Abstract
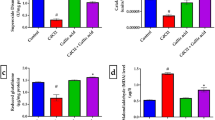
The present study aimed to investigate uranium hazardous effects, as well as the ameliorative effects of cardamom on the cerebellum and midbrain of adult male albino rats at different time intervals. The administration of 40 mg/Kg of uranyl acetate dehydrate (UAD) caused a significant decrease in Pi, K+ ions contents and GSH level while, a significant increase in Na+ and Ca2+ ions content and MDA level were observed, which may be due to the decrease in ATP synthesis and the inhibition of Na+/K+ ATPase activity. Also, UDA has the ability to increase lipid peroxidation and decrease GSH synthesis. On the other hand, the administration of 250 mg/kg of cardamom extract induced significant increase in Pi, K+ ions content and GSH level, while it caused a significant decrease in Na+, Ca2+ ions content and MDA level which may be due to the increase of ATP synthesis and its anticonvulsant and antioxidant effects. It could be concluded that the pre -and post-treatment with cardamom could ameliorate the hazardous effects of UAD intoxication.
Similar content being viewed by others
References
Kamath, D., Arunkumar, D., Avinash, N. & Samshuddin, S. Determination of total phenolic content and total antioxidant activity in locally consumed food stuffs in Moundbird, Karnataka and India. Adv. Appl. Sci. Res. 6, 99–102 (2015).
Aqil, F., Ahmed, I. & Mehmood, Z. Antioxidant and free radical scavenging properties of twelve traditionally used Indian medicinal plants. Turk. J. Biol. 30, 177–183 (2006).
Smarth, R. M. & Kumar, A. Radioprotection of Swiss albino mice by plant extract Mentha piperita (Linn.). J. Radiat. Res. 44, 101–109 (2003).
Lahlou, S., Figuereido, A. F., Magalhães, P. J. C. & LealCardoso, J. H. Cardiovascular effects of 1,8-cineole, a terpenoid oxide present in many plant essential oils, in normotensive rats. Can. J. Physiol. Pharmacol. 80, 125–130 (2002).
Vishwakarma, S., Chandan, K., Caroline, R. & Khushbu, S. Comparative study of Qualitative Phytochemical screening and antioxidant activity of Mentha arvensis, Elettaria cardamomum and Allium porrum. Indo Ameri. J. Pharma. Res. 4, 2538–2556 (2014).
Abdel-Kader, S., Abdel-Rahman, M., Bauomi, A., Mohammaden, T. & Rezk, M. M. Antioxidant Potentials of (Elletaria cardamomum) Cardamom against Uranium Hazards. Int. J. Basic. Life. Sci. 3, 122–138 (2015).
Mahady, G. B. et al. In vitro susceptibility of Helicobacter pylori to botanical extracts used traditionally for the treatment of gastrointestinal disorders. Photother. Res. 19, 988–991 (2005).
Bhat, N. et al. Comparison of the efficacy of cardamom (Elettaria cardamomum) with pioglitazone on dexamethasone-induced hepatic steatosis, dyslipidemia, and hyperglycemia in albino rats. J. Adv. Pharma. Techno. Res. 3, 136–140 (2016).
Suneetha, W. J. & Krishnakantha, T. P. Cardamom extract as inhibitor of human platelet aggregation. Phytother. Res. 19, 437–440 (2005).
Sengupta, A., Ghosh, S. & Bhattachar-jee, S. Dietary cardamom inhibits the formation of azoxymethane induced aberrant crypt foci in mice and reduces COX-2 and iNOS expression in the colon. Asian. Pac. J. Cancer Prev. 6, 118–122 (2005).
Gilani, A. H., Jabeen, Q., Khan, A. U. & Shah, A. J. Gut modulatory, blood pressure lowering, diuretic and sedative activities of cardamom. J. Ethnopharmacol. 115, 463–472 (2008).
WNA. “World Nuclear As-sociation: World Uranium Mining Production. London. http://www.worldnuclear.org/uploadedFiles/Pocket%20 (2013).
WNA., World Nuclear Association: WNA Public Information Service. London. http://www.world-nuclear.org/infomap.aspx (2014).
Keith, L., Faroon, O. M. & Fowler, B. A. in Hand book of toxicology of metal (eds Gunnar F. Nordberg) 1307-1345 (Rollins School of Public Health Fairbanks, USA 2015).
Hensley, K. Neuro-inflammatory aberrations of arachidonate pathway in ALS. Neuroscience. Bioachm. Biophys. Res. Commun. 82, 70–74 (2004).
Castro-Fernandez, C., Maya-Nunez, G. & Conn, P. M. Beyond the signaling sequence: Protein routing in health and disease. Endocr. Rev. 26, 25–40 (2005).
Valko, M., Rhodes, C. J., Moncol, J., Izakovic, M. & Mazur, M. Free radicals, metals and antioxidants in oxidative stress induced cancer. Chem. Biol. Interact. 160, 1–40 (2006).
McDiarmid, M. et al. The Gulf War depleted uranium cohort at 20: bioassay results and novel approachesto fragment surveillance. Health Phys. 104, 347–361 (2013).
Tissandie, E. et al. In vivo effects of chronic contamination with depleted uranium on vitamin D3 metabolism in rat. Biochim. Biophys. Acta., 1770, 266–272 (2007).
Katz, S. A. The Chemistry and Toxicology of Depleted Uranium. Toxics. 2, 50–78 (2014).
Arnault, E., Doussau, M. & Pesty, A. Natural uranium disturbs mouse folliculogenesis in vivo and oocyte meiosis in vitro. Toxicology 247: 80–87 (2008).
Abdel-Rahman, M., Bauomi, A., Abdel-Kader S., Mohammaden, T. & Rezk, M. M. Effect of Cardamom (Elletaria cardamomum) on Brain Ions and Acetylcholine Esterase Enzyme of Albino Rats Ingested Uranium. Int. J. Basic. Life. Sci. 3, 122–138 (2015).
Paquet, F. et al. Accumulation and distribution of uranium in rats after chronic exposure by ingestion. Health. Phys. 90, 139–147 (2006).
Kalinich, J. & Kasper, C. Do metals that translocate to the brain exacerbate traumatic brain injury? Med.l Hypotheses 82, 558–562 (2014).
Radcliffe, P. M. et al. Acute sodium tungstate inhalation is associated with minimal olfactory transport of tungsten (188W) to the rat brain. Neurotoxicology 30, 445–450 (2009).
Guyton, A. G. & Hall, J. E. in Text book of medical physiology (eds Reproductive and hormonal faction of male) 996–1011 (2006).
Oliver, J. A further study of the regenerated epithelium in chronic uranium nephritis: an anatomical investigation of its function. J. Exp. Med. 23, 301–321 (1916).
Taulan, M. et al. Renal toxicogenomic response to chronic uranyl nitrate insult in mice. Environ. Health Perspect. 112, 1628–1635 (2004).
Ericinska, M. & Silver, I. A. Ions and energy in mammalian brain. Prog. Neurobiol. 43, 37–71 (1994).
Franco, A. A., Odom, R. S. & Rando, T. A. Regulation of antioxidant enzymes gene expression in response to oxidative stress and during differentiation of mouse skeletal muscle. Free Radic. Biol. Med. 50, 2093–2098 (1999).
Shaki, F., Hosseini, M., Ghazi-Khansari, M. & Pourahmad, J. Toxicity of depleted uranium on isolated rat kidney mitochondria. Biochimica. et Biophysica. Acta. 1820, 1940–1950 (2012).
Beal, M. F. Aging, energy and oxidative stress in neurodegenerative diseases. Ann. Neurol. 38, 357–366 (1995).
Abou-Donia, M. B. et al. Uranyl acetate-induced sensorimotor deficit and increased nitric oxide generation in the central nervous system in rats. Pharmacol. Biochem. Behav. 72, 881–890 (2002).
Bloom. in The pharmacological basis of therapeutics (eds Lazo, J. S. & Parker, K. L.) 31, 7-341 (New York, USA. 2006).
Boice, J.D. et al. A cohort study of uranium millers and miners of Grants, New Mexico, 1979-2005. J. Radiol. Prot. 28, 303–325 (2006).
Lestaevel, P. et al. Changes in sleep-wake cycle after chronic exposure to uranium in rats. Neurotoxol. Teratol. 27, 835–840 (2005).
Houpert, P., Lestaevel, P., Bussy, C., Paquet, F. & Gourmelo, P. Enriched but not depleted uranium affects central nervous system in long-term exposed rat. J. Neurotoxicol. 26, 1015–1020 (2005).
Houpert, P. et al. Heterogeneous accumulation of uranium in the brain of rats. Radiat. Prot. Dosimetry. 127, 86–89 (2007).
Kang, J. & Pervaiz, S. Mitochondria: Redox Metabolism and Dysfunction. Biochem. Res. Inter. 2012, 1–14 (2012).
Lwasa, S. & Bwowe, F. Exploring the Economic Potential of Cardamom (Elettaria cardamomum) as an alternative and promising income source for Uganda’s smallholder farmers. ACSS Sci. Conf. Proc. 8, 1317–1321 (2007).
Winarsi, H., Sasongko, N. D., Purwanto, A. & Nuraeni, I. Effect of cardamom leaves extract as antidiabetic, weight lost and hypocholesterolemic to alloxan-induced Sprague Dawley diabetic rats. Int. Food Res. J. 21, 2253–2261 (2014).
Singh, G., Kapoor, I., & Singh, P. Effect of volatile oil and oleoresin of anise on the shelf life of yogurt. J. Food Process. Pres. 35, 47–49 (2011).
González-Trujano, M. F. et al. Evaluation of the antinociceptive effect of Rosmarinus officinalis L. using three different experimental models in rodents. J. Ethnopharmacol. 111, 476–482 (2007).
Gilani, A. H., Jabeen, Q., Ghayur, M. N., Janbaz, K. H. & Akhtar, M. S. Studies on the antihypertensive, antispasmodic, bronchodilator and hepatoprotective activities of the Carum copticum seed extract. J. Ethnopharmacol. 98, 127–135 (2005).
Fleckenstein, A. Specific pharmacology of Ca++ in myocardium cardiac pacemakers and vascular smooth muscle. Rev. Pharmacol. and Toxicol. 17, 149–166 (1977).
Kovacic, P. & Jacintho, J. D. Reproductive toxins, pervasive theme of oxidative stress and electron transfer. Curr. Med. Chem. 8, 863–892 (2001).
Chitra, K. C., Latchoumycandane, C. & Mathur, P. P. Induction of oxidative stress by bisphenol A in the epididymal sperm of rats. Toxicology 14, 119–127 (2003).
Ghosh, S., Kumar, A., Pandey, B. N. & Mishra, K. P. Acute exposure of uranyl nitrate causes lipid peroxidation and histopathological damage in brain and bone of Wistar rat. J. Environ. Pathol. Tox. 26, 255–261 (2007).
Linares, V. et al. Pro-oxidant effects in the brain of rats concurrently exposed to uranium and stress. Toxicology 236, 82–91 (2007).
Ahmed, H. H., Abdel-Rahman, M., Ali, R. A. & Abdel Moniem, A. Protective effect of Ginkgo Biloba Extract and Pumpkin Seed Oil Against Neurotoxicity of Rotenone in Adult Male Rats. J. Appl. Sci. Res. 5, 622–635 (2002).
Manchester, L. C. et al. High levels of melatonin in the seeds of edible plants. Possible function in germ tissue protection. Life Sci. 67, 23–29 (2000).
Toklu, H., Deniz, M., Yüksel, M., Keyer-Uysal, M. & Şener, G. The protective effect of melatonin and amlodipine against cerebral ischemia/reperfusion-induced oxidative brain injury in rats. Marmara Med. J. 22, 034–044 (2009).
Ozturk, M., Aydoğmus-Ozturk, F., Duru, M. E. & Topcu, G. Antioxidant activity of stem and root extracts of Rhubarb (Rheum ribes): An edible medicinal plant. Food Chem. 103, 623–630 (2008).
Ak, T. & Gulcin, I. Antioxidant and radical scavenging properties of curcumin. Chem. Biol. Int. 174, 27–37 (2008).
Chen, X. & Ahn, D. U. Antioxidant activities of six natural phenolics against lipid oxidation induced by Fe+ or ultraviolet light. J. Am. Oil Chem. Soc. 75, 1717–1721 (1998).
Kunwar, T., Kumar, N. & Kothiyal, P. Effect of Elettaria cardamomum hydroethanolic extract on learning and memory in Scopolamine induced amnesia. World J. Pharmaceut. Sci. 3, 75–85 (2015).
Litwinienko, G. & Ingold, K. Solvent effects on the rates and mechanisms of reaction of phenols with free radicals. Acc. Chem. Res. 40, 222 (2007).
Schmidt, T. J., Muller, E. & Fronczek, F. R. New allocedrane type sesquiterpene hemiketals and further sesquiterpene lactones from fruits of Illicium floridanum. J. Nat. Prod. 64, 411–414 (2001).
Hossain, M. B., Brunton, N. P., Barry-Ryan, C., Martin- Diana, A. B. & Wilkinson, M. Antioxidant activity of spice extracts and phenolics in parisom to synthetic antioxidant Rasayan. J. Chem. 4, 751–756 (2008).
Betarbet, R., Sherer, T. B., MacKenzie, G., Garcia-Osuna, M., Panov, A. V. & Greenamyre, T. J. Chronic systemic pesticide exposure produces feature of Parkinson’s disease. Nat. Neurosci. 3, 1301–1306 (2000).
Domingo, J. L., Llobet, J. M., Tomas, J. M. & Corbella, J. Acute toxicity of uranium in rats and mice. Bull. Environ. Contam. Toxicol. 39, 168–174 (1987).
Alhaider, A. A., Al-Mofelh, I. A., Mossa, J. S., Al-Sohaibani, M. O., Qureshi, S. & Rafatullah, S. Pharmacological and safety evaluation studies on Elettaria cardamomum: An important ingredient of gahwa (Arabian coffee). Arb. J. Pharmacol. Sci. 47-58, 47–58 (2005).
Glowinski, L. J. & Iversen, L. L. Regonal studies of catecholamines in the rat brain. I. Disposition of Hanoradrenaline, Ha-dopamine and Ha-dopa in various regions of brain. J. Neurochem. 13, 655–669 (1966).
Yellepeddi, V. in Lippincott Illustrated Reviews: Pharmacology 6th eds (Philadilphia, Baltimora, Newyork. 2015).
FAO “Food Administration Organizationn”. Committee for Inland fisheries of Africa. Report of the third session of the working party on pollution and Hisheries. Accra, Ghana, 25-29 (1992).
Cicik, B. & Engin, K. The effects of cadmium on levels of glucose in serum and glycogen reserves in the liver and muscle tissues of Cyprinus carpio (L., 1758). Turk. J. Vet. Arum. Sci. 29, 113–117 (2005).
Marczenko, Z. in Spectrophotometric determination of elements (Ellis Harwood Ltd, Chicheste, 1976).
Dai, H. et al. Transient binding of an activator BH3 domain to the Bak BH3-binding groove initiates Bak oligomerization. J. Biol. Chem. 289, 89–99 (2014).
Ellman, G. L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 82, 70–77 (1959).
Ohkawa, H., Ohishi, N. & Yagi, K. Assay for lipid peroxidation in animal tissues by thiobarbituric acid reaction. Ann. Biochem. 95, 351–358 (1979).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdel-Rahman, M., Rezk, M.M. & Kader, S.A. The role of cardamom on the hazardous effects of depleted uranium in cerebellum and midbrain of albino rats. Toxicol. Environ. Health Sci. 9, 64–73 (2017). https://doi.org/10.1007/s13530-017-0305-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13530-017-0305-5