Abstract
Purpose of Review
Gallant efforts are ongoing to achieve sustained antiretroviral therapy (ART)–free HIV remission in the HIV-infected person; however, most, if not all, current human clinical studies have primarily focused these efforts on targeting viral persistence in CD4 T cells in blood and tissue sanctuaries. The lack of myeloid centered HIV clinical trials, either as primary or secondary end points, has hindered our understanding of the contribution of myeloid cells in unsuccessful trials but may also guide successes in future HIV eradication clinical strategies.
Recent Findings
Recent advances have highlighted the importance of myeloid reservoirs as sanctuaries of HIV persistence and therefore may partially be responsible for viral recrudescence following ART treatment interruption in several clinical trials where HIV was not detectable or recovered from CD4 T cells. Given these findings, novel innovative therapeutic approaches specifically focused on HIV clearance in myeloid cell populations need to be vigorously pursued if we are to achieve additional cases of sustained ART-free remission.
Summary
This review will highlight new research efforts defining myeloid persistence and recent advances in HIV remission and cure trials that would be relevant in targeting this compartment and make an argument as to their clinical relevancy as we progress towards sustained ART-free HIV remission in all HIV-infected persons.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Jambo KC, Banda DH, Kankwatira AM, Sukumar N, Allain TJ, Heyderman RS, et al. Small alveolar macrophages are infected preferentially by HIV and exhibit impaired phagocytic function. Mucosal Immunol. 2014;7(5):1116–26. https://doi.org/10.1038/mi.2013.127.
Cai Y, Sugimoto C, Arainga M, Midkiff CC, Liu DX, Alvarez X, et al. Preferential destruction of interstitial macrophages over alveolar macrophages as a cause of pulmonary disease in simian immunodeficiency virus-infected rhesus macaques. J Immunol. 2015;195(10):4884–91. https://doi.org/10.4049/jimmunol.1501194.
Damouche A, Lazure T, Avettand-Fenoel V, Huot N, Dejucq-Rainsford N, Satie AP, et al. Adipose tissue is a neglected viral reservoir and an inflammatory site during chronic HIV and SIV infection. PLoS Pathog. 2015;11(9):e1005153. https://doi.org/10.1371/journal.ppat.1005153.
Zalar A, Figueroa MI, Ruibal-Ares B, Bare P, Cahn P, de Bracco MM et al. Macrophage HIV-1 infection in duodenal tissue of patients on long term HAART. Antiviral Res. 2010;87(2):269–71. https://doi.org/10.1016/j.antiviral.2010.05.005.
Josefsson L, von Stockenstrom S, Faria NR, Sinclair E, Bacchetti P, Killian M et al. The HIV-1 reservoir in eight patients on long-term suppressive antiretroviral therapy is stable with few genetic changes over time. Proc Natl Acad Sci U S A. 2013;110(51):E4987–96. https://doi.org/10.1073/pnas.1308313110.
Ganor Y, Real F, Sennepin A, Dutertre CA, Prevedel L, Xu L et al. HIV-1 reservoirs in urethral macrophages of patients under suppressive antiretroviral therapy. Nat Microbiol. 2019. https://doi.org/10.1038/s41564-018-0335-z.
Bernard-Stoecklin S, Gommet C, Corneau AB, Guenounou S, Torres C, Dejucq-Rainsford N et al. Semen CD4+ T cells and macrophages are productively infected at all stages of SIV infection in macaques. PLoS Pathog. 2013;9(12):e1003810. https://doi.org/10.1371/journal.ppat.1003810.
Kitagawa M, Lackner AA, Martfeld DJ, Gardner MB, Dandekar S. Simian immunodeficiency virus infection of macaque bone marrow macrophages correlates with disease progression in vivo. Am J Pathol. 1991;138(4):921–30.
Gill V, Shattock RJ, Freedman AR, Robinson G, Griffin GE, Gordon-Smith EC et al. Macrophages are the major target cell for HIV infection in long-term bone marrow culture and demonstrate dual susceptibility to lymphocytotropic and monocytotropic strains of HIV-1. Br J Haematoly. 1996;93(1):30–7.
Cenker JJ, Stultz RD, McDonald D. Brain microglial cells are highly susceptible to HIV-1 infection and spread. AIDS Res Hum Retrovir. 2017;33(11):1155–65. https://doi.org/10.1089/AID.2017.0004.
Eugenin EA, Clements JE, Zink MC, Berman JW. Human immunodeficiency virus infection of human astrocytes disrupts blood-brain barrier integrity by a gap junction-dependent mechanism. J Neurosci. 2011;31(26):9456–65. https://doi.org/10.1523/JNEUROSCI.1460-11.2011.
Hutter G, Nowak D, Mossner M, Ganepola S, Mussig A, Allers K, et al. Long-term control of HIV by CCR5 Delta32/Delta32 stem-cell transplantation. N Engl J Med. 2009;360(7):692–8. https://doi.org/10.1056/NEJMoa0802905.
Henrich TJ, Hu Z, Li JZ, Sciaranghella G, Busch MP, Keating SM, et al. Long-term reduction in peripheral blood HIV type 1 reservoirs following reduced-intensity conditioning allogeneic stem cell transplantation. J Infect Dis. 2013;207(11):1694–702. https://doi.org/10.1093/infdis/jit086.
Mavigner M, Watkins B, Lawson B, Lee ST, Chahroudi A, Kean L, et al. Persistence of virus reservoirs in ART-treated SHIV-infected rhesus macaques after autologous hematopoietic stem cell transplant. PLoS Pathog. 2014;10(9):e1004406. https://doi.org/10.1371/journal.ppat.1004406.
Henrich TJ, Hatano H, Bacon O, Hogan LE, Rutishauser R, Hill A, et al. HIV-1 persistence following extremely early initiation of antiretroviral therapy (ART) during acute HIV-1 infection: an observational study. PLoS Med. 2017;14(11):e1002417. https://doi.org/10.1371/journal.pmed.1002417.
Persaud D, Gay H, Ziemniak C, Chen YH, Piatak M Jr, Chun TW, et al. Absence of detectable HIV-1 viremia after treatment cessation in an infant. N Engl J Med. 2013;369(19):1828–35. https://doi.org/10.1056/NEJMoa1302976.
Luzuriaga K, Gay H, Ziemniak C, Sanborn KB, Somasundaran M, Rainwater-Lovett K, et al. Viremic relapse after HIV-1 remission in a perinatally infected child. N Engl J Med. 2015;372(8):786–8. https://doi.org/10.1056/NEJMc1413931.
Colby DJ, Trautmann L, Pinyakorn S, Leyre L, Pagliuzza A, Kroon E, et al. Rapid HIV RNA rebound after antiretroviral treatment interruption in persons durably suppressed in Fiebig I acute HIV infection. Nat Med. 2018;24(7):923–6. https://doi.org/10.1038/s41591-018-0026-6.
• Honeycutt JB, Thayer WO, Baker CE, Ribeiro RM, Lada SM, Cao Y, et al. HIV persistence in tissue macrophages of humanized myeloid-only mice during antiretroviral therapy. Nat Med. 2017;23(5):638–43. https://doi.org/10.1038/nm.4319. Using the myeloid-only mouse model, this group demonstrates HIV infection of tissue macrophages is rapidly suppressed by ART. However remarkably, post treatment interruption, a delayed viral rebound was observed and consistent with the establishment of persistent infection in tissue macrophages.
Crowe S, Zhu T, Muller WA. The contribution of monocyte infection and trafficking to viral persistence, and maintenance of the viral reservoir in HIV infection. J Leukoc Biol. 2003;74(5):635–41. https://doi.org/10.1189/jlb.0503204.
Igarashi T, Brown CR, Endo Y, Buckler-White A, Plishka R, Bischofberger N, et al. Macrophage are the principal reservoir and sustain high virus loads in rhesus macaques after the depletion of CD4+ T cells by a highly pathogenic simian immunodeficiency virus/HIV type 1 chimera (SHIV): implications for HIV-1 infections of humans. Proc Natl Acad Sci U S A. 2001;98(2):658–63. https://doi.org/10.1073/pnas.021551798.
Micci L, Alvarez X, Iriele RI, Ortiz AM, Ryan ES, McGary CS, et al. CD4 depletion in SIV-infected macaques results in macrophage and microglia infection with rapid turnover of infected cells. PLoS Pathog. 2014;10(10):e1004467. https://doi.org/10.1371/journal.ppat.1004467.
Kumar A, Abbas W, Herbein G. HIV-1 latency in monocytes/macrophages. Viruses. 2014;6(4):1837–60. https://doi.org/10.3390/v6041837.
Gaskill PJ, Zandonatti M, Gilmartin T, Head SR, Fox HS. Macrophage-derived simian immunodeficiency virus exhibits enhanced infectivity by comparison with T-cell-derived virus. J Virol. 2008;82(3):1615–21. https://doi.org/10.1128/JVI.01757-07.
Zhu T, Muthui D, Holte S, Nickle D, Feng F, Brodie S, et al. Evidence for human immunodeficiency virus type 1 replication in vivo in CD14(+) monocytes and its potential role as a source of virus in patients on highly active antiretroviral therapy. J Virol. 2002;76(2):707–16.
Churchill MJ, Wesselingh SL, Cowley D, Pardo CA, McArthur JC, Brew BJ, et al. Extensive astrocyte infection is prominent in human immunodeficiency virus-associated dementia. Ann Neurol. 2009;66(2):253–8. https://doi.org/10.1002/ana.21697.
Gonzalez-Scarano F, Martin-Garcia J. The neuropathogenesis of AIDS. Nat Rev Immunol. 2005;5(1):69–81. https://doi.org/10.1038/nri1527.
• Lamers SL, Rose R, Ndhlovu LC, Nolan DJ, Salemi M, Maidji E, et al. The meningeal lymphatic system: a route for HIV brain migration? J Neuro-Oncol. 2015;22:275–81. https://doi.org/10.1007/s13365-015-0399-y. Key publication which directly visualizes, by RNAscope, HIV RNA in macrophages of cerebellum from post-mortem brain tissue of an ART-suppressed HIV-infected individual. Presents compelling evidence for the myeloid compartment as critical contributors, despite the ongoing controversy, in the brain as a latent source of replication-competent virus.
Koppensteiner H, Brack-Werner R, Schindler M. Macrophages and their relevance in human immunodeficiency virus type I infection. Retrovirology. 2012;9:82. https://doi.org/10.1186/1742-4690-9-82.
Soulet D, Rivest S. Bone-marrow-derived microglia: myth or reality? Curr Opin Pharmacol. 2008;8(4):508–18. https://doi.org/10.1016/j.coph.2008.04.002.
Beschorner R, Nguyen TD, Gozalan F, Pedal I, Mattern R, Schluesener HJ, et al. CD14 expression by activated parenchymal microglia/macrophages and infiltrating monocytes following human traumatic brain injury. Acta Neuropathol. 2002;103(6):541–9. https://doi.org/10.1007/s00401-001-0503-7.
• Honeycutt JB, Wahl A, Baker C, Spagnuolo RA, Foster J, Zakharova O, et al. Macrophages sustain HIV replication in vivo independently of T cells. J Clin Investig. 2016;126(4):1353–66. https://doi.org/10.1172/JCI84456. Demonstrates that macrophages can sustain HIV replication in the absence of T cells in the myeloid only mouse model. HIV-infected macrophages were distributed in various tissues including the brain. This publication thus also contributes to evidence for the myeloid compartment as critical contributors, despite the ongoing controversy, in the brain as a latent source of replication-competent virus.
Ellery PJ, Tippett E, Chiu YL, Paukovics G, Cameron PU, Solomon A, et al. The CD16+ monocyte subset is more permissive to infection and preferentially harbors HIV-1 in vivo. J Immunol. 2007;178(10):6581–9. https://doi.org/10.4049/jimmunol.178.10.6581.
Valcour VG, Shiramizu BT, Shikuma CM. HIV DNA in circulating monocytes as a mechanism to dementia and other HIV complications. J Leukoc Biol. 2010;87(4):621–6. https://doi.org/10.1189/jlb.0809571.
• Avalos CR, Price SL, Forsyth ER, Pin JN, Shirk EN, Bullock BT, et al. Quantitation of productively infected monocytes and macrophages of simian immunodeficiency virus-infected macaques. J Virol. 2016;90(12):5643–56. https://doi.org/10.1128/JVI.00290-16. Presents a robust method for modified quantitative virus outgrowth assay to assess the contribution of productively infected monocytes and macrophages to HIV- and SIV-infected cells in vivo.
Castellano P, Prevedel L, Eugenin EA. HIV-infected macrophages and microglia that survive acute infection become viral reservoirs by a mechanism involving Bim. Sci Rep. 2017;7(1):12866. https://doi.org/10.1038/s41598-017-12758-w
Sonza S, Mutimer HP, Oelrichs R, Jardine D, Harvey K, Dunne A, Purcell DF, Birch C, Crowe SM. Monocytes harbour replication-competent, non-latent HIV-1 in patients on highly active antiretroviral therapy. Aids. 2001;15(1):17–22.
• Avalos CR, Abreu CM, Queen SE, Li M, Price S, Shirk EN, et al. Brain macrophages in simian immunodeficiency virus-infected, antiretroviral-suppressed macaques: a functional latent reservoir. MBio. 2017;8(4). https://doi.org/10.1128/mBio.01186-17. This publication demonstrates the presence of the latent macrophage reservoir in brains of SIV-infected ART-treated macaques. Also presents compelling evidence for the myeloid compartment as critical contributors, despite the ongoing controversy, in the brain as a latent source of replication-competent virus.
Finzi D, Hermankova M, Pierson T, Carruth LM, Buck C, Chaisson RE, et al. Identification of a reservoir for HIV-1 in patients on highly active antiretroviral therapy. Science. 1997;278(5341):1295–300.
Finzi D, Blankson J, Siliciano JD, Margolick JB, Chadwick K, Pierson T, et al. Latent infection of CD4+ T cells provides a mechanism for lifelong persistence of HIV-1, even in patients on effective combination therapy. Nat Med. 1999;5(5):512–7. https://doi.org/10.1038/8394.
Patel SS, Duby AD, Thiele DL, Lipsky PE. Phenotypic and functional characterization of human T cell clones. J Immunol. 1988;141(11):3726–36.
Wong JK, Hezareh M, Gunthard HF, Havlir DV, Ignacio CC, Spina CA, et al. Recovery of replication-competent HIV despite prolonged suppression of plasma viremia. Science. 1997;278(5341):1291–5.
Kuzmichev YV, Veenhuis RT, Pohlmeyer CW, Garliss CC, Walker-Sperling VE, Blankson JN. A CD3/CD28 microbead-based HIV-1 viral outgrowth assay. J Virus Erad. 2017;3(2):85–9.
Bullen CK, Laird GM, Durand CM, Siliciano JD, Siliciano RF. New ex vivo approaches distinguish effective and ineffective single agents for reversing HIV-1 latency in vivo. Nat Med. 2014;20(4):425–9. https://doi.org/10.1038/nm.3489.
• Fun A, Mok HP, Wills MR, Lever AM. A highly reproducible quantitative viral outgrowth assay for the measurement of the replication-competent latent HIV-1 reservoir. Sci Rep. 2017;7:43231. https://doi.org/10.1038/srep43231. Novel method streamlining the viral outgrowth assay using a dual co-receptor expressing cell line, SupT1-CCR5, to replace the PBMC co-culture. Reduces the protocol to a single-step resting CD4+ T cell purification with a custom antibody cocktail significantly reduce labour and improves assay reproducibility.
Laird GM, Eisele EE, Rabi SA, Lai J, Chioma S, Blankson JN, et al. Rapid quantification of the latent reservoir for HIV-1 using a viral outgrowth assay. PLoS Pathog. 2013;9(5):e1003398. https://doi.org/10.1371/journal.ppat.1003398.
Rosenbloom DI, Elliott O, Hill AL, Henrich TJ, Siliciano JM, Siliciano RF. Designing and interpreting limiting dilution assays: general principles and applications to the latent reservoir for human immunodeficiency virus-1. Open Forum Infect Dis. 2015;2(4):ofv123. https://doi.org/10.1093/ofid/ofv123.
• Massanella M, Yek C, Lada SM, Nakazawa M, Shefa N, Huang K, et al. Improved assays to measure and characterize the inducible HIV reservoir. EBioMedicine. 2018;36:113–21. https://doi.org/10.1016/j.ebiom.2018.09.036. Excellent review of modified methods to quantify the HIV reservoir, including an optimized quantitative viral outgrowth assay.
Procopio FA, Fromentin R, Kulpa DA, Brehm JH, Bebin AG, Strain MC, et al. A novel assay to measure the magnitude of the inducible viral reservoir in HIV-infected individuals. EBioMedicine. 2015;2(8):874–83. https://doi.org/10.1016/j.ebiom.2015.06.019.
Bruner KM, Wang Z, Simonetti FR, Bender AM, Kwon KJ, Sengupta S, et al. A quantitative approach for measuring the reservoir of latent HIV-1 proviruses. Nature. 2019. https://doi.org/10.1038/s41586-019-0898-8.
Fernandez G, Zaikos TD, Khan SZ, Jacobi AM, Behlke MA, Zeichner SL. Targeting IkappaB proteins for HIV latency activation: the role of individual IkappaB and NF-kappaB proteins. J Virol. 2013;87(7):3966–78. https://doi.org/10.1128/JVI.03251-12.
Gallastegui E, Marshall B, Vidal D, Sanchez-Duffhues G, Collado JA, Alvarez-Fernandez C, et al. Combination of biological screening in a cellular model of viral latency and virtual screening identifies novel compounds that reactivate HIV-1. J Virol. 2012;86(7):3795–808. https://doi.org/10.1128/JVI.05972-11.
Iglesias-Ussel M, Vandergeeten C, Marchionni L, Chomont N, Romerio F. High levels of CD2 expression identify HIV-1 latently infected resting memory CD4+ T cells in virally suppressed subjects. J Virol. 2013;87(16):9148–58. https://doi.org/10.1128/JVI.01297-13.
Fromentin R, Bakeman W, Lawani MB, Khoury G, Hartogensis W, DaFonseca S, et al. CD4+ T cells expressing PD-1, TIGIT and LAG-3 contribute to HIV persistence during ART. PLoS Pathog. 2016;12(7):e1005761. https://doi.org/10.1371/journal.ppat.1005761.
Banga R, Procopio FA, Noto A, Pollakis G, Cavassini M, Ohmiti K, et al. PD-1+ and follicular helper T cells are responsible for persistent HIV-1 transcription in treated aviremic individuals. Nat Med. 2016;22:754–61. https://doi.org/10.1038/nm.4113.
Hogan LE, Vasquez J, Hobbs KS, Hanhauser E, Aguilar-Rodriguez B, Hussien R, et al. Increased HIV-1 transcriptional activity and infectious burden in peripheral blood and gut-associated CD4+ T cells expressing CD30. PLoS Pathog. 2018;14(2):e1006856. https://doi.org/10.1371/journal.ppat.1006856.
• Abdel-Mohsen M, Kuri-Cervantes L, Grau-Exposito J, Spivak AM, Nell RA, Tomescu C, et al. CD32 is expressed on cells with transcriptionally active HIV but does not enrich for HIV DNA in resting T cells. Sci Transl Med. 2018;10(437):eaar6759. https://doi.org/10.1126/scitranslmed.aar6759. Key publication providing critical evidence as a rebuttal for CD32 as an insufficient marker of HIV infection. Demonstrates that although CD32 is expressed on cells with transcriptionally active HIV, the expression does not enrich for HIV DNA in resting T cells.
Cohn LB, da Silva IT, Valieris R, Huang AS, Lorenzi JCC, Cohen YZ, et al. Clonal CD4(+) T cells in the HIV-1 latent reservoir display a distinct gene profile upon reactivation. Nat Med. 2018;24(5):604–9. https://doi.org/10.1038/s41591-018-0017-7.
• Veenstra M, Byrd DA, Inglese M, Buyukturkoglu K, Williams DW, Fleysher L, et al. CCR2 on peripheral blood CD14(+)CD16(+) monocytes correlates with neuronal damage, HIV-associated neurocognitive disorders, and peripheral HIV DNA: reseeding of CNS reservoirs? J NeuroImmune Pharmacol. 2018. https://doi.org/10.1007/s11481-018-9792-7. Critical publication providing evidence of CCR2 expressing CD14+CD16+ monocytes as potential peripheral blood biomarkers of neurocognitive impairment in the context of ART-supressed chronic HIV infection.
Siliciano JD, Siliciano RF. HIV-1 eradication strategies: design and assessment. Curr Opin HIV AIDS. 2013;8(4):318–25. https://doi.org/10.1097/COH.0b013e328361eaca.
Archin NM, Bateson R, Tripathy MK, Crooks AM, Yang KH, Dahl NP, et al. HIV-1 expression within resting CD4+ T cells after multiple doses of vorinostat. J Infect Dis. 2014;210(5):728–35. https://doi.org/10.1093/infdis/jiu155.
Archin NM, Liberty AL, Kashuba AD, Choudhary SK, Kuruc JD, Crooks AM, et al. Administration of vorinostat disrupts HIV-1 latency in patients on antiretroviral therapy. Nature. 2012;487(7408):482–5. https://doi.org/10.1038/nature11286.
Elliott JH, McMahon JH, Chang CC, Lee SA, Hartogensis W, Bumpus N, et al. Short-term administration of disulfiram for reversal of latent HIV infection: a phase 2 dose-escalation study. Lancet HIV. 2015;2(12):e520–9. https://doi.org/10.1016/S2352-3018(15)00226-X.
Elliott JH, Wightman F, Solomon A, Ghneim K, Ahlers J, Cameron MJ, et al. Activation of HIV transcription with short-course vorinostat in HIV-infected patients on suppressive antiretroviral therapy. PLoS Pathog. 2014;10(10):e1004473. https://doi.org/10.1371/journal.ppat.1004473.
• Olesen R, Vigano S, Rasmussen TA, Sogaard OS, Ouyang Z, Buzon M, et al. Innate immune activity correlates with CD4 T cell-associated HIV-1 DNA decline during latency-reversing treatment with panobinostat. J Virol. 2015;89(20):10176–89. https://doi.org/10.1128/JVI.01484-15. Evidence provided for mediators of innate immune activity as critical contributors to modulate the effects of latency-reversing agents on the viral reservoir.
Sogaard OS, Graversen ME, Leth S, Olesen R, Brinkmann CR, Nissen SK, et al. The depsipeptide romidepsin reverses HIV-1 latency in vivo. PLoS Pathog. 2015;11(9):e1005142. https://doi.org/10.1371/journal.ppat.1005142.
Spivak AM, Andrade A, Eisele E, Hoh R, Bacchetti P, Bumpus NN, et al. A pilot study assessing the safety and latency-reversing activity of disulfiram in HIV-1-infected adults on antiretroviral therapy. Clin Infect Dis. 2014;58(6):883–90. https://doi.org/10.1093/cid/cit813.
Lu HK, Gray LR, Wightman F, Ellenberg P, Khoury G, Cheng WJ, et al. Ex vivo response to histone deacetylase (HDAC) inhibitors of the HIV long terminal repeat (LTR) derived from HIV-infected patients on antiretroviral therapy. PLoS One. 2014;9(11):e113341. https://doi.org/10.1371/journal.pone.0113341.
Gray LR, Cowley D, Crespan E, Welsh C, Mackenzie C, Wesselingh SL, et al. Reduced basal transcriptional activity of central nervous system-derived HIV type 1 long terminal repeats. AIDS Res Hum Retrovir. 2013;29(2):365–70. https://doi.org/10.1089/AID.2012.0138.
Chateauvieux S, Eifes S, Morceau F, Grigorakaki C, Schnekenburger M, Henry E, et al. Valproic acid perturbs hematopoietic homeostasis by inhibition of erythroid differentiation and activation of the myelo-monocytic pathway. Biochem Pharmacol. 2011;81(4):498–509. https://doi.org/10.1016/j.bcp.2010.11.011.
De Felice L, Tatarelli C, Mascolo MG, Gregorj C, Agostini F, Fiorini R, et al. Histone deacetylase inhibitor valproic acid enhances the cytokine-induced expansion of human hematopoietic stem cells. Cancer Res. 2005;65(4):1505–13. https://doi.org/10.1158/0008-5472.CAN-04-3063.
Doyon G, Zerbato J, Mellors JW, Sluis-Cremer N. Disulfiram reactivates latent HIV-1 expression through depletion of the phosphatase and tensin homolog. AIDS. 2013;27(2):F7–F11. https://doi.org/10.1097/QAD.0b013e3283570620.
Diaz L, Martinez-Bonet M, Sanchez J, Fernandez-Pineda A, Jimenez JL, Munoz E, et al. Bryostatin activates HIV-1 latent expression in human astrocytes through a PKC and NF-kB-dependent mechanism. Sci Rep. 2015;5:12442. https://doi.org/10.1038/srep12442.
• Gray LR, On H, Roberts E, Lu HK, Moso MA, Raison JA, et al. Toxicity and in vitro activity of HIV-1 latency-reversing agents in primary CNS cells. J Neuro-Oncol. 2016;22(4):455–63. https://doi.org/10.1007/s13365-015-0413-4. Significant investigation of toxicity and potency of a panel of commonly used LRA including: panobinostat, romidepsin, vorinostat, disulfrim. Good evaluation of LRA potential activation of HIV transcription in latently infecteted cells of the CNS.
Rasmussen TA, Tolstrup M, Moller HJ, Brinkmann CR, Olesen R, Erikstrup C, et al. Activation of latent human immunodeficiency virus by the histone deacetylase inhibitor panobinostat: a pilot study to assess effects on the central nervous system. Open Forum Infect Dis. 2015;2(1):ofv037. https://doi.org/10.1093/ofid/ofv037.
Ostios-Garcia L, Faig J, Leonardi GC, Adeni AE, Subegdjo SJ, Lydon CA, et al. Safety and efficacy of PD-1 inhibitors among HIV-positive patients with non-small cell lung cancer. J Thorac Oncol. 2018;13(7):1037–42. https://doi.org/10.1016/j.jtho.2018.03.031.
Gay CL, Bosch RJ, Ritz J, Hataye JM, Aga E, Tressler RL, et al. Clinical trial of the anti-PD-L1 antibody BMS-936559 in HIV-1 infected participants on suppressive antiretroviral therapy. J Infect Dis. 2017;215(11):1725–33. https://doi.org/10.1093/infdis/jix191.
• Mendoza P, Gruell H, Nogueira L, Pai JA, Butler AL, Millard K, et al. Combination therapy with anti-HIV-1 antibodies maintains viral suppression. Nature. 2018;561(7724):479–84. https://doi.org/10.1038/s41586-018-0531-2. Foundational publication in broadly neutralizing antibody efficacy. Combination treatment with 3BNC117 and 10-1074 targeting independent sites on the HIV-1 envelope mediated significant neutralization of HIV-1 infection during analytical treatment interruption.
Hessell AJ, Hangartner L, Hunter M, Havenith CE, Beurskens FJ, Bakker JM, et al. Fc receptor but not complement binding is important in antibody protection against HIV. Nature. 2007;449(7158):101–4. https://doi.org/10.1038/nature06106.
Bournazos S, Klein F, Pietzsch J, Seaman MS, Nussenzweig MC, Ravetch JV. Broadly neutralizing anti-HIV-1 antibodies require Fc effector functions for in vivo activity. Cell. 2014;158(6):1243–53. https://doi.org/10.1016/j.cell.2014.08.023.
Riddler SA, Zheng L, Durand CM, Ritz J, Koup RA, Ledgerwood J, et al. Randomized clinical trial to assess the impact of the broadly neutralizing HIV-1 monoclonal antibody VRC01 on HIV-1 persistence in individuals on effective ART. Open Forum Infect Dis. 2018;5(10):ofy242. https://doi.org/10.1093/ofid/ofy242.
• Byrareddy SN, Arthos J, Cicala C, Villinger F, Ortiz KT, Little D, et al. Sustained virologic control in SIV+ macaques after antiretroviral and alpha4beta7 antibody therapy. Science. 2016;354(6309):197–202. https://doi.org/10.1126/science.aag1276. Seminal publication reporting that ART plus infusions of a primatized monoclonal antibody against the α4β7 integrin conferred effective SIV control and moderate subsequent reconstitution of their immune systems occurred without a need for further therapy.
• Uzzan M, Tokuyama M, Rosenstein AK, Tomescu C, SahBandar IN, Ko HM, et al. Anti-alpha4beta7 therapy targets lymphoid aggregates in the gastrointestinal tract of HIV-1-infected individuals. Sci Transl Med. 2018;10(461):eaau4711. https://doi.org/10.1126/scitranslmed.aau4711. Presents the first human study of anti-α4β7 therapy in a cohort of HIV-1 infected subjects with mild inflammatory bowel disease.
Tiisala S, Paavonen T, Renkonen R. Alpha E beta 7 and alpha 4 beta 7 integrins associated with intraepithelial and mucosal homing, are expressed on macrophages. Eur J Immunol. 1995;25(2):411–7. https://doi.org/10.1002/eji.1830250216.
Vojnov L, Martins MA, Bean AT, Veloso de Santana MG, Sacha JB, Wilson NA, et al. The majority of freshly sorted simian immunodeficiency virus (SIV)-specific CD8(+) T cells cannot suppress viral replication in SIV-infected macrophages. J Virol. 2012;86(8):4682–7. https://doi.org/10.1128/JVI.06324-11.
• Clayton KL, Collins DR, Lengieza J, Ghebremichael M, Dotiwala F, Lieberman J, et al. Resistance of HIV-infected macrophages to CD8(+) T lymphocyte-mediated killing drives activation of the immune system. Nat Immunol. 2018;19(5):475–86. https://doi.org/10.1038/s41590-018-0085-3. An extensive body of compelling evidence for cytolytic failure for CD8 mediated killing of HIV-infected macrophages. Highlights the consequential hyperinflammatory conditions that poor killing of macrophages suggesting a myeloid-driven mechanism for ongoing chronic inflammation.
Rainho JN, Martins MA, Cunyat F, Watkins IT, Watkins DI, Stevenson M. Nef is dispensable for resistance of simian immunodeficiency virus-infected macrophages to CD8+ T cell killing. J Virol. 2015;89(20):10625–36. https://doi.org/10.1128/JVI.01699-15.
• Pegu A, Asokan M, Wu L, Wang K, Hataye J, Casazza JP, et al. Activation and lysis of human CD4 cells latently infected with HIV-1. Nat Commun. 2015;6:8447. https://doi.org/10.1038/ncomms9447. Compelling evidence for synergistic activation when a highly potent broadly neutralizing antibody is linked to an activating anti-human CD3 antibody. Demonstrates the feasibility of targeted activation and reduction in the number of latently infected T cells ex vivo and in non-human primate model.
• Chew GM, Fujita T, Webb GM, Burwitz BJ, Wu HL, Reed JS, et al. TIGIT marks exhausted T cells, correlates with disease progression, and serves as a target for immune restoration in HIV and SIV infection. PLoS Pathog. 2016;12(1):e1005349. https://doi.org/10.1371/journal.ppat.1005349. Indicates a novel role for TIGIT during HIV disease pathogenesis in immune exhaustion. Also investigates TIGIT immune-exhaustive role rhesus macaques providing evidence for feasibility of further investigating TIGIT blockade as a means of improving CD8+ T cell function in non-human primate model of HIV/AIDS.
Fujita T, Burwitz BJ, Chew GM, Reed JS, Pathak R, Seger E, et al. Expansion of dysfunctional Tim-3-expressing effector memory CD8+ T cells during simian immunodeficiency virus infection in rhesus macaques. J Immunol. 2014;193(11):5576–83. https://doi.org/10.4049/jimmunol.1400961.
Bui JK, Mellors JW. Reversal of T-cell exhaustion as a strategy to improve immune control of HIV-1. AIDS. 2015;29(15):1911–5. https://doi.org/10.1097/QAD.0000000000000788.
Tomescu C, Mavilio D, Montaner LJ. Lysis of HIV-1-infected autologous CD4+ primary T cells by interferon-alpha-activated NK cells requires NKp46 and NKG2D. AIDS. 2015;29(14):1767–73. https://doi.org/10.1097/QAD.0000000000000777.
Deeks SG, Wagner B, Anton PA, Mitsuyasu RT, Scadden DT, Huang C, et al. A phase II randomized study of HIV-specific T-cell gene therapy in subjects with undetectable plasma viremia on combination antiretroviral therapy. Mol Ther. 2002;5(6):788–97. https://doi.org/10.1006/mthe.2002.0611.
Mitsuyasu RT, Anton PA, Deeks SG, Scadden DT, Connick E, Downs MT, et al. Prolonged survival and tissue trafficking following adoptive transfer of CD4zeta gene-modified autologous CD4(+) and CD8(+) T cells in human immunodeficiency virus-infected subjects. Blood. 2000;96(3):785–93.
Yang OO, Tran AC, Kalams SA, Johnson RP, Roberts MR, Walker BD. Lysis of HIV-1-infected cells and inhibition of viral replication by universal receptor T cells. Proc Natl Acad Sci U S A. 1997;94(21):11478–83.
Perez EE, Wang J, Miller JC, Jouvenot Y, Kim KA, Liu O, et al. Establishment of HIV-1 resistance in CD4+ T cells by genome editing using zinc-finger nucleases. Nat Biotechnol. 2008;26(7):808–16. https://doi.org/10.1038/nbt1410.
• Zhang Y, Yin C, Zhang T, Li F, Yang W, Kaminski R, et al. CRISPR/gRNA-directed synergistic activation mediator (SAM) induces specific, persistent and robust reactivation of the HIV-1 latent reservoirs. Sci Rep. 2015;5:16277. https://doi.org/10.1038/srep16277. First reporting of successful demonstration for latent HIV-1 provirus reactivation by dCas9-SAM guided by target-specific MS2-mediated single guide RNA. This approach specifically targeted the enhancer of the HIV-1 LTR promoter and may inform next generation LRA or block and lock strategies.
Hu W, Kaminski R, Yang F, Zhang Y, Cosentino L, Li F, et al. RNA-directed gene editing specifically eradicates latent and prevents new HIV-1 infection. Proc Natl Acad Sci U S A. 2014;111(31):11461–6. https://doi.org/10.1073/pnas.1405186111.
Wang S, Huber PW, Cui M, Czarnik AW, Mei HY. Binding of neomycin to the TAR element of HIV-1 RNA induces dissociation of Tat protein by an allosteric mechanism. Biochemistry. 1998;37(16):5549–57. https://doi.org/10.1021/bi972808a.
Mestre B, Arzumanov A, Singh M, Boulme F, Litvak S, Gait MJ. Oligonucleotide inhibition of the interaction of HIV-1 Tat protein with the trans-activation responsive region (TAR) of HIV RNA. Biochim Biophys Acta. 1999;1445(1):86–98.
• Kessing CF, Nixon CC, Li C, Tsai P, Takata H, Mousseau G, et al. In vivo suppression of HIV rebound by didehydro-Cortistatin a, a “block-and-lock” strategy for HIV-1 treatment. Cell Rep. 2017;21(3):600–11. https://doi.org/10.1016/j.celrep.2017.09.080. Excellent first reporting of a compound with “block and lock” efficacy mediating suppression of viral rebound after treatment interruption in a humanized HIV-infected mouse model.
Mousseau G, Kessing CF, Fromentin R, Trautmann L, Chomont N, Valente ST. The Tat inhibitor didehydro-Cortistatin A prevents HIV-1 reactivation from latency. MBio. 2015;6(4):e00465. https://doi.org/10.1128/mBio.00465-15.
Adams M, Sharmeen L, Kimpton J, Romeo JM, Garcia JV, Peterlin BM, et al. Cellular latency in human immunodeficiency virus-infected individuals with high CD4 levels can be detected by the presence of promoter-proximal transcripts. Proc Natl Acad Sci U S A. 1994;91(9):3862–6.
Chatziandreou N, Farsakoglu Y, Palomino-Segura M, D,Antuono R, Pizzagalli DU, Sallusto F, et al. Macrophage death following influenza vaccination initiates the inflammatory response that promotes dendritic cell function in the draining lymph node. Cell Rep. 2017;18(10):2427–40. https://doi.org/10.1016/j.celrep.2017.02.026.
Cohen N, Stolarsky-Bennun M, Amir-Kroll H, Margalit R, Nussbaum G, Cohen-Sfady M, et al. Pneumococcal capsular polysaccharide is immunogenic when present on the surface of macrophages and dendritic cells: TLR4 signaling induced by a conjugate vaccine or by lipopolysaccharide is conducive. J Immunol. 2008;180(4):2409–18. https://doi.org/10.4049/jimmunol.180.4.2409.
Bar-On Y, Gruell H, Schoofs T, Pai JA, Nogueira L, Butler AL, et al. Safety and antiviral activity of combination HIV-1 broadly neutralizing antibodies in viremic individuals. Nat Med. 2018;24(11):1701–7. https://doi.org/10.1038/s41591-018-0186-4.
Li B, Xu L, Pi C, Yin Y, Xie K, Tao F, et al. CD89-mediated recruitment of macrophages via a bispecific antibody enhances anti-tumor efficacy. Oncoimmunology. 2017;7(1):e1380142. https://doi.org/10.1080/2162402X.2017.1380142.
Levine BL, Humeau LM, Boyer J, MacGregor RR, Rebello T, Lu X, et al. Gene transfer in humans using a conditionally replicating lentiviral vector. Proc Natl Acad Sci U S A. 2006;103(46):17372–7. https://doi.org/10.1073/pnas.0608138103.
Acknowledgements
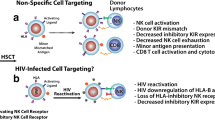
The authors would also like to acknowledge Thomas Premeaux for his construction and design of Fig. 1.
Funding
LCN is supported by the National Institutes of Health (NIH), National Institutes of Mental Health (R01MH112457, R01MH104141), the National Institute of Neurological Disorders and Stroke (R21 NS106970), and the National Institute on Minority Health and Health Disparities (U54 MD007601). The funders of this study had no role in the design of the report.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
LCN has received research support from Tobira Therapeutics and serves as advisory board member for ViiV Healthcare. Brooks I. Mitchell and Elizabeth I. Laws have nothing to disclose.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Central Nervous System and Cognition
Rights and permissions
About this article
Cite this article
Mitchell, B.I., Laws, E.I. & Ndhlovu, L.C. Impact of Myeloid Reservoirs in HIV Cure Trials. Curr HIV/AIDS Rep 16, 129–140 (2019). https://doi.org/10.1007/s11904-019-00438-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11904-019-00438-5