and generally too complex. Data on rotation and soil texture were obtained from regions’ environmental agencies: Regional Agency of Services for Agriculture and Forestry (Ente Regionale per i Servizi all’Agricoltura e alle Foreste, ERSAF) for Lombardy and Agriculture Registry Office (Anagrafe Agricola del Piemonte) for Piedmont, while records on water-seeding were supplied by the Ente Nazionale Risi. Raw data were provided in different formats and were therefore processed to fit our database, i.e., to obtain single data of WS, RR, PC, and PS for each municipality. Both WS and RR data were available for the period 2013–2015. The 3-year period was considered adequate because the diffusion of water-seeding and crop rotation had been relatively constant for several years before 2015.
WS data were received already aggregated at municipality level and expressed as a percentage of hectares of water-seeded rice in relation to the total area of each municipality. The percentage was then averaged over the 3 years. RR was instead calculated starting from the “land use database” of Lombardy and Piedmont Regions: data were expressed as hectares of land use type per cadastral plot. A cadastral plot is a physically continuous piece of territory located in a municipality with a single owner, quality, and culture class. A plot was considered “rotated” when not cropped with rice for at least one of the 3 years. Hectares of rotated plots were then summed and expressed as a percentage of the total area of each municipality.
PC and PS were calculated starting from data of texture of different soil types present in each municipality. A prevalent soil texture per municipality was then calculated by doing a weighted average of clay, sand, and silt content based on the no. of hectares of each soil type. Descriptive maps were then produced using QGIS software 2.14.9 to graphically display the variation in WS, PC, and RR in the study area. Herbicide strategy at municipality level was not considered among the predictors because data are scant, not homogeneous, and generally too complex.
2.2 Statistical analyses
Three different methods were selected: stepwise discriminant analysis, stepwise logistic regression, and artificial neural network. The stepwise discriminant analysis separates objects or observations in classes, or allocates new observations in already defined ones; the aim of the analysis was to define the probability of correctly classifying a resistant (1) or non-resistant (0) municipality on the basis of selected predictors. Logistic regression is a model that estimates the probability of a binary response on the basis of one or more independent variables; stepwise backward selection of the predictors was adopted for both analyses. The cut-off threshold of the probability was set according to the results of the receiver operating characteristic (ROC) curve analysis (Maiorano et al. 2009).
Neural network allows finding non-linear behavior of the system under analysis that cannot be discovered with traditional regression-based methods as logistic regression and discriminant analysis. Neural network has minimal requirement on model structure and assumptions because the form of the relationships is determined during the learning process (Haykin 2009). We used a multilayer perceptron network, using RR, WS, PS, and PC as variables to predict resistance (binary, 0/1). We used a 7/3 ratio to split the entire dataset in training and testing sets. The creation of the two sets was carried out by random choice and repeated 10 times. One hidden layer was used, with 2 to 4 neurons, using the hyperbolic tangent activation function (Vogl et al. 1988) and the softmax function for the output layer to assign decimal probabilities to the two output classes (Priddy and Keller 2005).
Resistance presence/absence was the dependent variable, while WS, RR, PC, and PS were the predictors. The analyses were done twice, first considering all five “resistant” weed species pooled together and then Echinochloa spp. only. All statistical analyses were performed with IBM SPSS 24 software. The alpha of the statistical tests was set at 0.05.
Previously created resistance maps were descriptive and gave important information but limited to the known diffusion of resistance in the municipalities. The next step was to generate stochastic maps of herbicide resistance risk evolution on the basis of the pseudoprobability of resistance calculated via the three statistical analyses. Specifically, we used the pseudoprobability obtained from the analysis with the highest prediction quality, namely the neural network which provided the most correct classification. QGIS software was then used to generate the maps based on the output of the analysis.
2.3 Echinochloa spp. case study
To verify whether the lack of herbicide-resistant weed populations observed in two areas (Fig. 2 areas L and S) was an artifact due to the nature of resistance monitoring done by GIRE, a random survey was done in these two areas. Echinochloa spp., the most troublesome weeds evolving herbicide resistance in rice crops, were chosen as case study. In September 2016, 20 populations of Echinochloa spp. were sampled following a grid of 5 × 5 km2. Seed samples were collected from plants of Echinochloa spp. present in the rice field closest to each node of the grid. The average size of the fields was about 2 ha, and each sample included seeds harvested from at least 5 plants. Samples from #300-L to #310-L were collected in area L while those from #311-S to #319-S came from area S. Sampling followed a density structured approach visually assessing the density of Echinochloa spp. in the sampled field. Infestation density was divided into four categories (very low: < 1 plant × 500 m−2, low: about 1 plant × 100 m−2, medium: about 1 plant × 10 m−2, high: about or more than 1 plant m−2). All seed samples were cleaned and dry stored at room temperature. Two whole-plant herbicide sensitivity experiments were then performed in greenhouse conditions following a standardized testing procedure (Panozzo et al. 2015a), in autumn 2016 (A) and a repetition in spring 2017 (S).
Seeds were chemically scarified for 20 min in sulfuric acid (96%) and then carefully rinsed with cold water. They were then placed in plastic boxes containing Pot Grown H peat (Klasmann Deilmann GmbH) and stored in a germination cabinet at 26/16 °C (day/night) under neon tubes providing a photosynthetic photon flux density (PFFD) of 15–30 μmol m−2 s−1 with a 12-h photoperiod.
At one leaf stage, seedlings were transplanted into pots with standard potting mix (60% silty loam soil, 15% sand, 15% perlite, 10% peat) and placed in a greenhouse, with the temperature ranging between 15 and 19 °C at night and 26–33 °C during the day. Light was supplemented using 400 W metal-halide lamps, which supplied about 450 μmol m−2 s−1 with a 16-h photoperiod.
All populations were tested for resistance to both ALS and acetyl coenzyme-A carboxylase (ACCase) inhibiting herbicides: penoxsulam (Viper, 40 g L−1, Dow Agrosciences, Bologna, Italy, recommended field dose 2 L ha−1), imazamox (Beyond, 40 g L−1, BASF Italia, recommended field dose 0.875 L ha−1), cyhalofop-butyl (Clincher One, 200 g L−1, Dow Agrosciences, recommended field dose 1.5 L ha−1), and profoxydim (Aura, 200 g L−1, BASF Italia, recommended field dose 0.5 L ha−1). Both imazamox and profoxydim were used along with the recommended surfactant Dash HC (methyl-palmitate and methyl-oleate 349 g L−1, BASF Italia) at 0.5% concentration and 0.9 L ha−1, respectively. Two known susceptible checks were included in the experiments. ALS herbicides were applied at two doses; the recommended field dose (1×) and three times that (3×), while ACCase herbicides were sprayed only at dose 1×. Plants were sprayed when they had reached 2–3 leaf stage (BBCH 12–13, Hess et al. 1997) using a precision bench sprayer delivering 300 L ha−1, at a pressure of 215 kPa, and a speed of 0.75 m s−1, with a boom equipped with three flat-fan (extended range) hydraulic nozzles (TeeJet, 11002). For each population, a non-treated control was included.
The experimental layout was a completely randomized design with three replicates of six plants each. Plant survival and shoot fresh weight were recorded 4 weeks after treatment and expressed as a percentage of the non-treated control. Plants were considered dead when they did not show any active growth, regardless of their color.
Populations were then divided into four categories: S when less than 5% of plants survived the 1× dose, SR when survival at 1× dose ranged between 5 and 20%, R when survival at 1× was > 20%, and RR when > 10% of plants survived the 3× dose and > 20% the recommended field dose (Panozzo et al. 2015a). To test whether the two experiments could be pooled, Levene’s test was performed: as both survival and fresh weight variances proved to be homogeneous (p = 0.22 and p = 0.74), data from the two experiments were pooled and analyzed together.
3 Results and discussion
3.1 Mapping and analyses of predictors
The dataset included a total of 232 municipalities, and in 115 (49.6%) of them, at least one resistant population had been found (GIRE 2018). Considering only Echinochloa spp., there were 78 municipalities (33.6%) where resistance had previously been confirmed.
By using the dynamic mapping system available on the Italian Herbicide Resistance Working Group website (GIRE 2018), it appeared that herbicide resistance in rice did not evolve evenly within the study area as it had not been reported in two relatively large pockets (Fig. 2 areas S and L) regardless of weed species or type of resistance. A non-homogeneous distribution of resistant populations was detected when all weeds (Fig. 2a), as well as when only Echinochloa spp., were considered.
Stepwise discriminant analysis including all weeds eliminated PS at the third step of the analysis, while for Echinochloa spp., only WS was retained after the first step. Discriminant analysis was able to correctly group 65.2% of “resistant” municipalities and 70.9% of “non-resistant” ones for all weeds, 64.1 and 65.6% for Echinochloa spp., respectively.
With the aim of setting the optimal cut-off value of probability in the logistic regression, we ran an ROC curve analysis using the predicted values of the logistic regression, which was first executed with a default cut-off probability value equal to 0.5, for both the datasets, i.e., the five weeds pooled together and the Echinochloa spp. one. Stepwise backward logistic regression performed on all weeds pooled together showed that WS, RR, and PC are highly correlated with resistance presence (p < 0.001, p = 0.003, and p = 0.009 respectively), whereas the correlation with PS resulted as not significant. For Echinochloa spp. alone, only WS resulted as significant (p < 0.001). RR and PC were negatively correlated with resistance while WS was positively correlated with it (see also Fig. 3a–c).
Descriptive maps of the three most important predictors (a–c) and resistance risk (d) maps of the area studied. a Percentage of rotation rate. b Percentage of clay in soil. c Percentage of water-seeding. d Map of resistance risk evolution created with neural network pseudoprobability considering all “resistant” weeds
Both statistical analyses highlighted the strict relation between the presence of resistance and the more traditional system of seeding rice in flooded paddies. This is reinforced by the observation that the five weed species that evolved herbicide-resistant populations are well adapted to humid and flooded conditions (Tabacchi and Viggiani 2017; Osuna et al. 2002). When all weeds were analyzed together, RR and PC were also significant and this is likely a consequence of using a larger dataset. It is clear that the three predictors are somehow inter-dependent, i.e., WS is less frequent in areas where PC is lower and RR is higher. The two virtually resistant-free areas S, and especially L (Fig. 2), display this pattern (Fig. 3a–c). Where WS is practiced, weed control strategies are generally based on fewer herbicide SoA and rely more on ALS inhibitors (Ferrero and Tinarelli 2008), thus increasing the herbicide selection pressure.
To our knowledge, this is the first study that determines the degree of correlation between herbicide resistance and a few important predictors at such a large scale (about 230,000 ha).
The maps generated by the GIRE website simply give a snapshot of diffusion of resistance based on complaint monitoring. Instead, we aimed to estimate the risk of resistance evolution in the various municipalities through an innovative approach such as neural network and generate a resistance risk map. With the aim of generating a resistance risk map, we used the pseudoprobability calculated through the neural network analysis, as the prediction model for resistance was better than that obtained with logistic regression and discriminant analysis. Using neural network, the effect of random choice of the cases was negligible for the training/testing sets and for the number of layers (10 attempts for each number of neurons); results of the simplest network are displayed in Table 1.
The prediction of the two cases was made using the percentage of rice cropping area under crop rotation (RR), the percentage of water-seeded (WS) area, the average percentage of sand (PS), and clay in soil (PC) as covariate predictors.
The neural network analysis confirmed that WS, PC, and RR are good predictors of resistance, with a normalized importance higher than 75%. In 48% of municipalities, the probability of resistance evolution is higher than 50%. Resistance risk is higher (risk > 60% in 64% of municipalities) in the central-western part of the study area (Piedmont region) than in the central-eastern area (Lombardy region, risk > 60% in 21% of municipalities) (Fig. 3d). It is worth mentioning that in the 60 municipalities where the risk is higher than 70%, the average WS and RR are about 88 and 16%, respectively. Comparison between the maps of WS, RR, and PC and the resistance risk map (Fig. 3) highlights again that the traditional rice cropping systems based on water-seeding and lack of rotation (Ferrero and Tinarelli 2008) are at higher risk. Therefore, contrary to what was presented in a recent article on a different cropping system (Hicks et al. 2018), we demonstrate that in areas where a combination of management strategies that increase system diversity are used, the evolution of resistance is slower.
3.2 Echinochloa spp. case study
The results of the screenings done on Echinochloa spp. populations sampled randomly in areas L and S (Fig. 2) disprove the initial hypothesis of lack of resistance in those areas. Only four accessions (300-L, 303-L, 307-L, and 310-L) resulted as still being susceptible to all four herbicides (Table 2), all of them coming from area L.
Sixteen populations proved to be resistant to at least one herbicide, whereas two accessions (313-S and 315-S) were multiple resistant to both ALS and ACCase inhibitors. The efficacy of penoxsulam was similar to that recorded for imazamox, while the efficacy of profoxydim was higher than cyhalofop (data not shown). ACCase herbicide resistance was weaker than ALS, and only two populations were resistant to this SoA. The dose effect for ALS herbicides was low (data not shown), indicating that a target-site-related resistance mechanism may be involved (Powles and Yu 2010; Panozzo et al. 2013; Scarabel et al. 2011). The results of the screening on randomly sampled populations proved that resistance is frequently present even in the two areas where it had not previously been recorded through complaint monitoring. However, most of the infestation densities recorded during sampling were medium to low (between about 1 plant × 10 m−2 and about 1 plant × 100 m−2), especially in area S. This suggests that the low level of infestation may not alarm farmers, so they do not complain about or report poor herbicide control. The low infestation density probably does not affect crop yield nor entail any economic loss. The generally low infestation levels are probably related to the higher level of diversity in the cropping systems (Renton et al. 2014; Norsworthy et al. 2012) practiced in these areas, especially in area L.; here, in fact, crop rotation and dry seeding are more frequent, mainly leading to the selection of different weed species (Juraimi et al. 2013).
4 Conclusions
We present a large dataset that meets the need to document and understand which agronomic factors drive herbicide resistance evolution in the field. By analyzing the available data on about 200,000 ha of Italian rice fields, we demonstrate that herbicide resistance is strongly correlated with management practices such as seeding type (water- or dry-seeded) and crop rotation, as well as soil clay content. Dry seeding and crop rotation rate are negatively correlated with resistance presence. Through the integration of complaint monitoring, mapping, and neural network analyses, we prove that a high risk of resistance evolution is associated with traditional rice cropping systems with intense rates of monoculture and in areas where water-seeding is widespread. Although the impact of agronomic practices and environmental factors on resistance evolution is well known, this is the first time that these interactions are analyzed at such a large scale.
Random sampling revealed that resistance is present even in the areas where previous monitoring based on farmers’ complaints had not revealed any resistant case. However, the density of resistant populations is medium-low, so possibly does not alarm rice farmers as they can manage the problem with practices that keep resistance at an acceptable level. This situation is confirmed by the resistance risk map, which shows that some risk is also present in these areas. It therefore appears that, although very useful for stakeholders, GIRE maps underestimate resistance.
We prove that resistance is present over the whole main rice growing area in Italy, likely related to the widespread and intense use of herbicides and standardization of cropping systems. Nevertheless, where appropriate agronomic practices such as dry seeding and crop rotation are implemented, and therefore diversity in the system is higher, the impact of resistance is lower and kept to an acceptable level.
The identification of concise, yet informative, agronomic predictors of diffusion of herbicide resistance can significantly facilitate effective management and improve sustainability.
References
Busi R, Vila-Aiub MM, Beckie HJ, Gaines TA, Goggin DE, Kaundun SS, Lacoste M, Neve P, Nissen SJ, Norsworthy JK, Renton M, Shaner DL, Tranel PJ, Wright T, Yu Q, Powles SB (2013) Herbicide-resistant weeds: from research and knowledge to future needs. Evol Appl 6(8):1218–1221. https://doi.org/10.1111/eva.12098
Délye C, Jasieniuk M, Le Corre V (2013) Deciphering the evolution of herbicide resistance in weeds. Trends Genet 29(11):649–658. https://doi.org/10.1016/j.tig.2013.06.001
Editorial (2018) Resistance is … complex. Nat Ecol Evol 2:405. https://doi.org/10.1038/s41559-018-0495-5
Evans JA, Tranel PJ, Hager AG, Schutte B, Wu C, Chatham LA, Davis AS (2016) Managing the evolution of herbicide resistance. Pest Manag Sci 72(1):74–80. https://doi.org/10.1002/ps.4009
Ferrero A, Tinarelli A (2008) Rice cultivation in the E.U. Ecological conditions agronomical practices. In: Capri E, Karpouzas DG (eds) Pesticide risk assessment in rice paddies: theory and practice. Elsevier B.V., Amsterdam, pp 1–23. https://doi.org/10.1016/B978-044453087-5.50002-3
Ferrero A, Vidotto F (2006) Weeds and weed management in Italian rice fields. In: Agro-economical traits of rice cultivation in Europe and India. Edizioni Mercurio, Vercelli, pp 55–72
Ferrero A, Vidotto F (2010) History of rice in Europe. In: Sharma SD (ed) Rice, origin antiquity and history. CRC Press, Boca Raton, pp 341–372. https://doi.org/10.1201/EBK1578086801-c11
GIRE - Italian Herbicide Resistance Working Group (2018) Database of herbicide resistance in Italy. www.resistenzaerbicidi.it. Accessed 15 Apr 2018
Gong QH, Zhang JX, Wang J (2018) Application of GIS-based back propagation artificial neural networks and logistic regression for shallow landslide susceptibility mapping in South China-take Meijiang river basin as an example. The Open Civil Engineering Journal 12(1):21–34. https://doi.org/10.2174/1874149501812010021
Haykin SO (2009) Neural networks and learning machines, 3rd edn. Pearson Publisher, London
Heap I (2014) Herbicide Resistant Weeds. In: Pimentel D, Peshin R (eds) Integrated Pest Management. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-7796-5_12
Heap I (2018) The International Survey of Herbicide Resistant Weeds. www.weedscience.org. Accessed 15 Apr 2018
Hess M, Barralis G, Bleiholder H, Buhr L, Eggers T, Hack H et al (1997) Use of the extended BBCH scale-general for the descriptions of the growth stages of mono and dicotyledonous weed species. Weed Res 37(6):433–441. https://doi.org/10.1046/j.1365-3180.1997.d01-70.x
Hicks HL, Common D, Coutts SR, Crook L, Hull R, Norris K, Neve P, Childs DZ, Freckerton RP (2018) The factors driving evolved herbicide resistance at a national scale. Nat Ecol Evol 2:529–536. https://doi.org/10.1038/s41559-018-0470-1
Holm L, Doll J, Holm E, Pancho J, Herberger J (1997) The world’s worst weeds: natural histories and distribution. Wiley, New York
Juraimi AS, Uddin MK, Anwar MP, Mohamed MTM, Ismail MR, Man A (2013) Sustainable weed management in direct seeded rice culture: a review. Aust J Crop Sci 7(7):989–1002
Lazcano C, Gómez-Brandón M, Revilla P, Domínguez J (2013) Short-term effects of organic and inorganic fertilizers on soil microbial community structure and function. Biol Fertil Soils 49(6):723–733. https://doi.org/10.1007/s00374-012-0761-7
Loddo D, Kudsk P, Costa B, Dalla Valle N, Sattin M (2018) Sensitivity analysis of Alisma plantago-aquatica L., Cyperus difformis L. and Schoenoplectus mucronatus (L.) Palla to penoxsulam. Agronomy 2018(8):220. https://doi.org/10.3390/agronomy8100220
Maiorano A, Reyneri A, Sacco D, Magni A, Ramponi C (2009) A dynamic risk assessment model (FUMAgrain) of fumonisin synthesis by Fusarium verticillioides in maize grain in Italy. Crop Prot 28(3):243–256. https://doi.org/10.1016/j.cropro.2008.10.012
Mansourian S, Darbandi EI, Mohassel MHR, Rastgoo M, Kanouni H (2017) Comparison of artificial neural networks and logistic regression as potential methods for predicting weed populations on dryland chickpea and winter wheat fields of Kurdistan province, Iran. Crop Prot 93:43–51. https://doi.org/10.1016/j.cropro.2016.11.015
Mortensen D, Egan J, Maxwell B, Ryan M, Smith R (2012) Navigating a critical juncture for sustainable weed management. Bioscience 62(1):65–84. https://doi.org/10.1525/bio.2012.62.1.12
Norris RF (1992) Relationship between inflorescence size and seed production in barnyardgrass (Echinochloa crus-galli). Weed Sci 40(1):74–78. https://doi.org/10.1017/S0043174500056988
Norsworthy JK, Ward SM, Shaw DR, Llewellyn RS, Nichols RL, Webster TM, Bradley KW, Frisvold G, Powles SB, Burgos NR, Witt WW, Barrett M (2012) Reducing the risks of herbicide resistance: best management practices and recommendations. Weed Sci 60(sp1):31–62. https://doi.org/10.1614/WS-D-11-00155.1
Orson J (1999) The cost to the farmer of herbicide resistance. Weed Technol 3(3):607–611. https://doi.org/10.1017/s0890037x0004628
Osuna MD, Vidotto F, Fischer AJ, Bayer DE, De Prado R, Ferrero A (2002) Cross-resistance to bispyribac-sodium and bensulfuron-methyl in Echinochloa phyllopogon and Cyperus difformis. Pestic Biochem Physiol 73(1):9–17. https://doi.org/10.1016/S0048-3575(02)00010-X
Panozzo S, Scarabel L, Tranel PJ, Sattin M (2013) Target-site resistance to ALS inhibitors in the polyploid species Echinochloa crus-galli. Pestic Biochem Physiol 105(2):93–101. https://doi.org/10.1016/j.pestbp.2012.12.003
Panozzo S, Scarabel L, Collavo A, Sattin M (2015a) Protocols for robust herbicide resistance testing in different weed species. J Vis Exp (101):e52923. https://doi.org/10.3791/52923
Panozzo S, Colauzzi M, Scarabel L, Collavo A, Rosan V, Sattin M (2015b) iMAR: an interactive web-based application for mapping herbicide resistant weeds. PLoS One 10(8):e0135328. https://doi.org/10.1371/journal.pone.0135328
Powles SB, Yu Q (2010) Evolution in action: plants resistant to herbicides. Annu Rev Plant Biol 61:317–347. https://doi.org/10.1146/annurev-arplant-042809-112119
Priddy K, Keller PE (2005) Artificial neural networks: an introduction. SPIE Press, Bellingham
Renton M, Busi R, Neve P, Thornby D, Vila-Aiub M (2014) Herbicide resistance modelling: past, present and future. Pest Manag Sci 70(9):1394–1404. https://doi.org/10.1002/ps.3773
Sattin M (2005) Herbicide resistance in Europe: an overview. In: Proc. BCPC International Congress – Crop Science & Technology, Glasgow, UK, pp 131–138
Scarabel L, Panozzo S, Varotto S, Sattin M (2011) Allelic variation of the ACCase gene and response to ACCase-inhibiting herbicides in pinoxaden target-site resistant Lolium spp. Pest Manag Sci 67(8):932–941. https://doi.org/10.1002/ps.2133
Scarabel L, Cenghialta C, Manuello D, Sattin M (2012) Monitoring and management of imidazolinone-resistant red rice (Oryza sativa L., var. sylvatica) in Clearfield® Italian paddy rice. Agron 2(4):371–383. https://doi.org/10.3390/agronomy2040371
Scarabel L, Cenghialta C, Panozzo S, Manuello D, Sattin M (2013) Resistance evolution and sustainability of the rice cropping system: the Italian case study. Proc. of the Conference “Global Herbicide Resistance Challenge”, Fremantle (Australia), 18–22 February 2013, p 105
Tabacchi M, Viggiani P (2017) Piante infestanti di risaie e canali. Botanica e riconoscimento. Edagricole, Bologna
Tabacchi M, Mantegazza R, Spada A, Ferrero A (2006) Morphological traits and molecular markers for classification of Echinochloa species from Italian rice fields. Weed Sci 54(6):1086–1093. https://doi.org/10.1614/WS-06-018R1.1
Vogl TP, Mangis JK, Rigler AK, Zink WT, Alkon DL (1988) Accelerating the convergence of the backpropagation method. Biol Cybern 59(4–5):257–263. https://doi.org/10.1007/BF00332914
Zhang Q, Zhang J, Yan D, Bao Y (2013) Dynamic risk prediction based on discriminant analysis for maize drought disaster. Nat Hazards 65(3):1275–1284. https://doi.org/10.1007/s11069-012-0406-z
Acknowledgements
The Piedmont and Lombardy Regions kindly provided the land use data and Ente Nazionale Risi the data relative to the seeding technique, i.e., water- and dry-seeded rice. We are grateful to all members of GIRE for contributing to herbicide resistance complaint monitoring and for stimulating discussions. Thanks also to Alison Garside for revising the English text.
Data availability statement
The datasets generated and analyzed during the current study are not publicly available due to the privacy law but are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Elisa Mascanzoni is an employee of DOW Agrosciences and a PhD candidate at the University of Padova. The whole research program is under the supervision of Maurizio Sattin of the Italian National Research Council (CNR) without any interference by DOW Agrosciences. The other authors declare that they have no conflict of interest.
About this article
Cite this article
Mascanzoni, E., Perego, A., Marchi, N. et al. Epidemiology and agronomic predictors of herbicide resistance in rice at a large scale. Agron. Sustain. Dev. 38, 68 (2018). https://doi.org/10.1007/s13593-018-0548-9
Accepted:
Published:
DOI: https://doi.org/10.1007/s13593-018-0548-9