Abstract
Autoxidation is a conversion pathway that has the potential to add value to multinuclear aromatic-rich coal liquids, heavy oils and bitumens, which are typically considered low-value liquids. In particular, autoxidation of these heavy materials could lead to products that may have petrochemical values, e.g., lubricity improvers and emulsifiers. Proper assessment of an oxidative transformation to ring-open the multinuclear aromatics present in heavy feeds relies on the understanding of the fundamentals of aromatic oxidation. This work reviews the selective oxidation chemistry of atoms that form part of an aromatic ring structure using oxygen (O2) as oxidant, i.e., the oxidation of aromatic carbons as well as heteroatoms contained in an aromatic ring. Examples of industrially relevant oxidations of aromatic and heterocyclic aromatic hydrocarbons are provided. The requirements to produce oxygenates involving the selective cleavage of the ring C–C bonds, as well as competing non-selective oxidation reactions are discussed. On the other hand, the Clar formalism, i.e., a rule that describes the stability of polycyclic systems, assists the interpretation of the reactivity of multinuclear aromatics towards oxidation. Two aspects are developed. First, since the interaction of oxygen with aromatic hydrocarbons depends on their structure, oxidation chemistries which are fundamentally different are possible, namely, transannular oxygen addition, oxygen addition to a carbon–carbon double bond, or free radical chemistry. Second, hydrogen abstraction is not necessary for the initiation of the oxidation of aromatics compared to that of aliphatics.
Similar content being viewed by others
Introduction
Processes for the oxidation of organic materials cover the entire selectivity spectrum ranging from total or near total combustion to give carbon oxides, to mild partial oxidation to functionalize organic molecules to produce chemical intermediates [1]. Selective oxidation is a direct and economic route to convert petroleum products into petrochemicals [2]. This type of chemistry is present in the large-scale production of commodities, as well as in the synthesis of small amounts of pharmaceuticals and fine chemicals [3]. Selective oxidation of alkanes (paraffins), alkenes (olefins) and aromatics feedstocks produce oxygenated compounds, such as alcohols, aldehydes, ketones, carboxylic acids, anhydrides and epoxides, which are important commodities in the petrochemical industry. The breadth of the industrial application of oxidation can be seen from books that discuss industrially practiced oxidation processes [4, 5].
The scope of this review is much narrower. The review is concerned only with oxidation of atoms that form part of an aromatic ring structure. This is a topic that received relatively little attention in the literature since the report by Tipson [6]. Oxidation of aromatic carbons as well as heteroatoms contained in aromatic rings will be considered.
Heavy aromatic feed materials can in principle be converted into higher value petrochemicals that are also mixtures, for example, lubricity improvers, emulsifiers and greases. Oxidized aromatic compounds may also be low-cost intermediates that can be used as feedstocks for the production of petrochemical products, or that are useful products in their own right. For example, autoxidation (oxidation using air) is commercially employed for asphalt hardening [7], which is an important process for producing asphalt for road construction. Autoxidation is also a conversion pathway by which value can potentially be added to other predominantly aromatic feed materials, such as coal liquids, petroleum vacuum residues, heavy aromatic oils and bitumens. Such complex mixtures are not normally considered for petrochemical production. Petrochemicals are usually produced from feed materials that are purified compounds, or well-defined mixtures of compounds. However, due to the low value associated with heavy aromatic feedstocks, there is an economic incentive to increase their quality if possible.
Oxidative transformation of heavy aromatic oils is likely challenging and one of the objectives of this review is to assess the potential of oxidation as pathway for the conversion of aromatics and in particular multinuclear aromatics.
When selecting an oxidant there are some points to consider: price, ease of handling, nature of the by-products and percentage of oxygen available. Depending on the scale, production options change. Bulk industrial oxidation processes are largely limited to air, but fine chemistry can afford the luxury of using more expensive oxidants, as illustrated by Table 1 adapted from [8]. When converting low-value heavy aromatic oils, air and possibly purified oxygen, are likely the only oxidants that make economic sense. Without proving the point, the review was restricted to O2 as oxidant. It also touches on the catalytic selective oxidation of multinuclear aromatic compounds.
Oxidation of aromatic hydrocarbons
The selective oxidation of aromatic hydrocarbons involves three general types of reactions [9], as illustrated by Fig. 1:
-
(a)
Formation of oxygenates without the cleavage of the aromatic rings, e.g., the formation of phenol or benzoquinone from benzene.
-
(b)
Formation of oxygenates accompanied by the cleavage of the aromatic rings, e.g., the formation of maleic anhydride from benzene.
-
(c)
Formation of oxygenates by oxidation of the side chains attached to the aromatic rings, e.g., the formation of benzaldehyde or benzoic acid from toluene.
Of these reactions, only (a) and (b) are considered in this review, since (c) does not involve the oxidation of an atom that is part of the aromatic ring structure.
Aromatic C–C bonds are strong and substitution reactions, reactions of type (a), are more readily achieved. For example, oxidation to produce quinonoids is well-described [10]. Reactions of type (a) may also lead to addition products that do not necessarily incorporate oxygen; these oxidative aromatic coupling reactions can be both intra- and intermolecular [11].
Even though aromatic oxidation involving splitting of the ring C–C bonds represented a milestone in the heterogeneous catalyzed oxidation technology [12], the literature on the topic is somewhat limited. The literature on ring scission is mostly focused on the discussion of a few examples of industrially successful selective catalytic oxidations, e.g., the commercial production of phthalic anhydride from the vapor-phase oxidation of naphthalene. This type of chemistry is of particular interest for the present work, due to its potential application for the conversion of heavy aromatic materials. As a result, the oxidation of some key aromatic hydrocarbons such as benzene, naphthalene, anthracene and phenanthrene will be discussed in the following sections. There are important differences in the way oxidation of these four compounds proceed.
Benzene oxidation
The vapor-phase oxidation of benzene to produce maleic anhydride, using air as oxidant, is one of the first successful catalytic oxidations of aromatic hydrocarbons. Commercial production of maleic anhydride from benzene started around 1933. Currently, the industrial process for benzene to maleic anhydride autoxidation is only of historical and educational interest [12]. The selective vapor-phase oxidation of n-butane over vanadium phosphorous oxide (VPO) catalysts has been the dominant chemical route to produce maleic anhydride since the mid-1970s [13]. Because of its availability and lower cost, n-butane became an attractive raw material [14].
The industrial routes for phenol production from benzene all employ intermediate products and there are no processes to produce phenol by direct oxidation of benzene [15].
Even though benzene is the simplest aromatic hydrocarbon, its oxidation to maleic anhydride is a complicated chemical reaction, with many side-products. Production of phenol, quinone and hydroquinone from benzene autoxidation has been reported [16,17,18]. The presence of small amounts of other compounds such as formaldehyde, biphenyl, acrylic and formic acid has also been mentioned [17,18,19]. In this case, maleic anhydride is the only product obtained in yields high enough for commercial production. In industrial operation benzene is oxidized with 67–72% of the theoretical yield of maleic anhydride [18].
The mechanism of benzene oxidation has been the subject of considerable research work. Yet, the literature indicates some disagreements regarding the nature of the intermediates. The generally adopted reaction network for benzene oxidation is presented in Fig. 2. This scheme is the basis of most kinetic studies. Benzene oxidizes in two independent ways to produce either maleic anhydride or total combustion products. Both reactions are the result of a number of short-lived intermediates [17]. Further oxidation of maleic anhydride also leads to the production of carbon oxides and water. In the absence of a catalyst, thermal decomposition of benzene is favored. Transition metal oxides have been successfully used to control the selectivity of the reaction and promote the production of maleic anhydride. Industrially, metal oxides are of interest because the temperature window, in which they catalyze the desired oxidation at appreciable rate, is wider than the one offered by most metal catalysts [16].
For benzene oxidation, vanadium, molybdenum and tungsten oxides have proved to be very selective [16, 18]. However, systems based on V2O5 have received special attention. According to the patent literature, the development of active and selective vanadium-based catalysts became an important research topic. A number of different catalyst compositions have been proposed and evaluated. Mixtures of vanadium and molybdenum oxides, in combination with one or more oxides of other elements, e.g., P, W, Co, Fe, Ni or their mixtures, or in combination with salts, e.g., sodium borate, have been tested in the temperature range 340–450 °C [20,21,22].
Industrial catalysts for benzene oxidation are based on mixtures of V2O5 and MoO3. The latter can be found in concentrations up to 30% mol [18]. Typical promoters used are oxides or salts of elements such as W, Bi, Sn, P, Ag, Cu, Na, B, Ti and Ni [23]. The special catalytic behavior of vanadium-based catalysts is related to the valence changes of the metal during reaction [16]. Some interesting discussion on the structure, the redox behavior and the phases formed in the catalyst during reaction as well as the interactions of oxygen and benzene on the catalysts surface can be found in [23, 24].
Mars and Van Krevelen studied the catalytic oxidation of some aromatic hydrocarbons, namely benzene, toluene, naphthalene and anthracene [25]. Their work suggested that the oxidation of these compounds occurred through a redox mechanism (appropriately known as the Mars–Van Krevelen mechanism), which involves two independent steps. First, the reaction between the aromatic hydrocarbon and the metal oxide catalyst (MO n ) takes place (Eq. 1). Second, the partially reduced catalyst (MOn−1) is reoxidized with gas phase oxygen (Eq. 2),
Due to the generality of the kinetics following from this mechanism, it is worthwhile providing a brief description. Assuming that the first step is proportional to the partial pressure of the aromatic hydrocarbon (pAr), i.e., first-order reaction, and to the fraction of catalyst surface covered with active oxygen (θ), the reaction rate can be expressed according to Eq. 3. On the other hand, if the rate of reoxidation of the catalyst is assumed to be proportional to a certain power (m) of oxygen partial pressure and to the catalyst surface area not covered with active oxygen (1 − θ), the reaction rate for the second step can be expressed according to Eq. 4,
In steady state, the reaction rates of these two steps (reduction–oxidation cycle) are equal. Also, if for one aromatic molecule, β molecules of oxygen are required, it can be written as
Combining Eq. 3 and 6, the overall reaction rate can be expressed as
The validity of the kinetic expression (Eq. 7) with m = 1 for the oxidation of benzene was confirmed by Mars and Van Krevelen [25]. Furthermore, this type of equation was also valid for the oxidation of naphthalene and anthracene; the value of the rate constant k2 of the reoxidation process on a V2O5–Mo3O/Al2O3 catalyst was found to be the same for the catalytic oxidation of benzene, naphthalene and anthracene.
The reaction kinetics of benzene oxidation over a number of vanadium-based catalysts have been reported. A valuable compilation of kinetic studies for benzene oxidation can be found in [17, 24]. Results from selected studies are presented in Table 2. At high concentrations of benzene, reactions (1) and (3) in Fig. 2 appear to be of pseudo-first order on benzene [23]. The effect of the mass transfer rate on the pseudo-first-order rate constants has been discussed [17]. The dependence of reaction (2) on the concentration of maleic anhydride is less clear [23]. It has been accepted that kinetic models derived from the reaction scheme presented in Fig. 2 are a good approximation when the concentration of benzene is high and the formation of side-products, such as phenol and quinone, can be neglected. More comprehensive studies have taken into account the oxidation of benzene as well as the oxidation of maleic anhydride, quinone, and phenol [18].
Naphthalene oxidation
The oxidation of naphthalene to phthalic anhydride is commercially one of the most important vapor-phase catalytic oxidations of aromatic compounds. The first commercial process started operation in 1916.
Industrially, naphthalene oxidation is presently carried out in fixed-bed reactors operating in the temperature range 340–380 °C and using supported vanadium oxide catalysts [26, 27]. Current processes are flexible and allow for using dual feedstocks, i.e., mixtures of naphthalene and o-xylene [26]. In recent years, the use of o-xylene as the main raw material has spread due to its lower cost, good availability and ease of transportation [28].
Compared to benzene, the presence of a second aromatic ring in the naphthalene molecule increases the complexity of the reaction network. Several parallel and consecutive reactions take place in this case. Even though naphthoquinone is a key intermediate in the reaction sequence, phthalic anhydride is not formed exclusively through this compound. Production of maleic anhydride and carbon oxides as secondary products has been reported [28].
Figure 3 presents the generally adopted reaction scheme. Naphthalene is oxidized by three independent routes. Production of phthalic anhydride and 1,4-naphthoquinone represents the most important primary reactions [17]. The distribution of these products may vary according to the initial point of attack in the naphthalene molecule [17]. It has been stated that for the sake of completeness, 1,2-naphthoquinone should be indicated as an intermediate in reaction (1); however, this compound is usually neglected because of its rapid oxidation to phthalic anhydride [27]. Naphthalene is also directly oxidized, to a much lesser extent, to maleic anhydride and carbon oxides [17, 27]. Secondary reactions are also important in this network. Typically, a large fraction of the 1,4-naphthoquinone formed is further oxidized to give phthalic anhydride, while a small portion contributes to the production of maleic anhydride and carbon oxides. Similarly, part of the phthalic anhydride formed by reactions (1) and (3) is oxidized to give maleic anhydride and carbon oxides. Production of aromatic acids, i.e., phthalic and benzoic acids has also been reported [16]. For reactor design purposes, the kinetic scheme of naphthalene oxidation may be simplified to reactions (1)–(4) [27], as discussed later on in this section.
It is believed that the stepwise oxidation of the naphthalene ring to form phthalic anhydride proceeds by a similar mechanism to that of benzene oxidation to form maleic anhydride. The formation of the 1,4-naphthoquinone (α-quinone), which is the equivalent of the quinone in benzene oxidation, supports this premise [16]. The formation of the 1,4-naphthoquinone also points out the difficulty of breaking the aromatic structure. The oxidation of the naphthalene molecule may be analyzed in three steps: addition of oxygen into the structure, breaking of one of the two rings in naphthalene and further destruction of the remaining benzene ring [16].
At ambient conditions, naphthalene is inert to air (oxygen) and cannot be partially oxidized. Therefore, the use of higher temperatures is required. In the absence of a catalyst, two scenarios are possible [16]. First, oxidation at high temperature (i.e., at a temperature high enough for the reaction rate to be significant) favors total combustion over partial oxidation. Second, oxidation at moderate conditions promotes addition reactions (condensation reactions) and polymerization of the intermediates due to the longer reaction times needed to achieve significant conversion. The use of an appropriate catalyst is essential to control the reaction selectivity.
Transition metal oxides of the fifth and sixth groups of the periodic table have been the candidates of choice. Vanadium and molybdenum oxides as well as tin and bismuth vanadates have been tested as catalysts for this reaction [16, 17]. Oxides of magnesium, aluminum, silicon, titanium, zirconium, copper and cobalt have shown rather poor performance in naphthalene oxidation [16]. Catalysts based on vanadium oxides are quite active and can cause over-oxidation to maleic anhydride or carbon oxides. Strict control of the reaction residence time or addition of less active oxides to the catalyst (e.g., manganese, copper or cobalt oxides) is common practices to avoid this problem [16]. For vanadium-based catalysts, yields to phthalic anhydride of more than 70% have been reported [17]. Similarly, selectivities towards phthalic anhydride of nearly 90% have been achieved using mixtures of V2O5 and K2SO4 supported on SiO2, i.e., commercial type of catalysts [29]. A great deal of information on the catalysts used for naphthalene oxidation is found in the patents while only some is found in the journal literature. There is no recent review dealing with commercially used catalysts for naphthalene oxidation. A valuable discussion on the parameters affecting the catalytic performance of vanadium-based catalysts (e.g., V2O5 concentration, active sites, role of alkali metal sulfates, among others) can be found in [28]. In addition, a compilation of the catalysts and conditions used in naphthalene oxidation can be found in [17].
The reaction mechanism and kinetics of naphthalene oxidation have been extensively studied. Yet, they are not completely elucidated. As mentioned in the previous section on benzene oxidation, it has been found that naphthalene oxidation also proceeds through a redox mechanism [25]. In other words, the aromatic hydrocarbon reacts with a metal oxide catalyst to give the oxidized products and a partially reduced catalyst; then, the catalyst is reoxidized by gas phase oxygen. Hence, the kinetic expression derived before, Eq. 7, can be used to describe this reaction too. At constant oxygen partial pressure, Eq. 7 can be rearranged to Eq. 8.
The latter expression shows that at low partial pressures of naphthalene (pN), the oxidation rate should approach first order in naphthalene concentration; whereas at high partial pressures of naphthalene, it should approach zero order in this reactant [17]. A number of kinetic studies using vanadium-based catalysts, namely V2O5 and K2S2O7 or K2SO4 supported on SiO2, are in agreement with this behavior. Mars and Van Krevelen investigated the effect of naphthalene and oxygen pressure on the rate of naphthalene oxidation [25]. According to their work, the rate is approximately first-order with respect to naphthalene at pN < 0.5 kPa, but it is almost independent of the naphthalene concentration at pN > 1 kPa. A similar trend was observed by Calderbank [30], who studied the reaction over the range of naphthalene concentrations of 2–10 kPa, and by Ioffe and Sherman [31], who explored the pressure range 0.7–1.5 kPa. On the other hand, the dependence of reaction rate on the concentration of oxygen was found to vary almost linearly with the O2 concentration in the region of oxygen partial pressures below 40 kPa [25].
Valuable compilations of kinetic studies for naphthalene oxidation were prepared by Dixon and Longfield [17] and Wainwright and Foster [28]. In general, reactions (1)–(4) in Fig. 3 appear to depend on the concentration of both oxygen and the organic reactant to an order between 0.5 and 1.0 [17]. The observed differences in individual studies are attributed to the variation in the reaction conditions rather than a real effect. There is little consistency in the reaction temperature, catalyst, reactor type and conversion achieved in the reported studies; therefore, comparisons are not easy [28].
Anthracene and phenanthrene oxidation
The catalytic vapor-phase autoxidation of anthracene to anthraquinone with yields of 85–90% has been described in patents in the 1930s and has found some industrial applications [2, 17]. The vapor-phase oxidation of anthracene with air is the preferred synthesis method to produce 9,10-anthraquinone [32], and > 90% yield is possible. However, the most widespread use of oxidation in relation to anthracene is the use of the autoxidation-and-reduction cycle of 9,10-dihydroxyanthracene and anthraquinone to produce hydrogen peroxide [2], as illustrated by Fig. 4.
Vapor-phase catalytic oxidations of anthracene and phenanthrene have been less extensively investigated compared to benzene and naphthalene oxidation. The additional aromatic ring further increases the complexity of the reaction network. Thus, depending on the position of attack and the extent of the reaction, oxidation products from the inner and outer rings are possible. As in other aromatic oxidations, achieving selectivity is a challenge and using an appropriate catalyst to promote the production of the desired products is necessary.
Almost complete conversion of anthracene with high selectivity to 9,10-anthraquinone is possible and 9,10-anthraquinone of 99% purity is industrially produced in this way [32]. Secondary and complete combustion products are also obtained is small amounts. Carbons in the 9- and 10-positions are the most reactive centers of attack in the anthracene molecule [6]. The attack of oxygen on these carbons results in the production of 9,10-anthraquinone. On the other hand, the unselective oxidation of the carbons in the 1- to 4-positions, i.e., carbons in the outer ring, gives ketone (1,4-anthraquinone) and anhydride (2,3-naphthalic anhydride and pyromellitic anhydride) species as by-products [33]. The production of small quantities of phthalic anhydride, maleic anhydride, CO and CO2 has also been reported [16, 33, 34]. The higher selectivity of the oxidation reaction can be explained in terms of the Clar formalism [35], noting that in anthracene additional stabilization of the outermost rings can be achieved if the π-electrons on the 9,10-positions are more localized.
The overall reaction for vapor-phase anthracene oxidation is shown in Fig. 5. The oxidation process involves the interaction of one hydrocarbon molecule with three oxygen atoms. However, the actual reaction mechanism is more complex than that. With the localization of the π-electrons on the 9,10-positions, not only does the transannular addition of oxygen take place, but two aromatic sextets are also formed. Even more, the removal of hydrogen from an aromatic carbon is not required for oxidation to take place. The addition of oxygen causes the carbons at the 9,10-positions to change from sp2 to sp3 hybridized carbons. In other words, these carbons are now benzylic aliphatic carbons with a much weaker C–H bond, which facilitates hydrogen abstraction by further reaction with oxygen.
It has been suggested that the catalytic conversion takes place in two steps, which are in agreement with the Mars–Van Krevelen mechanism [17, 25]. First, anthracene is oxidized by a metal oxide catalyst producing anthraquinone and water; during this step the oxide catalyst is reduced. Second, the catalyst is reoxidized by gas phase oxygen to return to its original state.
Vanadium oxide, iron vanadate or vanadic acid doped with alkali or alkaline earth metal ions are suitable catalysts for anthracene oxidation [16, 33, 34]. Industrially, the reaction has been carried out at temperatures in the range 320–390 °C and using catalysts consisting of V2O5, K2SO4 and Fe2O3 on pumice; yields of 95–97 wt% of anthraquinone were reported [17]. A reaction scheme derived from the oxidation of anthracene using V2O5–MoO3–P2O5 and V2O5–Fe2O3 as catalysts can be found [33]. In this case, oxidation at the inner and outer rings of anthracene leads to 9,10-anthraquinone, 1,4-anthraquinone and 2,3-naphthalic anhydride. At high conversion, the selectivities towards these compounds decreases and production of phthalic anhydride, pyromellitic anhydride and CO2 is favored.
The liquid phase oxidation of anthracene and 9,10-anthraquinone has been investigated by thermal analysis [36]. In this study, a number of metal oxides (V2O5, MoO3, Fe2O3 and NiO) were evaluated as catalysts. Among them, V2O5 showed the highest catalytic contribution during reaction; it measurably accelerated the autoxidation of both aromatic compounds. Experiments to determine whether this oxide just activated the O2 from air, or whether oxidation took place by transfer of lattice oxygen were also performed. Interestingly, reaction under nitrogen atmosphere confirmed that V2O5 was capable of using the lattice oxygen for oxidation of anthracene and 9,10-anthraquinone in the liquid phase. A Mars–Van Krevelen mechanism was likely.
Phenanthrene is used for industrial production of 9,10-phenanthrene quinone as well as diphenic acid by vapor-phase oxidation over vanadium-based catalysts [33]. Same as anthracene, the 9 and 10 carbons in the phenanthrene molecule are much more reactive than the other ones, and so the 9- and 10-positions are the preferred centers of attack [6]. As in the case of anthracene, this is explained by the Clar formalism [35]. Selective oxidation on these carbons gives the 9,10-dione; whereas further reaction produces anhydride (diphenic anhydride), lactone (2-hydroxy-diphenyl-2′-carboxylic acid lactone) and ketone (9-fluorenone) species as side-products [33]. When oxygen attacks the carbons on the outer rings (not preferred pathway), 1,2-naphthalic anhydride is produced [33]. On the other hand, a number of experimental studies taking place in the temperature range 370–440 °C and using vanadium pentoxide, mixtures of vanadium and molybdenum or iron oxides and tin vanadates as oxidation catalysts resulted in the production of phthalic anhydride and phthalic acid [17].
The reaction network for phenanthrene oxidation is presented in Fig. 6. The scheme was derived from the study of the reaction over V2O5 supported on SiO2 with and without K2SO4 [37]. Phenanthrene is oxidized by two independent routes, which are governed by the position of attack, i.e., oxidation in the inner or outer rings.
Compared to the benzene or naphthalene cases, there are a lot fewer publications dealing with the kinetics of the vapor-phase anthracene and phenanthrene oxidation. Mars and Van Krevelen found that at anthracene partial pressures > 0.1 kPa, the reaction rate was essentially independent with respect to the concentration of the aromatic reactant [25]. In addition, the reaction rate showed some dependence on oxygen over the entire concentration range (oxygen partial pressures up to 100 kPa). Pyatnitskii derived a kinetic expression for aromatic oxidations [38] based on the work of Mars and Van Krevelen. In his work, the two independent steps of the redox mechanism (Eqs. 1 and 2) were written as
where a and b are the orders with respect to adsorbed oxygen and to oxygen vacancies on the catalyst surface, respectively. These equations reduce to Eqs. 3 and 4 when a = 1 and b = 1. In steady state,
where the number of oxygen molecules required to oxidize one aromatic molecule (Ar) to the products P1, P2, …, P i , is represented by the stoichiometric coefficient (v) as follows:
where S i is the selectivity for P i and v i corresponds to the number of oxygen molecules necessary to oxidize one aromatic molecule to P i . By replacing Eqs. 10 and 11 in Eq. 5, it can be seen that the surface concentration of oxygen is a function of the ratio of the partial pressures of oxygen and the aromatic hydrocarbon (\(P_{{O_{2} }} /P_{\text{Ar}}\)):
For the simplest case a = b = 1, and combining Eqs. 11 and 14, the total rate of oxidation can be expressed as
The kinetic expression (Eq. 15) derived in Pyatnitskii’s work describes the oxidation of anthracene on V2O5–K2SO4 with Fe2O3 and SiO2, and on CoMoO4/SiO2 [38]. On the other hand, the kinetics of the oxidation of phenanthrene are best described with empirical power law equations [38]. It is worth mentioning that the rate of oxidation of phenanthrene is less than that of anthracene over the same V2O5–K2SO4/SiO2 catalyst.
Reactor engineering in aromatic oxidation
Vapor-phase catalytic aromatic oxidations are strongly exothermic reactions. Furthermore, the heat released is always greater than the calculated heat of reaction of the main reaction, due to side reactions leading to the formation of CO2. For instance, while the heat of oxidation of naphthalene to phthalic anhydride corresponds to 1880 kJ/mol, in practice, due to over-oxidation the total heat liberated can reach 2900 kJ/mol [39].
Aromatic oxidation requires large amounts of heat to be removed from the reaction zone to maintain the temperature and keep the overall process under control. An uncontrolled reaction would cause a severe reduction of yields (complete combustion would be favored) as well as the loss of catalyst life [23]. Furthermore, it is a self-amplifying problem, i.e., an increase in temperature causes more combustion reactions that would further increase the temperature. Proper reactor design and heat management are therefore key aspects in industrial processes. Reaction at near-isothermal conditions is preferred to maintain optimal catalyst efficiency and product selectivity. Multitubular reactors and fluidized beds with heat exchange internals have been widely used for this purpose; after all, these configurations are suitable to minimize the temperature gradients across the catalyst bed.
Most of the processes for industrial production of maleic anhydride from benzene use multitubular reactors. A typical reactor contains a bundle of 10,000–15,000 vertical tubes, ~ 25 mm in diameter, enclosed by a jacket through which cooling medium circulates [23]. During operation, the reaction gas flows through the tubes and over the catalyst, while the process pressure is kept between 0.15 and 0.25 MPa [19]. Circulating heat-transfer fluids or agitated eutectic salt mixtures are commonly used to deal with the large duty [23]. Steam, which can be used in other operations, is generated from the heat removed. Nearly 27 MJ of heat are dissipated per each tonne of reacted benzene, which translates to the production of almost 10 tonnes of saturated steam per tonne of reacted benzene [19]. Considering that the reaction is conducted at temperatures > 300 °C, production of high-pressure steam, typically 4–5 MPa steam, is possible.
A typical flow diagram of a process using a multitubular reactor for benzene oxidation [23] is presented in Fig. 7. In this configuration, a mixture of benzene and preheated air is reacted in the multitubular reactor. The conversion of benzene is not complete. The feed-product heat exchange decreases the duty of the feed preheating. On the other hand, switch condensers allow for recovering close to 90% of the maleic anhydride produced in the reaction. Refining of this product is completed later on by batch distillation. The remaining 10% of maleic anhydride is recovered in a two-stage scrubber unit using water and dilute alkali. Fumaric acid is also obtained in this process. The unconverted benzene is separated from the exhaust air by adsorption with activated carbon, and it is returned as part of the reactor feed.
Control of the reaction temperature during catalytic aromatic oxidation can also be achieved using fluidized bed reactors [23]. Some commercial plants employ this technology to oxidize naphthalene to phthalic anhydride. Thus, during typical operation, liquid naphthalene is fed at the bottom of the catalyst bed; after instant evaporation, it distributes over the entire fluidized bed. Oxygen fed through a distributor plate is mixed with the vaporized naphthalene. The reaction temperature is kept in the range 345–385 °C through heat exchanger tubes in the fluidized bed. Moreover, a uniform temperature profile is obtained because of the intense agitation and mixing of the catalyst in the fluidized bed. High-pressure steam (> 4 MPa) is produced from the heat removed [40].
The increase in size for aromatic hydrocarbons, which is represented by the number of aromatic rings in a molecule, has an effect on the calculated (theoretical) heat of reaction. According to Table 3, the heat liberated during oxidation to the main oxidation product decreases in the order benzene > naphthalene > anthracene. As the molecules become heavier, fewer oxygen atoms react per mole of aromatic substrate to produce the main product, and so considerably less heat is released. Irrespective of the aromatic feed, successful selective oxidation requires proper heat management.
Oxidation of heterocyclic aromatics
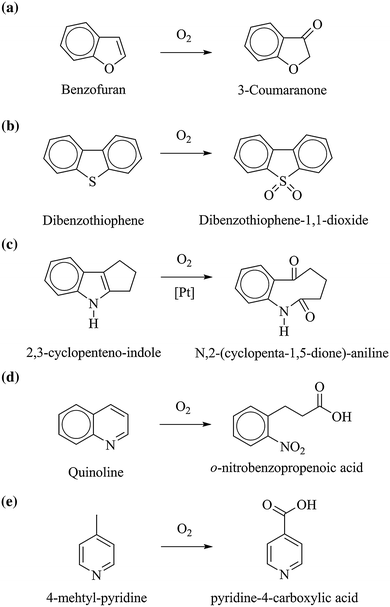
The selective oxidation of heterocyclic aromatics involves the same types of reaction as noted for the aromatic hydrocarbons [9], but added differentiation is required to distinguish between carbon oxidation and heteroatom oxidation. The general types of reactions are (Fig. 8):
Examples of heteroaromatic oxidation involving a carbon oxidation without the cleavage of the aromatic rings, b heteroatom oxidation without the cleavage of the aromatic rings, c cleavage of carbon–carbon bonds of the aromatic rings, d cleavage of carbon–heteroatom bonds of the heterocyclic aromatic rings, e oxidation of the side chains attached to the aromatic rings
-
(a)
Formation of oxygenates by carbon oxidation without the cleavage of the aromatic rings, e.g., formation of coumaranone from benzofuran and the polymerization of pyrrole.
-
(b)
Formation of oxygenates by heteroatom oxidation without the cleavage of the aromatic rings, e.g., formation of benzothiophene-1,1-dioxide from benzothiophene.
-
(c)
Formation of oxygenates accompanied by the cleavage of carbon–carbon bonds of the aromatic rings, e.g., formation of N,2-(cyclopenta-1,5-dione)-aniline from 2,3-cyclopenteno-indole [41]. However, it should be noted that oxidative carbon–carbon scission in heterocyclic aromatic rings appears to be rare.
-
(d)
Formation of oxygenates accompanied by the cleavage of carbon-heteroatom bonds of the heterocyclic aromatic rings, e.g., o-nitrobenzo-propenoic acid from quinoline.
-
(e)
Formation of oxygenates by oxidation of the side chains attached to the aromatic rings.
-
(f)
e.g., oxidation of 4-methyl-pyridine to pyridine-4-carboxylic acid.
Of these reaction types, only (a)–(d) will be considered. It is also possible for oxidation of the multinuclear aromatics to take place on a carbocyclic ring that does not contain a heteroatom, as was discussed in the previous section. One variation on reaction types (a) and (b) is reactions leading to the formation of addition products. Oxidative addition does not necessarily lead to oxygen incorporation into the product.
Even though selective oxidation is a direct and economic route to convert aromatic hydrocarbons into valuable chemicals, the scenario for heterocyclic aromatics is a little different. Oxidation with air of these compounds as a synthesis route to obtain added value products is not reported in standard texts [42, 43]. Their oxidation chemistry is rather discussed as part of the crude oil processing and fuel stability. For instance, sulfur- and nitrogen-containing compounds are typically undesired materials in crude oil. The oxidation of these compounds (i.e., oxidative desulfurization and oxidative denitrogenation) to give chemically modified products with properties that favor their separation or removal has been explored [44, 45]. Furthermore, it is known that autoxidation of nitrogen heterocyclic aromatics plays a role in the formation of heavy addition products, e.g., nitrogen-containing compounds seem to be related with the formation of sludge in fuels [46], thereby affecting fuel storage stability.
The oxidation of the distinct aromatic heterocyclic compound classes is considered in the following sections. Emphasis is placed on the chemistry of five-membered heterocyclic compounds that contain sulfur, nitrogen, and oxygen, as well as on their benzologs. Some comments regarding the chemistry of pyridine, a nitrogen-containing six-membered heterocyclic compound, are also included as part of the discussion.
Oxidation of aromatic S-heterocyclic compounds
The first class of aromatic heterocyclic compounds considered in this work corresponds to the sulfur-containing compounds, which are represented by thiophene and its benzologs, e.g., benzothiophene and dibenzothiophene (Fig. 9). These molecules are typically found in the heavier fractions of crude oil [47]. Refining of crude oil to final products, such as fuels and some petrochemicals, requires desulfurization of the oil. However, the sulfur contained in aromatic rings is more difficult to remove than aliphatic sulfur using traditional technologies, e.g., hydrodesulfurization (HDS) and thermal conversion [47].
Oxidation of thiophene and its benzologs as an alternative approach to HDS for sulfur removal has received a lot of attention. Oxidative desulfurization (ODS) involves two separate steps. First, the reaction between an oxidant and sulfur alters the nature of the sulfur-containing compounds. Second, the properties of the oxidized products are exploited to facilitate their removal through a separation process [44]. Oxidation with air has been extensively evaluated.
Autoxidation of S-heterocyclic aromatics involves the formation of hydroperoxides by oxidation of aliphatic compounds that are already present or are added to the reaction mixture. Hydroperoxides are key intermediates formed in situ by the oxygen. The thiophenic sulfur is subsequently oxidized by the hydroperoxides, rather than by oxygen directly (free radical chemistry). The chemistry taking place during the first step of this conversion illustrates the oxidative behavior of S-heterocyclic compounds in the presence of air. The thiophenic sulfur is oxidized to sulfoxides and sulfones (Fig. 10).
Moreover, thiophenes that could be considered refractive to HDS, are readily oxidized because of their increased electron density on the sulfur atom [48]. Temperatures below 200 °C and pressures near atmospheric pressure are usual for oxidation of heterocyclic sulfur. Typical conditions for oxidative desulfurization processes are given in Table 4.
Autoxidation of S-heterocyclic aromatics can also be performed using a catalyst or oxygen carrier, instead of employing free radical autoxidation. Catalysts facilitate the decomposition of hydroperoxides, and in doing so, accelerate the propagation step during autoxidation. Oxygen carriers, on the other hand, are more active oxidation agents than oxygen. Depending on their nature, they can be viewed as a type of catalyst which can be regenerated using molecular oxygen, i.e., oxidation by the Mars–Van Krevelen mechanism; this characteristic makes their behavior similar to the one observed for metal oxides in the selective oxidation of aromatic compounds, as discussed in the previous section. Additional details on the topic can be found in [54].
The C–S bond strength is decreased when the sulfur is oxidized, which in principle allow the cleavage of the C–S bonds by catalytic [55], or by thermal decomposition. These assertions are based on the published homolytic bond dissociation energies for sulfones. In the case of a diaryl sulfone, which is representative of the sulfone group in dibenzothiophene-1,1-dioxide, the bond dissociation energy is reportedly 288 kJ/mol [56]. However, this value was found to be inconsistent with the thermal stability of diaryl sulfones that suggested a much stronger bond [57].
Autoxidation of sulfur-containing compounds can also lead to the formation of addition products. For instance, model oil consisting of n-heptane and dibenzothiophene reacted with air at 145–170 °C and atmospheric pressure resulted in the formation of some heavier products [58]. In this case, besides the typical products involving the formation of both sulfoxides (dibenzothiophene-1-oxide) and sulfones (dibenzothiophene-1,1-dioxide), the formation of sediments was observed. This precipitation process involves a type of chemistry analogous to that explaining the gum formation, a common problem that undermines storage stability of transportation fuels. In the proposed reaction pathway, a sulfoxide reacts with a hydroperoxide to give an intermediate product that may be decomposed by reaction with another sulfoxide to yield an addition product [58].
The thiophene ring system, unless carrying electron-donating substituents, is relatively stable to atmospheric oxidation [59], and resist the action of moderate oxidizing agents [42, 43]. Oxygen and peracids are known to preferentially attack the sulfur atom [43], while stronger oxidants are required to actually attack the aromatic carbons. Ozone, for example, attacks the double bond between the aromatic carbons which results in the cleavage of the ring; thus, oxidation of benzothiophene with ozone yields o-mercaptobenzaldehydae [59]. Similarly, strong oxidants, e.g., nitric acid, are able to break down the aromatic ring to give maleic and oxalic acids, while the ring sulfur is oxidized to sulfuric acid [59].
It is evident that oxidation chemistry of sulfur-containing compounds, using O2 as oxidant, is simpler that the one observed for aromatic compounds, due to the high selectivity of autoxidation to oxidize the heterocyclic sulfur. In this case, there are no complex reaction networks. The formation of oxygenates by oxidation of the heteroatom is actually the main oxidative transformation. Even for addition reactions to take place, oxidation of the sulfur plays a role and autoxidation of the aromatic carbons is not observed except at extreme (combustion) conditions.
Oxidation of aromatic N-heterocyclic compounds
The second class of aromatic heterocyclic compounds examined in this work corresponds to the nitrogen-containing compounds. Examples of N-heterocyclic compounds are pyrrole, indole and pyridine (Fig. 9). Aromatic nitrogen-containing compounds present in crude oil are normally classified in two categories: basic (derivatives of pyridine) and non-basic (derivatives of pyrrole). Irrespective of the category, these compounds are associated with a number of different problems during oil processing and their removal is desirable [45].
Oxidation of nitrogen-containing compounds has been explored as an alternative approach to traditional practices such as hydrodenitrogenation (HDN) by hydrotreating. Even though the fundamental idea behind this process, i.e., oxidative denitrogenation, is very similar to those of oxidative desulfurization (the aim is to produce chemically modified products that can be more efficiently extracted from oils using polar solvents), very little has been reported on the topic [45].
Few studies dealing with the oxidation of industrial feedstocks have been reported, and in those cases the oxidation of the S- and N-containing compounds has been performed simultaneously [60, 61]. The purpose of oxidative denitrogenation is mainly to remove basic nitrogen-containing compounds. Typical reaction conditions involve temperatures around 70 °C and atmospheric pressure. Peracetic acid or mixtures of hydrogen peroxide and acetic acid have been used as oxidizing agents to convert pyridines into N oxides. While sulfones and sulfoxides are easily obtained during oxidative desulfurization, products of polymeric nature are obtained after the oxidation of N-heterocyclic aromatics [60]. It will be shown that this is due to the oxidation of pyrroles and not due to the oxidation of pyridines.
Because the oxidation chemistry of pyrroles and pyridines is quite different, some comments for each class of compound will be made in the following sub-sections in a separate manner.
Oxidation of pyrroles
Simple pyrroles are easily oxidized. Compounds containing a pyrrole ring, such as indoles and carbazoles, have a negative effect on the oxidative stability of jet fuels [62]. The effect of pyrroles on color and sediment formation in fuels has been documented and explained through a number of oxidation and polymerization reactions [63]. These are all industrially important fouling reactions.
However, the nature of the products depends on the strength of the oxidizing agent. Strong oxidants, such as chromium trioxide in aqueous sulfuric acid, often lead to complex breakdown products. When the ring survives, maleimide derivatives are obtained [42]. Milder oxidants such as hydrogen peroxide convert pyrroles to pyrrolines [59]. In the case of oxygen, which is the oxidant of interest in this work, pyrrolic compounds autoxidize to form dark colored pigments known as “pyrrole black” [64, 65]. In oil fractions exposed to air, pyrroles autoxidize to form the so-called “red tars” [43, 64]. Irrespective of the name, pyrrole black or red tar, these are all mixtures of pyrrole addition products. Oxidative addition of pyrroles is due to aromatic carbon oxidation [64], a variation on the reaction type (a) shown in Fig. 8.
The autoxidation of 2,5-dimethylpyrrole is presented in Fig. 11 to illustrate how a typical reaction of a pyrrolic compound with air takes place. The first step corresponds to the formation of a molecular association complex (I) between the oxygen and the pyrrolic compound. Then, the electron transfer from the pyrrole nucleus of the complex to the oxygen produces a charge-transfer complex (II), which is suggested to be in equilibrium with an endoperoxide (III) [65]. Alkylated pyrroles and pyrroles having electron-donating substituents are more susceptible to oxidation than those having electron-withdrawing substituents [64]. Pyrroles that have a ketone or ester substituent are also more resistant to ring degradation and yield side-chain oxidation products [42].
The oxidation chemistry of pyrrole and the corresponding benzopyrrole (indole) is quite similar. The presence of the benzene ring changes the preferred position of attack for oxidants from carbon in the 2- and 5-positions in the former to carbon in the 3-position in the latter [59]; however, it does not suppress the susceptibility of the pyrrole ring to give addition products [64]. The carbon adjacent to the nitrogen atom in a pyrrole ring is readily oxidized, which ultimately leads to oxidative coupling and formation of polymeric or addition products [64].
Depending on the conditions of the reaction, indole addition may or may not involve the incorporation of oxygen. In the presence of air and light, indole is autoxidized to give indoxyl which further reacts to produce indigo [43]. Indigo is a molecule containing two oxygen atoms and it is the reaction product from two indole molecules. The nature of the reaction products found after the catalytic oxidation of indole using Mn(II)porphyrins as catalyst, suggested that the mechanism of indole addition involves both oxygen incorporation and hydrogen disproportionation [66]. On the other hand, selectivity close to 90% towards oxidative addition product formation was found during the low temperature air oxidation of indole [67]. In this case, no evidence for oxygen incorporation in the products was found suggesting that the addition mechanism did not require oxygen incorporation even though oxygen was necessary for the reaction to proceed.
Oxidation of pyridines
The lone pair of electrons on the nitrogen atom that is available for bonding, without disturbing the aromaticity of the ring, makes pyridine chemistry quite distinct from that of pyrrole [67, 68]. Pyridines are therefore bases. Because the pyridine ring is π-electron deficient and oxidants act as electron acceptors, the oxidation of the ring is difficult [43]. In fact, strong oxidants, e.g., neutral aqueous potassium permanganate, and vigorous conditions are required for its breakdown. Side chains may be oxidized to the corresponding carboxylic acid group without breaking the ring [42]. The nitrogen atom, on the other hand, is a center of high electron density and as such can be easily oxidized to give pyridine N-oxide [43]. Alkaline hydrogen peroxide and various peracids have been used with this purpose [69].
It has been reported that quinoline is not autoxidized under mild reaction conditions (oxidation with air at 130 °C for 6 h) [67]. Oxygen, hydrogen peroxide and hydroperoxides in the absence of carboxylic acids are incapable of oxidizing the pyridinic nitrogen. The acid–base interaction between the peracid and the pyridine is consequently important for the reaction to proceed.
Oxidation of aromatic O-heterocyclic compounds
The last class of aromatic heterocyclic compounds considered in this work, corresponds to the oxygen-containing compounds, for example, furan and benzofuran (Fig. 9).
Of the mononuclear five-membered heterocyclic compounds, i.e., thiophene, pyrrole and furan, furan has the least aromatic character [68]. As a result, its chemistry is, to some extent, different from that described for thiophene and pyrrole. Furan is particularly sensitive to oxidation. In the presence of oxygen or air, it is unstable [43]. The autoxidation of furan involves the 1,4-addition of oxygen to the diene system to give the transannular peroxide. Further reaction opens the aromatic ring by producing succinaldehyde (Fig. 12) [43].
On the other hand, the vapor-phase catalytic oxidation of furan and its derivatives, using air as oxidant, produces maleic acid as the main reaction product [70]. Formation of small amounts of acetic and oxalic acids have also been reported [71]. Typical oxidation conditions involve the use of vanadium-based catalysts for reaction in the temperature range 290–410 °C [70, 72]. During catalytic oxidation, furan derivatives have to undergo oxidative elimination of the side chain [72]. Substituents are oxidized to furan carboxylic acids, which are then decarboxylated to furan (Fig. 13). As of this point, the reaction involves the formation of an endoperoxide, followed by the formation of malealdehyde and finally maleic acid [71]. Substituents on the aromatic ring influence the reaction rate and decrease the yield to maleic acid [72].
Details on the oxidation of furan using singlet oxygen (photochemical oxidation) as well as other oxidants common to the organic chemists can be found in [42, 71, 73].
The reactivity of benzofuran is different from that of furan. The oxidation of benzofuran with air is not reported on in standard texts [42, 43, 62], or in specialized texts [74]. In fact, it seems that oxidation of this oxygen-containing compound requires the use of a catalyst to enable oxidative addition (coupling) reactions [75,76,77]. On the other hand, low temperature autoxidation of benzofuran resulted in the formation of addition products [67]. In this case, the experimental evidence indicated that coumaranone, the ketone obtained by oxidation of benzofuran, was a key intermediate in the oxidative dimerization process. Carbon–carbon coupling through the 2- and 3-positions of the furan ring in this ketone compound led to the addition product.
Discussion
Reactivity of aromatic hydrocarbons to oxidation
The aromatic sextet
Even though aromatic compounds have been the center of countless studies, it is valuable to refer to two specific contributions in relation to the relative stability of aromatic compounds compared to cyclic aliphatic compounds and conjugated polyenes. Hückel’s rule, given by the formula 4n + 2, where n is zero or an integer, successfully explained the stability of benzene, compared to other monocyclic conjugated polyenes [78]. On the other hand, the Clar’s π-sextet rule has enabled a description of the stability of polycyclic systems [79].
According to Clar’s rule, the aromatic sextet or π-sextet is denoted by a circle. The appropriate way to draw Clar structures is explained in [80]. Due to the importance of the Clar description of multinuclear aromatics as way to explain the oxidation of aromatics, a brief description is included here. Three simple rules are followed to build aromatic structures [80, 81]:
-
(1)
Circles denoting π-sextets cannot be drawn in adjacent benzenoid rings. This representation would erroneously indicate the presence of 12 π-electrons, when in reality only 10 π-electrons are present.
-
(2)
On ignoring the rings with π-sextets, all other benzenoid rings must have a Kekulé structure. Hence, rings might have one or two double bonds, or might be empty but there must not be unpaired electrons.
-
(3)
Conditioned to the above constrains, a Clar structure must contain the maximum number possible of π-sextets. When dealing with a linear string of benzenoid rings (e.g., anthracene), the π-sextet can occupy any of the rings, which is designated using an arrow.
Clar’s rule classifies aromatic rings into four categories [35], as illustrated by Fig. 14:
-
(1)
Rings having localized π-sextets: Aromatic rings having a localized π-sextet, also known as benzenoid rings, display benzenoid-like stability [82]. Benzenoid rings are considered to be the most aromatic centers in the polyaromatic hydrocarbons; other rings are less aromatic in comparison, and so are chemically more reactive than the benzenoid rings [79].
-
(2)
Rings sharing a migrating π-sextet: Some multinuclear aromatics can be represented by more than one Clar structure. The aromatic sextet in naphthalene can exist in any of the two rings. However, resonance makes the rings in this molecule equal in terms of electron density, and so it is not possible to assign the π-sextet to any one ring alone. Instead, the π-sextet migrates between the rings as denoted by the arrow in Fig. 14b. In linear acenes, the benzenoid character of the π-sextet is diluted when increasing the length of the hydrocarbon [82]; higher acenes are much more reactive due to migration of the π-sextet and loss of the aromatic nature.
-
(3)
Rings with localized double bonds: In some cases, the presence of rings with localized π-sextets leads to the formation of true double bonds. For instance, the outer rings in phenanthrene exhibit a local aromaticity and have more aromatic character than the central ring [79]; as a result, two external π-sextets and a central double bond are formed (Fig. 14c). This double bond is as reactive as any olefinic double bond [35].
-
(4)
Empty rings: The so-called empty rings refer to those rings without π-electrons. They are found in “fully benzenoids” structures (Fig. 14d).
For multinuclear aromatics in which different Clar structures are possible, the one with the greatest number of aromatic sextet carbons is preferred [83]. Aromatic compounds with a higher fraction of aromatic sextet carbons are more stable, compared to those with a higher fraction of isolated double bonds [83].
Influence of structure on oxidation reactivity
The Clar formalism is a valuable tool to rationalize the reactivity of multinuclear aromatics towards oxidation. Formulas with π-sextets not only indicate the stability of a molecule but also point out to the reactive positions on the ground state [82]. Thus, the structure of an aromatic compound can be related with the type of oxidation chemistry observed; it also explains the differences in reactivity among aromatic hydrocarbons. Figure 15 presents the structures of multinuclear aromatics consisting of three fused rings arranged in different geometric configurations. The oxidation chemistry of these compounds, using oxygen as oxidant, will be discussed to illustrate the use of the Clar formalism in explaining oxidation reactivity of multinuclear aromatics.
The triangular building principle results in aromatic hydrocarbons that form radicals [82]. The first member of this series corresponds to perinaphthyl (Fig. 15a). Four resonance structures of this compound have been reported [84]. An aromatic sextet and double bonds are possible, but more importantly, a permanent free radical is present. Due to the diradical nature of oxygen, it can directly perform addition without prior hydrogen abstraction or any other electronic rearrangement (Fig. 16a). This radical–radical coupling should have the lowest activation energy when comparing the oxidation chemistry of (a) (b) and (c) in Fig. 15. Once the oxygen is added, the resonance in perinaphthyl is restricted. Only two of the rings can share a π-sextet, while, a double bond is formed on third one. Oxygen attack is likely to occur in the latter (reactive position).
The angular configuration of phenanthrene leads to the formation of two aromatic sextets in the outer rings (Fig. 15b). The carbon atoms in these rings are quite stable, and so difficult to oxidize. On the contrary, the double bond in the middle ring is very reactive. Oxygen can perform direct addition without previous hydrogen abstraction to give a diradical species: a free radical located on one of the carbons involved in the double bond, and another radical situated on the second oxygen of the O2 molecule that was added (Fig. 16b). Further oxidation involves some electronic rearrangement and bond breaking. The hydrogen bonded to the carbon initially attacked can migrate to stabilize the free charge on the second carbon. Similarly, additional hydrogen can stabilize the radical on the second oxygen atom (from the O2 molecule added) forming a hydroperoxide. The now weaker O–O bond in the hydroxide species can be broken to give a ketone. On the other hand, oxidation of the second carbon of the double bond might involve hydrogen abstraction. It is expected that oxidation of phenanthrene requires more energy than oxidation of perinaphthyl.
Because anthracene is a linear aromatic hydrocarbon, one migrating π-sextet is shared between the three rings (Fig. 15c). Although all carbons should have a similar reactivity, oxygen preferentially attacks the middle ring, namely the 9,10-positions. This corresponds to the resonance structure with two aromatic sextets in the outer rings and a diradical on the 9,10-positions. During autoxidation, the resonance electronic structure of the molecule is disturbed and the two π-electrons at the 9- and 10-position become localized, allowing the oxygen to be added to these two non-adjacent carbons through a para-attack (Fig. 16c). Moreover, the transannular addition of oxygen is favored by the formation of two aromatic sextets in the outer rings of the oxidized product, i.e., 9,10-anthraquinone. The reactivity of the para-position, which is caused by the localization of two π-electrons, is important in the addition reactions of acenes [82]. The transannular addition of oxygen to the 9,10-positions in anthracene takes place more readily than oxygen addition to the 9,10-position in phenanthracene [6].
By comparing the oxidation chemistry of aromatic compounds consisting of three fused rings, it becomes evident that the structure influences the way in which oxygen can interact with a given substrate. The reactivity towards oxidation for linear, angular and condensed aromatic hydrocarbons is different.
Pre-oxidative behavior the non-catalytic oxidation of multinuclear aromatics
Based on the work that appeared around the time that Tipson [6] was writing his review, advances were made in the understanding of how oxygen interacts with aromatics. The first “pre-oxidation” step, which also explained why fluorescence of aromatics was quenched in the presence of dissolved oxygen, was that the oxygen acted as an electron acceptor, with the aromatic molecule being the electron donor [6]. The initiation of oxidation of aromatic hydrocarbons appears to be different in its sequence to that found in aliphatic hydrocarbon oxidation. The latter involves hydrogen abstraction from the hydrocarbon being oxidized to form the free radicals required to start the chain process [85, 86]. Hydrogen abstraction is not necessary for the pre-oxidative step in aromatic oxidation.
In anthracene, the oxygen first adds in a transannular fashion to two carbons, which causes the carbons to change from sp2 to sp3 hybridized carbons. Only after this step is the hydrogen on the sp3 hybridized carbon (non-aromatic hydrogen) abstracted, or transferred. Intermediate products, such as anthrone and oxanthrone were identified and reported in reaction products from the oxidation of anthracene [87]. In phenanthrene, the oxygen first adds to one of the carbons in the double bond. This reaction can be described as radical addition to an olefin, which causes C–O bond formation with one of the carbons of the C=C group and the formation of a carbon centered radical on the other carbon. These carbons are no longer aromatic, but aliphatic, which facilitates hydrogen migration, transfer and/or abstraction. In perinaphthyl, the oxygen can directly perform addition; radical–radical coupling is energetically favorable. It can be seen that regardless of the type of oxidation chemistry taking place, i.e., transannular oxygen addition, oxygen addition to a double bond or free radical chemistry, the first “pre-oxidation” step in aromatic oxidation does not involve hydrogen abstraction as is the case in aliphatic autoxidation. In fact, it facilitates subsequent hydrogen abstraction.
Reactivity of O2 towards heterocyclic aromatics
It has been shown that oxygen can directly interact with multinuclear aromatics (carbocyclic rings) to oxidize the aromatic carbons. The way in which O2 interacts is influenced by the peculiarities of the structure of the aromatic substrate, which dictates the most reactive positions (See “Influence of structure on oxidation reactivity”). The reactivity towards oxidation in heterocyclic hydrocarbons is in turn influenced by the presence of a heteroatom in an aromatic ring.
In the case of S-heterocyclic aromatic compounds, autoxidation in the liquid phase involves the formation of hydroperoxides species by oxidation of molecules other that the S-heterocyclic aromatic. Oxidation of thiophenes is achieved by the hydroperoxides, rather than by oxygen itself. Furthermore, the process is highly selective and the oxidation of the heterocyclic sulfur, to give asulfoxides and sulfones, is the main oxidative transformation. Vapor-phase oxidation of S-heterocyclic aromatic compounds requires different conditions and takes place by a catalytic process. In this case, the oxidation of the S-containing compounds yields sulfur dioxide and sulfur trioxide and smaller aromatic compounds compared to the initial molecules. Metal oxides or salts of metals having several oxidation states are used as catalysts [88].
Autoxidation of N-heterocyclic aromatic compounds follows different chemical routes depending on the basic or non-basic character of the nitrogen atom. Oxidation of the pyridinic nitrogen with air is difficult. Other oxidants, e.g., peracids, have been used to convert pyridines into N oxides. On the other hand, oxidation of pyrroles is readily achieved by oxygen. In this case, the main oxidative transformation involves the oxidation of the aromatic carbons to form products of polymeric nature (addition products), in which oxygen might or might not be present.
In the case of O-heterocyclic aromatic compounds, the reactivity towards autoxidation of furan and its derivatives is quite different of that of benzofuran and its benzologs. Oxygen can readily attack the aromatic carbons of furan to ring-open the structure. On the other hand, autoxidation of the aromatic carbons of the 5-membered ring in benzofuran leads to addition products.
Catalysis in aromatic oxidation
Transition metal oxides, in particular vanadium oxides, are the most important catalysts in industrially successful processes for the selective oxidation of aromatics with air (see “Oxidation of aromatic hydrocarbons”).
A detailed discussion on the catalytic chemistry of vanadium oxides dealing with topics such as redox behavior, geometry of the catalyst surface, role of the carrier, among others, can be found in [89].
The vapor-phase oxidation of aromatic hydrocarbons occurs mainly by the two-step Mars–Van Krevelen mechanism [25]. In the first step, the aromatic hydrocarbon is oxidized by oxygen from the oxide lattice. Desorption of the oxygenated product forms an oxygen vacancy and leaves an active site in a reduced state at the surface of the catalyst [1]. In the second step, reoxidation of the catalyst active site takes place. This process is achieved either by incorporation of gas phase oxygen or by diffusion of bulk lattice oxygen [1, 90].
Gaseous oxygen is adsorbed on metal oxides surfaces in the form of activated electrophilic species, i.e., O− (oxide) or O2− (superoxide) species, before being incorporated into the lattice in the form of nucleophilic oxygen, i.e., O2− (oxide ion) species [8]. The contribution of non-lattice oxygen to oxidation has not been completely ruled out [89]. The role of the different oxygen species in catalytic oxidation processes has been extensively discussed and a rigorous discussion on the topic can be found in [91]. For metal oxides with a significant mobility of lattice oxygen, regeneration of the catalyst by diffusion of bulk oxide ions is very efficient; in these cases, the dead time of the active sites is short and the turn-over frequency is high [1].
It has been suggested that the activity and selectivity of metal oxides towards hydrocarbon oxidation depends on the metal–oxygen (M–O) bond strength within the catalyst [92]. Oxides with strong M–O bonds are likely to retain their oxygen making them inactive for oxidation. On the contrary, oxides with weak M–O bonds are likely to give their oxygen away, which might lead to rapid but unselective oxidation. To achieve high selectivity, oxides with M–O bonds of intermediate strength should be present in the catalyst.
M–O bond strengths have been measured using different approaches. Andersen and Kung [93] correlated the differential heat of reoxidation, measured by calorimetry, with the reaction selectivity for the oxidative dehydrogenation of butane on V2O5/Al2O3. In this case, the selectivity was low when the heat of reoxidation was small but it increased with the increase in the heat of reoxidation. Results suggested that the heat of reoxidation measures the M–O bond strength and indicates the ease of the removal of lattice oxygen. Simons et al. [94] studied the interaction between 1-butene and butadiene with a series of metal oxides (MnO2, V2O5, CuO, Co3O4, Fe2O3, NiO, TiO2, SnO2, ZnO and Cr2O3) over the temperature range 300–600 °C. They related the M–O bond strength with Qo, a single parameter that represented the heat necessary to dissociate ½ O2 from the oxide catalysts. Their work indicated that catalytic activity decreased with increasing the M–O bond strength, i.e., high values of Qo, and that a maximum of selectivity could be obtained for intermediate M–O bond strength, i.e., values of Qo of 210–250 kJ mol−1 (50–60 kcal mol−1).
Regardless of the complexity of the chemistry of metal oxide catalysts in aromatic oxidation, there are a few comments that are worth making. First, the action of oxygen as oxidant of multinuclear aromatics includes the addition to a double bond, addition to p-centers (transannular addition) and formation of o-, pseudo-p- or p-diones (Fig. 17) [6]. These primary pathways do not rely on the presence of a metal oxide catalyst, e.g., V2O5. However, the selective formation of oxygenates accompanied by the cleavage of the aromatic rings has only been successful at industrial level when catalyzed by metal oxides, e.g., V2O5. Second, catalytic aromatic oxidation has a significant effect on minimizing undesired side reactions, i.e., network of free radical reactions and total combustion. Metal oxide catalysts may enable transition states that would not require the aromatic carbons to assume a free radical nature. Also, catalysts enable conversion processes requiring lower oxidation temperatures, which inherently reduce losses due to combustion reactions. Third, metal oxides accelerate selective oxidation of aromatics. By accelerating selective oxidation compared to other oxidation reactions, catalysts facilitate the production of intermediate oxidation products, namely the desired products, to be isolated in commercial yields under controlled reaction conditions.
Implications for industrial use
Production of oxygenated chemicals by the oxidation of aromatic hydrocarbons with oxygen can be considered an industrially proven technology (See “Oxidation of aromatic hydrocarbons”). These processes make use of purified hydrocarbons as starting materials. The same principle applies to oxidation with oxygen for the manufacturing of smaller volume chemical products, which were not reviewed.
As explained in the introduction, the review was motivated by the potential use of autoxidation to add value to materials that contain a high concentration of multinuclear aromatics in a mixture, such as heavy oil, bitumen, and coal liquids. The application that appears to have the most scope for value addition is the use of O2 instead of H2 as reagent for ring-opening of multinuclear aromatics. The purpose of ring-opening is to ultimately convert the multinuclear aromatics into smaller and/or less refractory molecules for refining to transport fuels and chemicals.
Oxidative degradation is a recurring theme in the literature on coal conversion, and in particular, the wet oxidative conversion of coal to lower molecular mass coal acids [95,96,97,98]. Although it was found that oxidation using O2 was slower compared to chemical oxidation [99], the outcome was the similar.
The oxidative ring-opening of some multinuclear aromatic compounds appears to be quite facile, for example, oxidation pathway of benzo[a]pyrene with O2 leads primarily to the ring-opened product [100]. Low temperature wet oxidative dissolution of coal with O2 was reported to achieve dissolution yields of around 70% [97, 98].
Analogous claims were made for petroleum derived asphaltenes, but these could not be substantiated [101]. Oxidative hardening appears to dominate the autoxidation of heavy petroleum fractions [7]. Conversion leading to oxidative addition reactions is related to the presence of specific compound classes [67].
The implication for the potential industrial application of autoxidation for ring-opening of multinuclear aromatics is far reaching. It implies that when addition-prone compound classes are present, a non-radical pathway for oxidative ring-opening must be employed. This is possible by catalytic oxidation, for example, by making use of metal oxide catalysts. It remains to be seen whether catalysts can be employed with heavy oil and bitumen in a way that would enable acceptable catalyst cycle times and catalyst life time for practical industrial application.
Conclusions
Heavy aromatic feed materials, e.g., coals liquids, bitumen and heavy oils could potentially benefit from conversion by oxidation with air. Oxygen may act as a reagent to ring-open multinuclear aromatics and give smaller and/or less refractory compounds for refining to transportation fuels and petrochemicals. Based on these premises, this work reviewed the autoxidation chemistry of the atoms that are part of an aromatic ring structure, i.e., aromatic carbons and heteroatoms contained in an aromatic ring.
The following observations were made from the available literature on selective catalytic oxidation of aromatic hydrocarbons:
-
(a)
Production of oxygenates accompanied by cleavage of the aromatic rings has only been successful at industrial scale when catalyzed by transition metal oxides, in particular vanadium oxides. Catalyzed processes operate in the temperature range 320–450 °C.
-
(b)
The catalytic action of metal oxides involves (1) minimizing the undesired reactions, i.e., free radical chemistry and total combustion, by limiting the formation of transition states that are of free radical nature (2) lowering the oxidation temperatures, which helps controlling the selectivity losses due to total combustion, and (3) accelerating selective oxidation compared to other oxidation reactions, which facilitates the isolation of intermediate oxidation products at commercial yields.
-
(c)
The chemistry of metal oxide catalysts in aromatic oxidation is complex. Even though processes have been described by the two-step Mars–Van Krevelen mechanism, a redox mechanism which states that oxidation takes place by transfer of lattice oxygen, the contribution of non-lattice oxygen to oxidation has not been completely ruled out.
-
(d)
The absence of a metal oxide catalyst is detrimental for the reaction selectivity. Depending on the temperature of reaction, non-catalyzed oxidation favors total combustion over partial oxidation or promotes addition reactions (condensation and polymerization reactions).
There are significant differences in the autoxidation chemistry of heterocyclic aromatics compared to that of multinuclear hydrocarbon aromatics (carbocyclic rings that do not contain heteroatoms). Some points to consider are:
-
(e)
The way in which oxygen interacts with heterocyclic aromatic compounds during oxidation is influenced by the nature of the heteroatom present in an aromatic ring.
-
(f)
Liquid-phase oxidation of S-heterocyclic aromatics is achieved by hydroperoxides species, rather than by oxygen itself. The oxidation of the aromatic sulfur to form sulfoxides and sulfones species is the main oxidative transformation.
-
(g)
Oxidation of N-heterocyclic aromatics depends on the basic or non-basic character of the nitrogen atom. Pyridines do not interact directly with oxygen and so oxidation to give N oxides requires the use of other oxidants, e.g., peracids. Pyrroles readily interact with oxygen; oxidation of the aromatic carbons leads to products that may or may not contain oxygen.
-
(h)
The reactivity of oxygen towards O-heterocyclic aromatic compounds is quite different for furan and it derivatives compared to that of benzofuran and its benzologs. Oxidation of furan readily ring opens the structure, whereas oxidation of benzofuran leads to addition products. The most common outcome is the formation of heavier products by oxygen-mediated addition reactions.
-
(i)
With exception of furan and its derivatives, the oxidation with air of heterocyclic aromatics does not lead to ring-opening except at severe non-selective oxidation conditions (total combustion conditions).
The Clar formalism is a valuable tool in the understanding of the fundamentals of aromatic oxidation and its implications for industrial use. Some relevant comments are:
-
(j)
The interaction of oxygen with multinuclear aromatics is particular to their structure. As a result, fundamentally different oxidation chemistries are possible, namely transannular oxygen addition, oxygen addition to a carbon–carbon double bond, or free radical chemistry.
-
(k)
The sequence during the initiation of the oxidation of aromatic hydrocarbons seems to be different compared to that found in aliphatic hydrocarbons. Regardless of the type of chemistry taking place, the first “pre-oxidation” step does not involve hydrogen abstraction.
-
(l)
Free radical oxidation can likely not be used for conversion of petroleum due to the loss in selectivity, e.g., addition reactions. The only realistic strategy for oxidative ring-opening of heavy aromatic feed materials is catalytic oxidation.
References
Warren BK, Oyama ST (eds) (1996) Heterogeneous hydrocarbon oxidation (ACS Symp. Ser. 638). American Chemical Society, Washington, DC
Sittig M (1962) Combine oxygen and hydrocarbons for profit. Gulf Publishing Company, Houston
Denisov ET, Mitskevich NI, Agabekov VE (1977) Liquid-phase oxidation of oxygen-containing compounds. Consultants Bureau, New York
Clerici MG, Kholdeeva OA (eds) (2013) Liquid phase oxidation via heterogeneous catalysis. Organic synthesis and industrial applications. Wiley, Hoboken
Duprez D, Cavani F (eds) (2014) Handbook of advanced methods and processes in oxidation catalysis. From laboratory to industry. Imperial College Press, London
Tipson RS (1965) Oxidation of polycyclic aromatic hydrocarbons. A review of the literature (National Bureau of Standards Monograph). US Government Printing Office, Washington, DC
Petersen JC (1993) Asphalt oxidation—an overview including a new model for oxidation proposing that physicochemical factors dominate the oxidation kinetics. Fuel Sci Technol Int 11:57–87
Centi G, Cavani F, Trifiro F (2001) Selective oxidation by heterogeneous catalysis. Kluwer Academic/Plenum Publishers, New York
Lücke B, Martin A (1999) The future role of aromatics in refining and petrochemistry. In: Proceedings Of the DGMK-conference on petrochemistry, Erlangen, Germany, p 139
Patai S (ed) (1974) The chemistry of the quinonoid compounds. John Wiley & Sons, London
Grzybowski M, Skonieczny K, Butenschön H, Gryko DT (2013) Comparison of oxidative aromatic coupling and the Scholl reaction. Angew Chem Int Ed 52:9900–9930
Lücke B, Narayana KV, Martin A, Jähnisch K (2004) Oxidation and ammoxidation of aromatics. Adv Synth Catal 346:4107–4124
Song N, Zhang D, Huang H, Zhao H, Tian F (1999) Behavior of vanadium–phosphorus-oxides catalyst for butane oxidation to maleic anhydride in a single-tube packed bed reactor. Catal Today 51:85–91
Cavani F, Trifirò F (1992) Some innovative aspects in the production of monomers via catalyzed oxidation processes. Appl Catal A 88:115–135
Benzene and its industrial derivatives; Hancock, E. G. Ed.; Ernest Benn: London, 1975
Marek LF, Hahn DA (1932) The catalytic oxidation of organic compounds in the vapor phase. Chemical Catalogue Company, New York
Dixon JK, Longfield JE (1960) Hydrocarbon oxidation. In: Emmett PH (ed) Catalysis. Vol. VII. Oxidation, hydration, dehydration and cracking catalysts. Reinhold, New York, pp 183–280
Bielański A, Haber J (1991) Oxygen in catalysis. Marcel Dekker Inc, New York
Lohbeck K, Haferkorn H, Fuhrmann W, Fedtke N (2012) Maleic and fumaric acids. In: Ullman’s encyclopedia of industrial chemistry. Wiley-VCH Verlag GmbH & Co, Weinheim
Bayne J, King IR, Richard M (1972) Laporte industries limited. Vapor phase oxidation of benzene to maleic anhydride using an aluminum phosphate supported catalyst. Pat Appl US 3697550
Di Cio A, Vitali A, Prolizenz AG (1975) Oxidation of benzene to maleic anhydride using a new catalyst. Pat Appl US 3917655
Barker RS, Halcon International Inc. (1975) Method of oxidizing benzene to maleic anhydride using a vanadium, molybdenum, boron, containing catalyst. Pat Appl US 3867412
Trivedi BC, Culbertson BM (1982) Maleic anhydride. Plenum Press, New York
Bielański A, Najbar M (1997) V2O5–MoO3 catalysts for benzene oxidation. Appl Catal A 157:223–261
Mars P, Van Krevelen DW (1954) Oxidations carried out by means of vanadium oxide catalysts. Chem Eng Sci Spec Suppl 3:41–59
Mason RT (2002) Naphthalene. In: Kirk-Othmer encyclopedia of chemical technology. John Wiley & Sons, Inc.
De Maria F, Longfield JE, Butler G (1961) Catalytic reactor design. Ind Eng Chem 53:259–266
Wainwright MS, Foster N (1979) Catalysts, kinetics and reactor design in phthalic anhydride synthesis. Catal Rev Sci Eng 19:211–292
Dálessandro AF, Farkas J (1956) The kinetics of the catalytic oxidation of naphthalene. Colloid Sci 11:653–670
Calderbank PH (1952) Catalytic oxidation of naphthalene. Ind Chem 28:291–295
Ioffe I, Sherman Y (1954) Issledovaniya V Oblasti Kinetiki i Mekhanizma Parofaznogo Okisleniya Aromaticheskikh Uglevodorodov. 5. Kinetika Okisleniya Naftalina Na Smeshannom Vanadievom Katalizatore. Zhurnal Fiz. Khimii 28:2095–2106
Cofrancesco AJ (2000) Anthraquinone. In: Kirk-othmer encyclopedia of chemical technology. John Wiley & Sons Inc., New York, pp 1–13. https://doi.org/10.1002/0471238961.0114200803150618.a01
Brückner A, Baerns M (1997) Selective gas-phase oxidation of polycyclic aromatic hydrocarbons on vanadium oxide-based catalysts. Appl Catal A 157:311–334
Vogel A (2000) Anthraquinone. In: Ullmann’s encyclopedia of industrial chemistry. Wiley-VCH, Weinheim, Germany. https://doi.org/10.1002/14356007.a02_347
Clar E (1972) The aromatic sextet. John Wiley & Sons, London
Montoya Sánchez N, De Klerk A (2014) Oxidative ring-opening over metal oxides. Prepr Pap Am Chem Soc Div Energy Fuels 59(2):558–561
Brooks JD (1955) The catalytic oxidation of phenanthrene. J Appl Chem 5:250–260
Pyatnitskii YI (1976) Heterogeneous catalytic oxidation of aromatic hydrocarbons in the gas phase. Russ Chem Rev 45(8):762–776
Vrbaski T (1963) Oxidation of aromatic compounds. US Patent 3,306,915
Lorz PM, Towae FK, Enke W, Jäckh R, Bhargava N, Hillesheim W (2007) Phthalic acid and derivatives. In: Ullmann's encyclopedia of industrial chemistry. Wiley-VCH, Weinheim, Germany. https://doi.org/10.1002/14356007.a20_181.pub2
Witkop B, Patrick JB, Rosenblum M (1951) Ring effects in autoxidation. A new type of Camps reaction. J Am Chem Soc 73:2641–2647
Joule JA, Mills K (2010) Heterocyclic Chemistry, 5th edn. Wiley, Chichester
Bansal RK (2008) Heterocyclic chemistry, 4th edn. Tunbridge Wells, Anshan Ltd
Javadli R, De Klerk A (2012) Desulfurization of heavy oil. Appl Petrochem Res 1:3–19
Prado GHC, Rao Y, De Klerk A (2017) Nitrogen removal from oil: a review. Energy Fuels 31:14–36
Hardy DR, Wechter MA (1994) Characterization of soluble macromolecular oxidatively reactive species (SMORS) from middle distillate diesel fuels: their origin and role in instability. Energy Fuels 8:782–787
Gray MR, Ayasse AR, Chan EW, Veljkovic M (1995) Kinetics of hydrodesulfurization of thiophenic and sulfide sulfur in Athabasca bitumen. Energy Fuels 9:500–506
Ma X, Sakanishi K, Mochida I (1996) Hydrodesulfurization reactivities of various sulfur compounds in vacuum gas oil. Ind Eng Chem Res 35:2487–2494
Paniv PM, Pysh’ev SV, Gaivanovich VI, Lazorko OI (2006) Current problems, nontraditional technologies, noncatalytic oxidation desulfurization of the kerosene cut. Chem Technol Fuels Oils 42:159–166
Xu X, Moulijn JA, Ito E, Wagemans R, Makke M (2008) Deep desulfurization of fossil fuels by air in the absence of a catalyst. Chem. Sustain. 1:817–819
Ma X, Zhou A, Song C (2007) A novel method for oxidative desulfurization of liquid hydrocarbon fuels based on catalytic oxidation using molecular oxygen coupled with selective adsorption. Catal Today 123:276–284
Murata S, Murata K, Kidena K, Nomura M (2004) A novel oxidative desulfurization system for diesel fuels with molecular oxygen in the presence of cobalt catalysts and aldehydes. Energy Fuel 18:116–121
Ford JF, Rayne TA, Adlington DG (1967) Desulfurization of hydrocarbons using oxidative and hydro-treatments. US Patent 3,341,448
Attar A, Corcoran WH (1978) Desulfurization of organic sulfur compounds by selective oxidation. 1. Regenerable and nonregenerable oxygen carriers. Ind Eng Chem Prod Res Dev 17:102
You N, Kim MJ, Jeong K, Jeong S, Park Y, Jeon J (2010) Catalytic removal of sulfur dioxide from dibenzothiophene sulfone over Mg–l mixed oxides supported on mesoporous silica. J Nanosci Nanotechnol 5:3663–3666
Herron JT (1988) Thermochemistry of sulfoxides and sulfones. In: Pata S, Rappoport Z, Stirling C (eds) The chemistry of sulphones and sulfoxides. John Wiley & Sons, Chichester, pp 95–106
Weh R, De Klerk A (2017) Thermochemistry of sulfones relevant to oxidative desulfurization. Energy Fuels 31:6607–6614
Javadli R, De Klerk A (2012) Desulfurization of heavy oils. Oxidative desulfurization (ODS) as potential upgrading pathway for oil sands derived bitumen. Energy Fuels 26:594–602
Katritzky AR (1985) Handbook of heterocyclic chemistry, 1st edn. Pergamon Press, Oxford
Ishihara A, Wang D, Dumeignil F, Amano H, Qian EW, Kabe T (2005) Oxidative desulfurization and denitrogenation of a light gas oil using an oxidation/adsorption continuous flow process. Appl Catal A 279:279–287
Shiraishi Y, Tachibana K, Hirai T, Komasawa I (2002) Desulfurization and denitrogenation process for light oils based on chemical oxidation followed by liquid–liquid extraction. Ind Eng Chem Res 41:4362–4375
Balster LM, Zabarnick S, Striebitch RC, Shafer LM, West ZJ (2006) Analysis of polar species in jet fuel and determination of their role in autoxidative deposit formation. Energy Fuels 20:2564–2571
Oswald AA, Noel F (1961) Role of pyrroles in fuel instability. J Chem Eng Data 6:294–301
Jones RA, Bean GP (1977) The chemistry of Pyrroles. Academic Press, London
Jones RA (1990) Pyrroles. Wiley, New York
Linhares M, Rebelo SL, Simões MMQ, Silva AMS, Graça M, Neves PMS, Cavaleiron JAS, Freire C (2014) Biomimetic oxidation of indole by Mn(III)porphyrins. Appl Catal A 470:427–433
Siddiquee MN, De Klerk A (2015) Heterocyclic addition reactions during low temperature autoxidation. Energy Fuels 29:4236–4244
Davies DT (1992) Aromatic heterocyclic chemistry. Oxford University Press, Oxford
Katritzky AR, Lagowski JM (1971) Chemistry of the heterocyclic N-Oxides. Academic Press, London
Milas NA, Walsh WL (1935) Catalytic oxidations I. Oxidations in the furan series. J Am Chem Soc 57:1389–1393
Badovskaya LA, Povarova LV (2009) Oxidation of furans (review). Chem Heterocycl Compd 45:1023–1034
Kreile DR, Slavinskaya VA, Shimanskaya MV, Lukevits EY (1969) The reactivity of furan compounds in vapor-phase catalytic oxidation. Chem Heterocycl Compd 5:579–581
Mensah TA, Ho C, Chang SS (1936) Products identified from photosensitized oxidation of selected furanoid flavor compounds. J Agric Food Chem 34:336–338
Mustafa A (1974) Benzofurans (the chemistry of heterocyclic compounds Vol. 29). Wiley, New York
Dao-Huy T, Haider M, Glatz F, Schnürch M, Milhovilovic MD (2014) Direct arylation of benzo[b]furan and other benzo-fused heterocycles. Eur J Org Chem 36:8119–8125
Dwight TA, Rue NR, Charyk D, Josselyn R, DeBoef B (2007) C–C bond formation via double C–H functionalization: aerobic oxidative coupling as a method for synthesizing heterocoupled biaryls. Org Lett 9:3137–3139
Do H-Q, Daugulis O (2009) An aromatic Glaser–Hay reaction. J Am Chem Soc 131:17052–17053
Waack R (1962) The stability of the “Aromatic Sextet”. J Chem Educ 39:469–472
Solà M (2013) Forty years of Clar’s aromatic π-sextet rule. Front Chem 1(22):1–8
Balaban AT (2004) Clar formulas: how to draw and how not to draw formulas of polycyclic aromatic hydrocarbons. Polycycl Aromat Compd 24:83–89
Jiang D, Chen Z (eds) (2013) Graphene chemistry: theoretical perspectives, first edition. John Wiley & Sons, United Kingdom
Clar E (1964) Polycyclic hydrocarbons (Vol 1). Academic Press, London
Ruiz-Morales Y, Mullins O (2007) Polycyclic aromatic hydrocarbons of asphaltenes analyzed by molecular orbital calculations with optical spectroscopy. Energy Fuels 21:256–265
Klein DJ, Rosenfeld V (2014) Forcing, freedom, and uniqueness in graph theory and chemistry. Croat Chem Acta 87:49–59
Emanuel NM, Denisov ET, Maizus ZK (1967) Liquid-phase oxidation of hydrocarbons. Plenum Press, New York (translated by B.J. Hazzard)
Sheldon RA, Kochi JK (1981) Metal-catalyzed oxidations of organic compounds. Academic Press, New York
David B, Boule P (1993) Phototransformation of PAHs adsorbed on SiO2 in aqueous suspension. In: Garrigues P, Lamotte M (eds) Polycyclic aromatic compounds. Synthesis, properties, analytical measurements, occurrence and biological effects. Gordon and Breach Science Publishers, Amsterdam, pp 127–133
Ismagilov Z, Yashnik M, Kerzhentsev M, Parmon V, Bourane A, Al-Shahrani FM, Hajji AA, Koseoglo OR (2011) Oxidative desulfurization of hydrocarbon fuels. Catal Rev 53:199–255
Grzybowska-Świerkosz B (ed) (1984) Vanadia catalysts for processes of oxidation of aromatic hydrocarbons. Polish Scientific Publishers, Warsaw
Kung HH (1989) Transition metal oxides: surface chemistry and catalysis; studies in surface science and catalysis, vol 45. Elsevier, Amsterdam, pp 169–199
Bielański A, Haber J (1979) Oxygen in catalysis on transition metal oxides. Catal Rev Sci Eng 19(1):1–41
Sachtler WMH, De Boer NH (1964) Proc. Intern. Congr. catalysis, Amsterdam 3:252
Andersen PJ, Kung HH (1992) Differential heat of reoxidation of reduced V2O/γ-Al2O3. J Phys Chem 96:3114–3123
Simons TGJ, Verheijen EJM, Batist PA, Schuit GC (1968) Oxidation on Metal Oxides Surfaces. In: Oxidation of organic compounds, vol 76. American Chemical Society, pp 261–275
Wender I, Heredy LA, Neuworth MB, Dryden IGC (1981) Chemical reactions and the constitution of coal. In: Elliott MA (ed) Chemistry of coal utilization. Second supplementary volume. Wiley, New York, pp 425–521
Hayatsu R, Scott RG, Winans RE (1982) Oxidation of coal. In: Part D, Trahanovsky WS (eds) Oxidation in organic chemistry. Academic Press, New York, pp 279–354
Hayashi J, Matsuo Y, Kusakabe K, Morooka S (1997) Depolymerization of lower rank coals by low-temperature O2 oxidation. Energy Fuels 11:227–235
Anderson KB, Crelling JC, Huggett WW, Perry D, Fullinghim T, McGill P, Kaelin P (2011) Oxidative hydrothermal dissolution (OHD) of coal and biomass. Prepr Pap Am Chem Soc Div Fuel Chem 56(2):310–311
Stock LM, Obeng M (1997) Oxidation and decarboxylation. A reaction sequence for the study of aromatic structural elements in Pocahontas No. 3 coal. Energy Fuels 11:987–997
Lee-Ruff E, Wang C (1993) Photooxydation of 6-methylbenzo[a]pyrene and related compounds. In: Garrigues P, Lamotte M (eds) Polycyclic aromatic compounds. Synthesis, properties, analytical measurements, occurrence and biological effects. Gordon and Breach Science Publishers, Amsterdam, pp 93–101
Montoya Sánchez N, De Klerk A (2016) Low-temperature oxidative asphaltenes liquefaction for petrochemicals: fact or fiction? Appl Petrochem Res 6:97–106
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Montoya Sánchez, N., de Klerk, A. Autoxidation of aromatics. Appl Petrochem Res 8, 55–78 (2018). https://doi.org/10.1007/s13203-018-0199-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13203-018-0199-4