Abstract
Allelopathy is an important ecological mechanism in natural and managed ecosystems. Its study is critical to understand natural plant behaviors, to isolate allelochemicals with herbicide potential, and to use the allelopathic genes in transgenic studies. Poaceae is an ecologically dominant plant family and it is economically important worldwide because its chemical diversity represents an important source to discover new molecules. From this viewpoint, Brazil is an interesting place to study, encompassing 197 genera of the Poaceae family, many of them being dominant in various biomes and some being native to Brazil. Here, we review the literature describing allelopathic activities involving grasses of the Poaceae family. We evaluate the experimental conditions used in these studies, we identify the allelochemicals involved, and, finally, we assess the applicability of allelopathy. Our main findings are (1) among the 47 Brazilian species studied for their allelopathic effects, only Bothriochloa barbinodis, Bothriochloa laguroides, Paspalum notatum, and Paspalum urvillei are native to Brazil; (2) 51% of the reviewed studies prepared extracts from the leaves and used lettuce as the target plant; and (3) 64% of the papers identified allelochemicals, of which 67% were phenolic acids. This first bibliographical survey on allelopathy in Poaceae species present in Brazil shows that less than 3% of the Brazilian species have been studied, suggesting it is an incipient research subject. Since this plant family is a valuable source of unknown natural products, refining such studies should contribute to a better understanding of the ecosystem relationships. Identification and isolation of grass allelochemicals should promote environmentally safer compounds with bioherbicide properties, in sustainable agriculture.
Similar content being viewed by others
-
1. Introduction
-
3.2 Target species
-
6. Conclusion
1 Introduction
The influence of some plant species on the growth of others in their vicinity remained an unexplained phenomenon (Rodrigues et al. 1992) until Theophrastus (300 B.C.) conceptualized it as allelopathy (Reigosa et al. 2013). In 1974, Elroy L. Rice defined allelopathy as the effect of one plant on another via the release of chemical compounds into the environment (Rice 1984). Currently, the most accepted concept of allelopathy is as “any process involving secondary metabolites produced by plants, algae, bacteria, and fungi that influence the growth and development of agriculture and biological systems” (Ias 1996).
Allelopathy is an important ecological mechanism in the natural and managed ecosystems. It is a phenomenon that influences the primary and secondary plant succession, encompassing all successional stages (Reigosa et al. 1999); the structure and composition of plant communities and the dynamics between different formations (Rizvi et al. 1992); the dominance of certain plant species that affect the local biodiversity (Reigosa et al. 1999); and agriculture, which is the target of most studies (Chou 1989). Considering the importance of allelopathy, several studies have been conducted on the subject, a great majority of which focused on species of economic interest. Allelopathic studies in Brazil also focused on agroecosystems, especially with cultivated and weedy plants. On the other hand, considering the territorial extent and the diversity of the Brazilian flora, studies on the allelopathic potential of native species are scarce (Ferreira et al. 1992).
The Poaceae family is ecologically the most dominant and economically the most important family in the world (Heywood 1978), with approximately 793 genera and 8,000–9,000 species (Sánchez-Moreiras et al. 2004). In Brazil, this family is represented by 197 genera and 1,368 species, many of which are native and dominant in several Brazilian biomes. However, despite the representativeness of this family, the knowledge about allelopathy and the allelochemicals present in these species is limited (Fig. 1).
In order to investigate works related to the allelopathic potential of Poaceae species present in Brazil, research literature were searched in the databases of Science Direct, the Portal of Periodicals of Capes (Coordination for the Improvement of Higher Education Personnel), and the Academic Google. For the search purpose, the following keywords were used: “allelopathy,” “allelochemicals,” “phytochemistry,” “chemical compounds,” and “bioprospecting,” besides the genera of Poaceae species present in Brazil, as described by Boldrini et al. (2010) and Pillar et al. (2009).
2 Poaceae genera native to Brazil with allelopathic effect
A total of 44 papers referring to 47 species were found for works related to the allelopathic effect of grasses present in Brazil (Table 1). Of these, 31 species occur in Brazil, but only four are native to the country, which include Bothriochloa barbinodis, Bothriochloa laguroides, Paspalum notatum, and Paspalum urvillei. The fact that only 2.56% of the Brazilian species have been studied in terms of their allelopathic potential shows that this area is still incipient for the Poaceae, especially, for the species native to the country.
Despite the popular belief of several authors that scientific studies on allelopathy has been successful in recent years (Reigosa et al. 2013), most studies so far refer to the interaction between crops and weeds and only few to the grasses native to Brazil.
Among the genera studied, Bothriochloa stands out as the most studied in relation to the allelopathic effect of its species, for which seven works were found (Table 1). Allelopathy in species of this genus is attributed to the production and release of essential oils and richness in sesquiterpenes and monoterpenes (Scrivanti 2010).
3 Experimental conditions
3.1 Plant organs used to prepare extracts
We found that most of the studies have investigated the allelopathic properties of the leaves of the Poaceae species. Only a small minority investigated seeds, inflorescences, and pollen (Fig. 2). These data are in agreement with those reported by Reigosa et al. (2013), who investigated the works of allelopathy in Brazil. According to them, the preference for leaves may reflect the fact that it is certainly easier to collect leaves than other parts and that leaves represent a large part of the litter produced by the vegetation biomass that directly impacts the growth of the seedling.
The leaves are considered to be the most metabolically active organ of a plant and, therefore, it is reasonable that they present greater diversity of allelochemicals and, consequently, greater allelopathic effect (Ribeiro et al. 2009). However, it is known that allelochemicals can be produced in different organs of a plant, including the stems, roots, flowers, and seeds (Parvez et al. 2003; Weston and Duke 2003), with varying concentrations from one organ to another (Hong et al. 2004). However, for grasses, the evidence indicates that the aerial part, followed by the roots and seeds, are the main sources of potentially allelopathic substances (Souza Filho 1995).
In seed allelopathy, it is worth considering that the seeds of several forage grass that contain phytotoxic compounds may inhibit the germination of other seeds in their vicinity, compromising the germination and establishment of one or more species in the mixture, which in turn compromises the performance of the pasture (Souza Filho and Alves 1998).
3.2 Target species
The effect of grass extracts was tested in different target species, including lettuce (Lactuca sativa) (Table 2), followed by cultivated species such as maize (Zea mays), wheat (Triticum aestivum), and alfalfa (Medicago sativa). Lettuce is considered as a bioindicator plant and has been used in several allelopathy researches because it presents with rapid germination and uniform initial growth, which are desirable attributes for experiments that compare the effects of different treatments (Reigosa et al. 2013).
Most of the past studies have evaluated the allelopathic effect of grasses on cultivated species, followed by native, forages, and invasive plants (Fig. 3). In the ecological context, once the allelopathy of a species is verified, this effect can be tested in species that live together in the field, as only then can the applicability of allelopathy and the interaction with neighboring plants be inferred. As pastures are constituted of several types of grasses, it is interesting to examine the allelopathic effect in other forage plants or in weeds.
3.3 Evaluation of allelopathic effect
The most common physiological parameters used to identify the allelopathic effects of grasses were germination (41.35%), followed by the root length (27.89%) and shoot length (15.38%) (Table 3). Although germination is largely considered to be less sensitive to the presence of allelochemicals (Oliveira et al. 2012), it is the most commonly used attribute in evaluations of allelopathy. As for the vegetative structures, the allelopathic effect is most observed in the root system (Yamagushi et al. 2011). However, the allelopathic effect is often not evident in the germination process or during the initial growth of the seedlings; therefore, the evaluation of the appearance of abnormalities becomes a valuable tool to study allelopathy (Ferreira and Aquila 2000). However, of all the studies evaluated, only 0.96% studies analyzed the presence of abnormalities in the target species, which may have underestimated the allelopathic effect of the tested plants.
Most of the research reported so far has been conducted under laboratory conditions or under controlled conditions. Although such studies are important for isolating variables and identifying the true factors involved in plant interactions, there is a need for further studies to describe the allelopathic properties of plants under natural conditions.
Based on this requirement, the type of research on allelopathy can be divided into two main categories: (i) the one that follows the concepts of an ecological approach, corresponding to studies on phenomena occurring in natural ecosystems (allelopathy sensu stricto) and (ii) the other one that follows the criteria and commercial and economic interests corresponding to studies based on interactions between cultivated species that do not occur naturally in the same habitat (applied allelopathy). In the first category, preliminary or complementary laboratory studies could replicate, under controlled conditions, the expected effects of rain or dew on substance leaching, mimicking the events in natural environment. In fact, the species under study should coexist in the same habitat (Reigosa et al. 2013).
Due to the complexity of this kind of study, only limited studies have successfully “completed the cycle” or, in other words, have shown the production of a specific metabolite by the allelopathic (donor) plant, its journey through the environment (soil, water, or atmosphere), its arrival at the target, and its influence on the affected (recipient) plant (Reigosa et al. 2013).
4 Allelochemicals in Poaceae genera native to Brazil
For the successful application of allelopathic properties of a plant, the identification of allelochemicals is required (Bhadoria 2011). In this bibliographical survey review, only thirty (30) papers were found on the identification of allelochemicals in 23 genera and thirty (30) species of Poaceae present in Brazil (Table 4). Among the species studied, 22 occur in Brazil and only one is native to this country, namely, Merostachys riedeliana (Table 4).
Chemicals that establish allelopathic influence are called allelochemicals and they are divided into classes, viz., water-soluble organic acids; simple unsaturated lactones; long-chain fatty acids and polyacetylenes; naphthoquinones, anthraquinones, and complex quinines; simple phenols; benzoic acid and derivatives; cinnamic acid derivatives; coumarins; flavonoids; condensed and hydrolysable tannins; terpenoids and steroids; amino acids and polypeptides; alkaloids and cyanohydrins; sulphides and glycosides; and purines and nucleosides (Rice 1984). Although allelochemicals may belong to any of these classes, they generally belong to the terpenoids (Llusià et al. 1996), phenolic compounds (Li et al. 2010), and alkaloids (Levitt and Lovett 1985), which are mainly responsible for allelopathy (Trezzi 2002; Taiz and Zeiger 2013).
The allelochemicals present in the Poaceae are diverse, ranging from phenols to quinones. The most clearly identified compounds of these can be broadly divided into four groups: phenolic acids, hydroxamic acids, alkaloids, and quinones (Sánchez-Moreiras et al. 2004). In this review, despite the great diversity of compounds (Table 4), the main allelochemicals were found to belong to the group of phenolic acids, flavonoids, alkaloids, and terpenoids, with a predominance of phenolic acids (67%) (Fig. 4) probably due to the methodological facility adopted to identify these compounds.
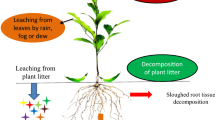
The plant that releases allelochemicals is known as the donor plant, whereas the plant that is influenced by the release of the allelochemicals is termed as the target plant or a recipient plant (Inderjit and Duke 2003). The release of the allelochemicals by the donor plant can happen through leaching, volatilization, and decomposition of the plant material or by release from the roots (Bhadoria 2011).
From the release process of the allelochemical by the donor plant to the effect in the recipient plant, several factors can influence the allelopathic activity. However, for the optimal use of allelopathy under field conditions, the influence of environmental factors needs to be investigated. In this context, the soil factor can be said to be the most important (Bhadoria 2011).
Understanding the effect of the relationships as well as the release form of the allelochemicals is crucial for designing alternatives for possible applications of these compounds. The first step is to know the potential of the species as well as to identify the compounds responsible for allelopathy. In this sense, a positive aspect observed in the last decades was that there was an increase both in the number of studies related to Brazilian Poaceae allelopathy and in those that isolated and identified allelochemicals from these species. In a review of allelopathy surveys conducted in Brazil, Reigosa et al. (2013) reached the same conclusion, that is, the last two decades has witnessed a proliferation of studies on the allelopathic properties of species introduced or cultivated in Brazil. These authors reported an evident increase in the number of publications, attributing it to the growing interest not only in allelopathic interactions in natural ecosystems and agroecosystems but also in products that can be derived from allelochemicals, such as natural herbicides and growth regulators.
The studies referring to the simple allelopathic activity of Brazilian Poaceae outnumber those that identify the allelochemicals (Fig. 5) owing to the comparatively greater ease, practicality, and lower costs of operations involved. In addition, bioprospecting for secondary metabolites in plants requires prior knowledge of biochemistry and molecular signaling among organisms, making them lengthier and more difficult to execute.
Although the search was made only for grasses present in Brazil, few of the works found in the survey performed the researches in this country. With respect to the allelopathy experiments, only 20% of the studies were performed in Brazil and a smaller percent (2%) of the allelochemical studies were developed in the country. This observation indicates that allelopathy in Brazil is an area of recent knowledge with much scope to expand.
5 Allelopathy and its applicability
Considering that Brazilian Poaceae can be found mainly in natural fields and cultivated pastures, it should be emphasized that allelopathy may play two extremely important roles in the pasture areas: (1) act as a management tool and (2) act as a supplier of basic structures for the production of bioherbicides (Souza Filho and Alves 1998).
Bioherbicides are environmentally safe alternatives, sources of new mechanisms of action, and have a structural diversity that has been attracting the attention of companies and researchers. Although research in the field of allelopathy, which aims at the search for bioherbicides, is only a recent process, some examples of natural compounds with potential use for weed control has already been established. For instance, in the case of Poaceae, the products 2-benzoxazolinone (BOA) and 6-methoxy-2,3-benzoxazolinone (MBOA) can be highlighted (Macías et al. 2007). In addition, we highlight sorgoleone—a lipid benzoquinone exuded from sorghum roots (Sorghum bicolor L.)—that reduces weed growth through its action on the PSII (Hejl and Koster 2004) by inhibiting the enzyme H+-ATPase in the roots. Sorgoleone affects the absorption of ions and the water balance of the plant, reducing its water absorption capacity (Soltys et al. 2013). It acts in a similar way to herbicides of the triazine class such as atrazine (Gniazdowska and Bogatek 2005).
On the other hand, allelopathy is important for the use of plants that control certain undesirable species (Resende et al. 2003). In this context, the identification of allelopathic forages and the knowledge of the mechanisms by which they exert their effects on the environment are of great importance as they provide a more adequate management of these plants in order to increase productivity and the persistence of pastures (Resende et al. 2003).
The reductions affected by forage grasses on the germination and development of the weeds assume an important aspect from the ecological perspective, because, with the decrease in the germination of the seeds, a reduction in the number of undesirable plants in the area were noted, thus reducing the competition power of these plants for water, light, and nutrients. Moreover, with the reduction in the development of the root system, the weeds have reduced their aggressive capacity. As a consequence of these two aspects, there is a greater possibility of establishing denser stands of desirable plants in cultivated pasture areas (Souza Filho and Alves 1998).
In the last few years, some studies involving the analysis of allelopathic activity have been developed with different species of forage grasses (Souza Filho et al. 2005). Studies involving forage grasses Brachiaria humidicola, Brachiaria decumbens, and Brachiaria brizantha cv. Marandu showed potentially allelopathic effects on the desmodium pastures (Desmodium adscendens), arrowleaf sida (Sida rhombifolia), and assa-fish (Vernonia polyanthes) (Souza Filho et al. 1997). The use of cover crops for the control of weeds is one of the earliest examples of the economic use of allelopathy. In addition to the suppressive effect of weeds, mulching has important effects on soil conservation and the maintenance of soil moisture (Medeiros 1989).
Considering these points, allelopathy assumes an important aspect from the point of view of pasture management as it allows not only the identification of forage species that can exert a certain level of control of certain undesirable species but also the establishment of grasses and legumes that are not strongly allelopathic to each other and which can compose more balanced pastures, with extremely favorable effects on their productivity and longevity (Wardle 1987). The difficulty in managing these two groups of physiologically different plants is one of the factors that prevent the establishment of intercropped pastures in several places across Brazil. Therefore, before recommending consortia with known species, it would be advisable to evaluate the allelopathic potential of the species considered (Rodrigues et al. 1992), considering that the allelopathic potential of the grasses can compromise the persistence of a legume in a consortium (Souza Filho and Alves 1998).
Another important role that allelopathy may play in terms of pasture management strategies and other crops is the possibility of obtaining pest- and disease-resistant species of plants. This bias opens up from the perspective of genetic transfer—through the manipulation of DNA—of this ability of a plant without major agronomic interests to a forage species of great interest to livestock, in which this characteristic was absent. The current phase of global research presents with practically no results in this regard. However, given the importance of this line of research, it is essential that studies be conducted in this sense (Souza Filho and Alves 1998).
The main problem in allelopathy works is to probe this phenomenon. It often happens due to the difficulty in separating allelopathy from competition. According to Duke (2015), most articles that claim to demonstrate allelopathy do not prove that it occurs. They only demonstrate that a crude extract of a plant species suspected to be allelopathic, or one or more compounds from such a plant, are phytotoxic in unrealistic bioassays that maximize the effects of the phytotoxin. This can be a first step in the proof of allelopathy, but all plants produce compounds that are weakly phytotoxic in simple bioassays conducted in the absence of soil. A successful demonstration of an allelopathic interaction has three components: (1) an ecological component—a demonstration that it exists in nature; (2) a chemical component—isolation, identification, and characterization of allelochemicals involved; and (3) a physiological component—identification of the interference mechanism at the biochemical, physiological, cellular, and molecular level (Inderjit and Weston 2000). Besides the difficulty of proving allelopathy, the studies that have been developed do not have an adequate standard methodology, which makes it difficult to compare them.
6 Conclusion
The allelopathic properties of Poaceae species present in Brazil are poorly studied. Moreover, most of the species described in studies as belonging to the genera native to Brazil are not actually native to the country. Most of the reported work was performed only under laboratory conditions with leaf extracts, using lettuce as a target plant and by evaluating simple growth characters such as germination. The number of papers that identified or isolated allelochemicals in Poaceae is lower than those that simply tested the allelopathic effect. Among the species studied, the great majority of identified allelochemicals belong to a group of phenolic acids. A fewer number of studies identified compounds responsible for allelopathy, reflecting the complexity and costs of this type of study.
Despite the increase in the number of studies on allelopathy in recent years, the fact that only a minority of these works have been performed in Brazil is worrisome as the information about the chemical composition and biological properties of native Poaceae is limited. Allelopathy is important to explain interactions both in the composition of natural ecosystems and in the interaction between cultivated and invasive plants. Therefore, to understand this phenomenon and to identify allelochemicals, it is fundamental to understand the natural behavior of plants and managed pastures, besides bioprospecting for allelochemicals with potential herbicide value. In addition, there is a need for further genetic and molecular studies of allelopathic plants toward increasing their protection against competitors as well as to identify allelopathic genes that can be used in transgeny.
References
Abdullah S, Gobilik J, Chong KP (2012) Preliminary phytochemical study and antimicrobial activity from various extract of Cynodon dactylon (L.) Pers. (bermuda) against selected pathogens. Int J Pharm Pharm Sci 4(5):227–230
Abu-Romman S, Ammari T (2015) Allelopathic effect of Arundo donax, a Mediterranean invasive grass. Plant Omics J 8(4):287–291
Ahmad W, Akbar M, Farooq U, Alia A, Khan F (2014) Allelopathic effects of aqueous extracts of Avena fatua on seed germination and seedling growth of Triticum aestivum (variety GW-273). J Environ Sci Toxicol Food Technol 8(2):38–42
Amini R, An M, Pratley J, Azimi S (2009) Allelopathic assessment of annual ryegrass (Lolium rigidum): bioassays. Allelopath J 24:67–76
An N, Pratley JE, Haig T (1996) Differential phytotoxicity of Vulpia species and their plant parts. Allelopath J 3(2):185–194
An N, Pratley JE, Haig T (2001) Phytotoxicity of Vulpia residues: III. Biological activity of identified allelochemicals from Vulpia myuros. J Chem Ecol 27(2):383–394
Asghari J, Tewari JP (2007) Allelopathic potentials of eight barley cultivars on Brassica juncea (L.) Czern. and Setaria viridis (L) p. Beauv. J Agric Sci Technol 9:165–176. https://doi.org/10.1023/A:1020853620102
Bai SB, Zhou GM, Wang YX (2013) Allelopathic potential of Phyllostachys edulis on two dominant tree species of evergreen broad-leaved forest in its invasive process. Chin J Environ Sci 34(10):4066–4072
Bartholomew OI, Maxwell E, Bitrus HJ (2013) Phytochemical compositions and in vitro antioxidant capacity of methanolic leaf extract of Axonopus compressus (P. Beauv.) Eur J Med Plant 3(2):254–265. https://doi.org/10.9734/EJMP/2013/1686
Bertoldi C, De Leo M, Ercoli L, Braca A (2012) Chemical profile of Festuca arundinacea extract showing allelochemical activity. Chemoecology 22:13–21. https://doi.org/10.1007/s00049-011-0092-4
Bhadoria PBS (2011) Allelopathy: a natural way towards weed management. Am J Exp Agr 1:7–20
Boldrini II, Ferreira PMA, Andrade BO, Schneider AA, Setubal RB, Trevisan R, Freitas EM (2010) Bioma Pampa diversidade florística e fisionômica. Editora Pallotti, Porto Alegre
Bomediano C (2013) The role of native species as biome resistance factors on molasses grass (Melinis minutiflora Beauv.) invasion in rupestrian fields in Minas Gerais State, Brazil. Dissertation, Universidade Federal de Minas Gerais. https://hdl.handle.net/1843/BUBD-A34GXH
Bostan C, Moisuc A, Radu F, Cojocariu L, Horablaga M, Sarateanu V (2010) Effect of Poa pratensis extracts on growing and development of perennial grasses seedlings. Res J Agr Sci 42:372–377
Bostan C, Butnariu M, Butu M, Ortan A, Butu A, Rodino S, Parvu C (2013) Allelopathic effect of Festuca rubra on perennial grasses. Rom Biotechl Lett 18(2):8190–8196
Carvalho SIC, Nascimento Júnior D, Alvarenga EM, Regazzi AJ (1993) Efeitos alelopáticos de Brachiaria brizantha cv. Marandu no estabelecimento de plantas de Stylosanthes guianensis var. vulgaris e cv. Bandeirantes Rev Bras Zootecn 22(6):930–937
Chou CH (1989) Allelopathic research of subtropical vegetation in Taiwan. IV. Comparative phytotoxic nature of leachate from four subtropical grasses. J Chem Ecol 15(7):2149–2159. https://doi.org/10.1007/BF01207445
Chou CH, Lee HF (1991) Allelopathic dominance of Miscanthus transmorrisonensis in an alpine grassland community in Taiwan. J Chem Ecol 17(11):2267–2281. https://doi.org/10.1007/BF00988007
Chou CH, Young CC (1975) Phytotoxic substances in twelve subtropical grasses. J Chem Ecol 1(2):183–193. https://doi.org/10.1007/BF00987867
Chung IM, Ali M, Ahmad A, Lim JD, Yu CY, Kim JS (2006) Chemical constituents of rice (Oryza sativa) Hulls and their herbicidal activity against duckweed (Lemna paucicostata Hegelm 381). Phytochem Anal 17:36–45. https://doi.org/10.1002/pca.879
Chung IM, Ahn JK, Yun SJ (2015) Identification of allelopathic compounds from rice (Oryza sativa L.) straw and their biological activity. Can J Plant Sci 58:815–821
De Almeida ARP, Rodrigues TJD, Santos JM (2000) Alelopatia de cultivares de Panicum maximum Jacq., sobre leguminosas forrageiras arbustivas e arbóreas. I - Avaliações em laboratório. Bol Ind Anim 57(2):113–127
de Medeiros ARM (1989) Determinação de potencialidades alelopáticas em agroecossistemas. Thesis, Universidade de São Paulo
Duke SO (2015) Proving allelopathy in crop-weed interactions. Weed Sci 63:121–132. https://doi.org/10.1614/WS-D-13-00130.1
Emeterio LS, Arroyo A, Canals RM (2004) Allelopathic potential of Lolium rigidum Gaud. on the early growth of three associated pasture species. Grass Forage Sci 59:107–112. https://doi.org/10.1111/j.1365-2494.2004.00410.x
Eussen JHH, Niemann GJ (1981) Growth inhibiting substances from leaves of Imperata cylindrica (L.) Beauv. Z Pflanz 102(3):263–266. https://doi.org/10.1016/S0044-328X(81)80229-2
Faria DA, Guarantini MTG (2011) Avaliação do potencial alelopático de Merostachys pluriflora um bambu nativo da Mata Atlântica. Sociedade de Ecologia do Brasil. http://www.seb-ecologia.org.br/xceb/resumos/664.pdf. Accessed 17 June 2015
Fernandes LAV, Miranda DLC, Sanquetta CR (2007) Potencial alelopático de Merostachys multiramea Hackel sobre a germinação de Araucaria angustifolia (Bert.) Kuntze. Rev Acad 5(2):139–146
Ferreira AG, Aquila MEA (2000) Alelopatia: uma área emergente da ecofisiologia. Rev Bras Fisiol Veg 12:175–204
Ferreira AG, Aquila MEA, Jacobi US, Rizvi V (1992) Allelopathy in Brazil. In: Rizvi SJH, Rizvi V (eds) Allelopathy: basic and applied aspects. Chapman & Hall, London, pp 243–250
Ghebrehiwot HM, Aremu AO, Staden JV (2014) Evaluation of the allelopathic potential of five South African mesic grassland species. Plant Growth Regul 72(2):155–162. https://doi.org/10.1007/s10725-013-9847-y
Gniazdowska A, Bogatek R (2005) Allelopathic interactions between plants: multisite action of allelochemicals. Acta Physiol 27(3):395–407. https://doi.org/10.1007/s11738-005-0017-3
Golpavar AR, Hadipanah A, Sepehri A, Salehi S (2015) Allelopathic effects of bermuda grass (Cynodon dactylon L. Pers.) extract on germination and seedling growth of basil (Ocimum basilicum L.) and common purslane (Portulaca oleracea L.). J Biodivers. Environ Sci 6(5):137–143
Hagan DL, Jose S, Lin CH (2013) Allelopathic exudates of cogongrass (Imperata cylindrica): implications for the performance of native pine savanna plant species in the Southeastern US. J Chem Ecol 39:312–322. https://doi.org/10.1007/s10886-013-0241-z
Hamidi R, Mazaheri D, Rahimian H, Alizadeh HM, Ghadiri H, Zeinaly H (2006) Inhibitory effects of wild barley (Hordeum spontaneum Koch.) residues on germination and seedling growth of wheat (Triticum aestivum L.) and its own plant. BIABAN J 11:35–43
Hejl AM, Koster KL (2004) The allelochemical sorgoleone inhibits root H+-ATPase and water uptake. J Chem Ecol 30(11):2181–2191. https://doi.org/10.1023/B:JOEC.0000048782.87862.7f
Heywood VH (1978) Flowering plants of the world. Mayflower Books, New York
Hong NH, Xuan TD, Eiji T, Khanh TD (2004) Paddy weed control by higher plants from Southeast Asia. Crop Prot 23(3):255–261. https://doi.org/10.1016/j.cropro.2003.08.008
Hoult AHC, Lovett JV (1993) Biologically active secondary metabolites of barley: a method for identification and quantification of hordenine and gramine in barley by high-performance liquid chromatography. J Chem Ecol 19(10):2245–2254. https://doi.org/10.1007/BF00979661
Hussain F, Ahmad B, Ilahi I (2010) Allelopathic effects of Cenchrus ciliaris L. and Bothriochloa pertusa (L.) A. Camus. Pak J Bot 42(5):3587–3604
IAS (1996) Constitution and Bylaw of IAS. IAS Newsletter, Cadiz
Inderjit, Duke SO (2003) Ecophysiological aspects of allelopathy. Planta 2017:529–539. https://doi.org/10.1007/s00425-003-1054-z
Inderjit, Weston LA (2000) Are laboratory bioassays for allelopathy suitable for prediction of field responses? J Chem Ecol 26(9):2111–2118. https://doi.org/10.1023/A:1005516431969
Ishimine Y, Nakama M, Matsumoto S (1987) Allelopathic potential of Paspalum urvillei STEUD., Bidens pillosa L. var. radiata SCHERFF., and Stellaria aquatica Scop., dominant weeds in sugarcane fields in the Ryukyu Islands. Weed Res 32(4):274–281
Jung WS, Kim KH, Ahn JK, Hahn SJ, Chung IM (2004) Allelopathic potential of rice (Oryza sativa L.) residues against Echinochloa crus-galli. Crop Prot 23:211–218. https://doi.org/10.1016/j.cropro.2003.08.019
Kannan D, Priyal SB (2015) Phytochemistry study in three selected ecotypes of Cenchrus ciliaris L. grass. Int J Multidiscip Res 1(1):56–59
Kato-Noguchi H, Kobayashi A, Ohno O, Kimura F, Fujii Y, Suenaga K (2014) Phytotoxic substances with allelopathic activity may be central to the strong invasive potential of Brachiaria brizantha. J Plant Physiol 171:525–530. https://doi.org/10.1016/j.jplph.2013.11.010
Khuzhaev VU (2004) Alkaloids of Arundo donax. XVIII. Nitrogenous bases in flowers of cultivars. Chem Nat Compd 40(5):516–517. https://doi.org/10.1007/s10600-005-0025-y
Koger GH, Bryson CT (2004) Effect of cogongrass (Imperata cylindrica) extracts on germination and seedling growth of selected grass and broadleaf species. Weed Technol 18:236–242. https://doi.org/10.1614/WT-03-022R1
Kremer JR, Ben-Hammouda M (2009) Allelopathic plants. 19. Barley (Hordeum vulgare L). Allelopath J 24(2):225–242
Levitt J, Lovett JV (1985) Alkaloids, antagonisms and allelopathy. Biol Agric Hortic 2(4):289–301. https://doi.org/10.1080/01448765.1985.9754443
Li ZH, Wang K, Ruan X, Pan CD, Jiang DA (2010) Phenolics and plant allelopathy. Molecules 15(12):8933–8952. https://doi.org/10.3390/molecules15128933
Llusià J, Estiarte M, Peñuelas J (1996) Terpenoids and plant communication. Butll Inst Cat Inst Nat 64:125–133
Ma Y, Zhang M, Li Y, Shui J, Zhou Y (2014) Allelopathy of rice (Oryza sativa L.) root exudates and its relations with Orobanche cumana Wallr. and Orobanche minor Sm. germination. J Plant Interact 9(1):722–730. https://doi.org/10.1080/17429145.2014.912358
Macías FA, Molinillo JMG, Varela RM, Galindo JCG (2007) Allelopathy—a natural alternative for weed control. Pest Manag Sci 63:327–348. https://doi.org/10.1002/ps.1342
Mahmoodzadeh H, Mahmoodzadeh M (2013) Allelopathic effects of Cynodon dactylon L. on germination and growth of Triticum aestivum. Annals Biol Res 5(1):118–123
Mahmoodzadeh H, Mahmoodzadeh M (2014) Allelopathic effects of rhizome aqueous extract of Cynodon dactylon L. on seed germination and seedling growth of legumes, Labiatae and Poaceae. Iran. J Plant Physiol 4(3):1047–1054
Martin LD, Smith AE (1994) Allelopathic potential of some warm-season grasses. Crop Prot 13(5):388–392. https://doi.org/10.1016/0261-2194(94)90055-8
Mbuthia KS (1997) Analysis of volatile phytochemical for wild non-host plant, Melinis minutiflora P. Beav., and wild host plant Pennisetum purpureum(K) sihumach of Chilo portellus (swinhoe). Dissertation, Kenyatta University
Mold R (2005) Testing the allelopathic effect of Festuca paniculata in Sub-Alpine Grasslands. La Station Alpine Joseph Fourier. http://www.jardinalpindulautaret.fr/sites/sajf/files/pdf/stage2005MOLD.pdf. Acessed 27 August 2015
Murphy SD, Aarssen LW (1995) Allelopathic pollen extract from Phleum pratense L. reduces germination (in vitro) off pollen of sympatric species. Int J Plant Sci 156(4):425–434. https://doi.org/10.1086/297264
Nascimento EA, Terrones MGH, Morais SAL, Chang R, Andrade GA, Santos DQ, Pereira BHA (2009) Allelopathic activity of Cenchrus echinatus L. extracts on weeds and crops. Allelopath J 24(2):363–372
Novo MCSS, Deuber R, Lago AA, Araújo RT, Santini A (2009) Efeito de extratos aquosos de estruturas de grama-seda no desenvolvimento inicial de plântulas de arroz, milho e trigo. Bragantia 68:665–672. https://doi.org/10.1590/S0006-87052009000300013
Ogie-Odia EA, Eseigbe D, Ilechie MN, Erhabor J, Ogbebor E (2010) Foliar epidermal and phytochemical studies of the grasses Cymbopogon citratus (Stapf.), Axonopus compressus (P. Beauv.) and Eragrostis tremula (S.W. Beauv.) in Ekpoma, Edo State, Nigeria. Sci World J 5(1):20–24
Oliveira SCC, Gualtieri SCJ, Domínguez FAM, Molinillo JMG, Montoya RV (2012) Estudo fitoquímico de folhas de Solanum lycocarpum A. St.-Hil (Solanaceae) e sua aplicação na alelopatia. Acta Bot Bras 26(3):607–618. https://doi.org/10.1590/S0102-33062012000300010
Parveen I, Wilson T, Donnison IS, Cookson AR, Hauck B, Threadgill MD (2013) Potential sources of high value chemicals from leaves, stems and flowers of Miscanthus sinensis ‘Goliath’ and Miscanthus sacchariflorus. Phytochemistry 92:160–167. https://doi.org/10.1016/j.phytochem.2013.04.004
Parvez SS, Parvez MM, Fujii Y, Gemma H (2003) Allelopathic competence of Tamarindus indica L. root involved in plant growth regulation. Plant Growth Regul 41:139–148. https://doi.org/10.1023/A:1027387126878
Pillar VP, Muller SC, Castilhos ZMS, Jacques AVA (2009) Campos Sulinos. Conservação e uso sustentável da biodiversidade. MMA, Brasília
Rasmussen JA, Rice EL (1971) Allelopathic effect of Sporobolus pyramidatus on vegetational patterning. Am Midl Nat 86:309–326. https://doi.org/10.2307/2423626
Reigosa MJ, Sanchéz-Moreiras A, González L (1999) Ecophysiological approach in allelopathy. Crc Rev Plant Sci 18(5):577–608. https://doi.org/10.1080/07352689991309405
Reigosa M, Gomes AS, Ferreira AG, Borghetti F (2013) Allelopathic research in Brazil. Acta Bot Bras 27(4):629–646. https://doi.org/10.1590/S0102-33062013000400001
Resende PC, Pinto JC, Evangelista AR et al. (2003) Alelopatia e suas interações na formação e manejo das pastagens. UFLA. http://livraria.editora.ufla.br/upload/boletim/tecnico/boletim-tecnico-54.pdf. Accessed 13 June 2015.
Ribeiro JP, Matsumoto RS, Tadao LK, Voltarelli VM, Lima MIS (2009) Efeitos alelopáticos de extratos aquosos de Crinum americanum L. Rev Bras Bot 32(1):183–188
Rice EL (1984) Allelopathy. Academic Press, New York
Rizvi SJH, Haque H, Singh VK, Rizvi V (1992) A discipline called allelopathy. In: Rizvi SJH, Rizvi V (eds) Allelopathy: basic and applied aspects. Chapman & Hall, London, pp 1–10
Rodrigues LRA, Rodrigues TJD, Reis RA (1992) Alelopatia em plantas forrageiras. UNESP/FUNEP, Jaboticabal
Samedani B, Juraimi AS, Rafii MY, Anuar AR, Awadz SAS, Anwar MP (2013) Allelopathic effects of litter Axonopus compressus against two weedy species and its persistence in soil. Sci World J 8:16–24. https://doi.org/10.1155/2013/695404
Sánchez-Moreiras AM, Weiss OA, Reigosa-Roger MJ (2004) Allelopathic evidence in the Poaceae. Bot Rev 69(3):300–319. https://doi.org/10.1663/0006-8101
Santos JCF, Souza IF, Mendes ANG, Morais AR, Conceição HEO, Marinho JTS (2002) Efeito de extratos de cascas de café e de arroz na emergência e no crescimento de caruru-de-mancha. Pesqui Agropecu Bras 37:783–790. https://doi.org/10.1590/S0100-83582001000200007
Scognamiglio M, Fiumano V, D’Abrosca B, Pacifico S, Messere A, Esposito A, Fiorentino A (2012) Allelopathic potential of alkylphenols from Dactylis glomerata subsp. hispanica (Roth) Nyman. Phytochem Lett 5:206–210. https://doi.org/10.1016/j.phytol.2011.12.009
Scrivanti LR (2010) Allelopathic potential of Bothriochloa laguroides var. laguroides (DC.) Herter (Poaceae: Andropogoneae). Flora 205:302–305. https://doi.org/10.1016/j.flora.2009.12.005
Scrivanti LR, Anton AM, Zygadlo JA (2011) Allelopathic potential of South American Bothriochloa species (Poaceae: Andropogoneae). Allelopath J 28(2):189–200
Shivakoti C, Ramanjaneyelu K, Ramesh A (2015) Preliminary phytochemical screening of Setaria verticillata. Indo Am J Pharm Res 5(6):2425–2429
Shui J, An Y, Ma Y, Ichizen N (2010) Allelopathic potential of switchgrass (Panicum virgatum L.) on perennial ryegrass (Lolium perenne L.) and alfalfa (Medicago sativa L.) Environ Manag 46:590–598. https://doi.org/10.1007/s00267-010-9454-x
Slater PD, Cregan TM, Cregan PD (1996) Allelopathic effects of Danthonia richardsonii (cv. taranna) and Phalaris aquatica (cv. sirolan) on Trifolium subterraneum (cv. seaton park). In: Eleventh Australian Weeds Conference Proceedings, 1996, Weed Science Society of Victoria Inc., Melbourne, Victoria, Australia, pp 279–282
Soltys D, Krasuska U, Bogatek R, Gniadowska A (2013) Allelochemicals as bioherbicides—present and perspectives. In: Price AJ, Kelton J (eds) Herbicides current research and case studies in use. InTech, Warsaw
Souza Filho APS (1995) Potencialidades alelopáticas envolvendo gramíneas e leguminosas forrageiras e plantas invasoras de pastagens. Thesis, Universidade Estadual de São Paulo
Souza Filho APS, Alves SM (1998) Alelopatia em ecossistema de pastagem cultivada. Embrapa-CPATU, Belém
Souza Filho APS, Rodrigues LRA, Rodrigues TJD (1997) Inibição da germinação e alongamento da radícula de invasoras de pastagens pelos extratos aquosos de gramíneas forrageiras tropicais. Past Trop 19(1):45–50
Souza Filho APS, Pereira AAG, Bayma JC (2005) Aleloquímico produzido pela gramínea forrageira Brachiaria humidicola. Planta Daninha 23(1):25–32. https://doi.org/10.1590/S0100-83582005000100004
Taiz L, Zeiger E (2013) Fisiologia Vegetal. ARTMED, Porto Alegre
Tan HY, Sieo CC, Abdullah N, Liang JB, Huang XD, Ho YW (2011) Effects of condensed tannins from Leucaena on methane production, rumen fermentation and populations of methanogens and protozoa in vitro. Anim Feed Sci Technol 169:185–193. https://doi.org/10.1016/j.anifeedsci.2011.07.004
Tang CS, Young CC (1982) Collection and identification of allelopathic compounds from the undisturbed root system of Bigalta limpograss (Hemarthria altissima). Plant Physiol 69:155–160
Tantiado RG, Saylo MC (2012) Allelopathic potential of selected grasses (family Poaceae) on the germination of lettuce seeds (Lactuca sativa). Int J Bio-Sci Bio-Technol 4(2):27–34
Torres R, Jose C, Shirasuna R, Grombone-Guaratini MD (2014) Phenolic acids and C-glycoside flavonoids in Merostachys riedeliana (bamboo). Planta Med 80:33–34. https://doi.org/10.1055/s-0034-1394976
Trezzi MM (2002) Avaliação do potencial alelopático de genótipos de sorgo. Thesis, Universidade Federal do Rio Grande do Sul
Wardle DA (1987) Allelopathy in the New Zealand grassland/pasture ecosystem. N Z J Exp Agric 15:243–255. https://doi.org/10.1080/03015521.1987.10425567
Weston LA, Duke SO (2003) Weed and crop allelopathy. Crit Rev Plant Sci 22:367–389. https://doi.org/10.1080/713610861
Xuan TD, Toyama T, Fukuta M, Khanh TD, Tawata S (2009) Chemical interaction in the invasiveness of cogon grass (Imperata cylindrica (L.) Beauv.) J Agric Food Chem 57:9448–9453. https://doi.org/10.1021/jf902310j
Yamagushi MQ, Gusman GS, Vestena V (2011) Efeito alelopático de extratos aquosos de Eucalyptus globulus Labill. e de Casearia sylvestris Sw. sobre espécies cultivadas. Semina 32(4):1361–1374. https://doi.org/10.5433/1679-0359
Yamamoto Y, Fugii Y (1997) Exudation of allelopathic compound from plant roots of sweet vernalgrass (Anthoxanthum odoratum). J Weed Sci Technol 42(1):31–35
Zain NMD, Yew OH, Sahid I, Seng CT (2013) Potential of napier grass (Pennisetum purpureum) extracts as a natural herbicide. Pak J Bot 45(6):2095–2100
Zheng H, He CQ, Xu QY et al. (2011) Interference of allelopathy about Spartina alterniflora to Scirpus mariqueter by effects of activated carbon on soil. Procedia Environ Sci 10:1835–1840
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Favaretto, A., Scheffer-Basso, S.M. & Perez, N.B. Allelopathy in Poaceae species present in Brazil. A review. Agron. Sustain. Dev. 38, 22 (2018). https://doi.org/10.1007/s13593-018-0495-5
Accepted:
Published:
DOI: https://doi.org/10.1007/s13593-018-0495-5