Abstract
Introduction
Oxygen is essential for metabolic processes and in the absence thereof alternative metabolic pathways are required for energy production, as seen in marine invertebrates like abalone. Even though hypoxia has been responsible for significant losses to the aquaculture industry, the overall metabolic adaptations of abalone in response to environmental hypoxia are as yet, not fully elucidated.
Objective
To use a multiplatform metabolomics approach to characterize the metabolic changes associated with energy production in abalone (Haliotis midae) when exposed to environmental hypoxia.
Methods
Metabolomics analysis of abalone adductor and foot muscle, left and right gill, hemolymph, and epipodial tissue samples were conducted using a multiplatform approach, which included untargeted NMR spectroscopy, untargeted and targeted LC–MS spectrometry, and untargeted and semi-targeted GC-MS spectrometric analyses.
Results
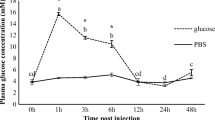
Increased levels of anaerobic end-products specific to marine animals were found which include alanopine, strombine, tauropine and octopine. These were accompanied by elevated lactate, succinate and arginine, of which the latter is a product of phosphoarginine breakdown in abalone. Primarily amino acid metabolism was affected, with carbohydrate and lipid metabolism assisting with anaerobic energy production to a lesser extent. Different tissues showed varied metabolic responses to hypoxia, with the largest metabolic changes in the adductor muscle.
Conclusions
From this investigation, it becomes evident that abalone have well-developed (yet understudied) metabolic mechanisms for surviving hypoxic periods. Furthermore, metabolomics serves as a powerful tool for investigating the altered metabolic processes in abalone.


Similar content being viewed by others
References
Badenhorst, C. P. S., Erasmus, E., Van der Sluis, R., Nortje, C., & Van Dijk, A. A. (2014). A new perspective on the importance of glycine conjugation in the metabolism of aromatic acids. Drug Metabolism Reviews, 46(3), 343–361.
Barkai, R., & Griffiths, C. (1988). An energy budget for the South African abalone Haliotis midae Linnaeus. Journal of Molluscan Studies, 54(1), 43–51.
Boardman, L., Sørensen, J. G., Koštál, V., Šimek, P., & Terblanche, J. S. (2016). Chilling slows anaerobic metabolism to improve anoxia tolerance of insects. Metabolomics, 12(12), 176.
Britz, P., & Venter, S. (2016). Aquaculture review: South Africa. World Aquaculture, 47(4), 19–28.
Carroll, J. L., & Wells, R. M. (1995). Strategies of anaerobiosis in New Zealand infaunal bivalves: Adaptations to environmental and functional hypoxia. New Zealand Journal of Marine and Freshwater Research, 29(2), 137–146.
Cook, P. A. (2014). The worldwide abalone industry. Modern Economy, 5(13), 1181–1186.
Dona, A. C., Jiménez, B., Schäfer, H., Humpfer, E., Spraul, M., Lewis, M. R., et al. (2014). Precision high-throughput proton NMR spectroscopy of human urine, serum, and plasma for large-scale metabolic phenotyping. Analytical Chemistry, 86(19), 9887–9894.
Dunn, W. B. (2008). Current trends and future requirements for the mass spectrometric investigation of microbial, mammalian and plant metabolomes. Physical Biology, 5(1), 011001.
Dunphy, B. J., Watts, E., & Ragg, N. L. (2015). Identifying thermally-stressed adult green-lipped mussels (Perna canaliculus Gmelin, 1791) via metabolomic profiling. American Malacological Bulletin, 33(1), 127–135.
Durazo-Beltrán, E., Viana, M. A. T., D’Abramo, L. R., & Toro-Vazquez, J. F. (2004). Effects of starvation and dietary lipid on the lipid and fatty acid composition of muscle tissue of juvenile green abalone (Haliotis fulgens). Aquaculture, 238(1), 329–341.
Ellis, S., & Steyn, H. (2003). Practical significance (effect sizes) versus or in combination with statistical significance (p values): Research note. Management Dynamics: Journal of the Southern African Institute for Management Scientists, 12(4), 51–53.
Feala, J. D., Coquin, L., Zhou, D., Haddad, G. G., Paternostro, G., & McCulloch, A. D. (2009). Metabolism as means for hypoxia adaptation: Metabolic profiling and flux balance analysis. BMC Systems Biology, 3(1), 91.
Fields, J. H. (1983). Alternatives to lactic acid: Possible advantages. Journal of Experimental Zoology Part A: Ecological Genetics and Physiology, 228(3), 445–457.
Fields, J. H., & Storey, K. B. (1987). Tissue-specific alanopine dehydrogenase from the gill and strombine dehydrogenase from the foot muscle of the cherrystone clam Mercenaria mercenaria (Linn.). Journal of Experimental Marine Biology and Ecology, 105(2–3), 175–185.
Fujimori, T., & Abe, H. (2002). Physiological roles of free d-and l-alanine in the crayfish Procambarus clarkii with special reference to osmotic and anoxic stress responses. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology, 131(4), 893–900.
Gäde, G. (1988). Energy metabolism during anoxia and recovery in shell adductor and foot muscle of the gastropod mollusc Haliotis lamellosa: Formation of the novel anaerobic end product tauropine. The Biological Bulletin, 175(1), 122–131.
Gäde, G., Carlsson, K.-H., & Meinardus, G. (1984). Energy metabolism in the foot of the marine gastropod Nassa mutabilis during environmental and functional anaerobiosis. Marine Biology, 80(1), 49–56.
Gäde, G., & Grieshaber, M. K. (1986). Pyruvate reductases catalyze the formation of lactate and opines in anaerobic invertebrates. Comparative Biochemistry and Physiology Part B: Comparative Biochemistry, 83(2), 255–272.
Garrett, R., & Grisham, C. (2010). Biochemistry, Brooks. (4th ed., pp. 511–812). Belmont, CA: Brooks Cole, Cengage Learning.
Green, S. R., & Storey, K. B. (2016). Regulation of crayfish, Orconectes virilis, tail muscle lactate dehydrogenase (LDH) in response to anoxic conditions is associated with alterations in phosphorylation patterns. Comparative Biochemistry and Physiology Part B: Biochemistry and Molecular Biology, 202, 67–74.
Grieshaber, M., Hardewig, I., Kreutzer, U., & Pörtner, H.-O. (1993). Physiological and metabolic responses to hypoxia in invertebrates. In Reviews of physiology, biochemistry and pharmacology (Vol. 125, pp. 43–147). Berlin: Springer.
Grubert, M. A., Dunstan, G. A., & Ritar, A. J. (2004). Lipid and fatty acid composition of pre-and post-spawning blacklip (Haliotis rubra) and greenlip (Haliotis laevigata) abalone conditioned at two temperatures on a formulated feed. Aquaculture, 242(1), 297–311.
Harcet, M., Perina, D., & Pleše, B. (2013). Opine dehydrogenases in marine invertebrates. Biochemical Genetics, 51(9–10), 666–676.
Harris, J. O., Maguire, G. B., Edwards, S., & Hindrum, S. M. (1998). Effect of ammonia on the growth rate and oxygen consumption of juvenile greenlip abalone, Haliotis laevigata Donovan. Aquaculture, 160(3), 259–272.
Hayat, S., Hayat, Q., Alyemeni, M. N., Wani, A. S., Pichtel, J., & Ahmad, A. (2012). Role of proline under changing environments: A review. Plant Signaling & Behavior, 7(11), 1456–1466.
Hochachka, P. W., Fields, J., & Mustafa, T. (1973). Animal life without oxygen: Basic biochemical mechanisms. American Zoologist, 13(2), 543–555.
Hooper, C., Day, R., Slocombe, R., Benkendorff, K., & Handlinger, J. (2014). Histopathology and haemolymph biochemistry following anaesthesia and movement in farmed Australian abalone (Haliotis rubra × Haliotis laevigata). Aquaculture, 422, 202–210.
Irwin, C., van Reenen, M., Mason, S., Mienie, L. J., Westerhuis, J. A., & Reinecke, C. J. (2016). Contribution towards a metabolite profile of the detoxification of benzoic acid through glycine conjugation: An intervention study. PLoS ONE, 11(12), e0167309.
Kreutzer, U., Siegmund, B. R., & Grieshaber, M. K. (1989). Parameters controlling opine formation during muscular activity and environmental hypoxia. Journal of Comparative Physiology B: Biochemical, Systemic, and Environmental Physiology, 159(5), 617–628.
Li, P., Mai, K., Trushenski, J., & Wu, G. (2009). New developments in fish amino acid nutrition: Towards functional and environmentally oriented aquafeeds. Amino Acids, 37(1), 43–53.
Lindeque, J. Z., Hidalgo, J., Louw, R., & van der Westhuizen, F. H. (2013). Systemic and organ specific metabolic variation in metallothionein knockout mice challenged with swimming exercise. Metabolomics, 9(2), 418–432.
Lindeque, J. Z., van Rensburg, P. J. J., Louw, R., van der Westhuizen, F. H., Florit, S., Ramírez, L., et al. (2015). Obesity and metabolomics: Metallothioneins protect against high-fat diet-induced consequences in metallothionein knockout mice. Omics: A Journal of Integrative Biology, 19(2), 92–103.
Liu, C., Shin, P., & Cheung, S. (2014). Comparisons of the metabolic responses of two subtidal nassariid gastropods to hypoxia and re-oxygenation. Marine Pollution Bulletin, 82(1), 109–116.
Loots, D. T., Erasmus, E., & Mienie, L. J. (2005). Identification of 19 new metabolites induced by ab normal amino acid conjugation in isovaleric acidemia. Clinical Chemistry, 51(8), 1510–1512.
Lu, J., Feng, J., Cai, S., & Chen, Z. (2017). Metabolomic responses of Haliotis diversicolor to organotin compounds. Chemosphere, 168, 860–869.
Lu, J., Shi, Y., Wang, S., Chen, H., Cai, S., & Feng, J. (2016). NMR-based metabolomic analysis of Haliotis diversicolor exposed to thermal and hypoxic stresses. Science of The Total Environment, 545, 280–288.
Mason, S., van Furth, A. M., Mienie, L. J., Engelke, U. F., Wevers, R. A., Solomons, R., et al. (2015). A hypothetical astrocyte–microglia lactate shuttle derived from a 1H NMR metabolomics analysis of cerebrospinal fluid from a cohort of South African children with tuberculous meningitis. Metabolomics, 11(4), 822–837.
Morash, A. J., & Alter, K. (2016). Effects of environmental and farm stress on abalone physiology: Perspectives for abalone aquaculture in the face of global climate change. Reviews in Aquaculture, 8(4), 342–368.
Mouton, A. (2017). Red tide devastates South African abalone farms. Aquaculture Magazine, 31, 32–35.
Müller, M., Mentel, M., van Hellemond, J. J., Henze, K., Woehle, C., Gould, S. B., et al. (2012). Biochemistry and evolution of anaerobic energy metabolism in eukaryotes. Microbiology and Molecular Biology Reviews, 76(2), 444–495.
Nollens, H. H., Keogh, J. A., & Probert, P. K. (2004). Haematological pathology of shell lesions in the New Zealand abalone, Haliotis iris (Mollusca: Gastropoda). Comparative Clinical Pathology, 12(4), 211–216.
O’omolo, S., Gäde, G., Cook, P., & Brown, A. (2003). Can the end products of anaerobic metabolism, tauropine and d-lactate, be used as metabolic stress indicators during transport of live South African abalone Haliotis midae. African Journal of Marine Science, 25(1), 301–309.
Pitcher, G., & Calder, D. (2000). Harmful algal blooms of the southern Benguela current: A review and appraisal of monitoring from 1989 to 1997. African Journal of Marine Science, 22, 255–271.
Prathomya, P., Prisingkorn, W., Jakovlić, I., Deng, F.-Y., Zhao, Y.-H., & Wang, W.-M. (2017). 1H NMR-based metabolomics approach reveals metabolic alterations in response to dietary imbalances in Megalobrama amblycephala. Metabolomics, 13(2), 17.
Ragg, N. L., & Taylor, H. H. (2006). Heterogeneous perfusion of the paired gills of the abalone Haliotis iris Martyn 1784: An unusual mechanism for respiratory control. Journal of Experimental Biology, 209(3), 475–483.
Rosenblum, E., Viant, M., Braid, B., Moore, J., Friedman, C., & Tjeerdema, R. (2005). Characterizing the metabolic actions of natural stresses in the California red abalone, Haliotis rufescens using 1H NMR metabolomics. Metabolomics, 1(2), 199–209.
Roznere, I., Watters, G. T., Wolfe, B. A., & Daly, M. (2014). Nontargeted metabolomics reveals biochemical pathways altered in response to captivity and food limitation in the freshwater mussel Amblema plicata. Comparative Biochemistry and Physiology Part D: Genomics and Proteomics, 12, 53–60.
Salway, J. G. (2004). Metabolism at a glance. Guildford: Wiley.
Sato, M., Takeuchi, M., Kanno, N., Nagahisa, E., & Sato, Y. (1993). Distribution of opine dehydrogenases and lactate dehydrogenase activities in marine animals. Comparative Biochemistry and Physiology Part B: Comparative Biochemistry, 106(4), 955–960.
Schmidt-Nielsen, K. (2007). Animal physiology: Adaptation and environment. New York: Cambridge University Press.
Schymanski, E. L., Jeon, J., Gulde, R., Fenner, K., Ruff, M., Singer, H. P., et al. (2014). Identifying small molecules via high resolution mass spectrometry: Communicating confidence. Environmental Science and Technology, 48(4), 2097–2098.
Shen, Y., Hixson, K. K., Tolic, N., Camp, D. G., Purvine, S. O., Moore, R. J., et al. (2008). Mass spectrometry analysis of proteome-wide proteolytic post-translational degradation of proteins. Analytical Chemistry, 80(15), 5819–5828.
Slabbert, R., & Roodt-Wilding, R. (2006). Non-destructive sampling of juvenile abalone using epipodial tentacles and mucus: Method and application. African Journal of Marine Science, 28(3–4), 719–721.
Sokolova, I. M., Frederich, M., Bagwe, R., Lannig, G., & Sukhotin, A. A. (2012). Energy homeostasis as an integrative tool for assessing limits of environmental stress tolerance in aquatic invertebrates. Marine Environmental Research, 79, 1–15.
Storey, K. B. (2004). Adventures in oxygen metabolism. Comparative Biochemistry and Physiology Part B: Biochemistry and Molecular Biology, 139(3), 359–369.
Storey, K. B. (2005). Functional metabolism: Regulation and adaptation. Hoboeken: Wiley.
Sumner, L. W., Amberg, A., Barrett, D., Beale, M. H., Beger, R., Daykin, C. A., et al. (2007). Proposed minimum reporting standards for chemical analysis. Metabolomics, 3(3), 211–221.
Taylor, H., & Ragg, N. (2005). The role of body surfaces and ventilation in gas exchange of the abalone, Haliotis iris. Journal of Comparative Physiology B, 175(7), 463–478.
Tripp-Valdez, M. A., Bock, C., Lucassen, M., Lluch-Cota, S. E., Sicard, M. T., Lannig, G., et al. (2017). Metabolic response and thermal tolerance of green abalone juveniles (Haliotis fulgens: Gastropoda) under acute hypoxia and hypercapnia. Journal of Experimental Marine Biology and Ecology, 497, 11–18.
Venter, L., van Rensburg, P. J. J., Loots, D. T., Vosloo, A., & Lindeque, J. Z. (2016a). Untargeted metabolite profiling of abalone using gas chromatography mass spectrometry. Food Analytical Methods, 9(5), 1254–1261.
Venter, L., Lindeque, Z., van Rensburg, P. J. J., Van der Westhuizen, F., Smuts, I., & Louw, R. (2015). Untargeted urine metabolomics reveals a biosignature for muscle respiratory chain deficiencies. Metabolomics, 11(1), 111–121.
Venter, L., Loots, D. T., Vosloo, A., Jansen van Rensburg, P., & Lindeque, J. Z. (2016b). Abalone growth and associated aspects: Now from a metabolic perspective. Reviews in Aquaculture, 2, 2016.
Venter, L., van Rensburg, P. J. J. , Loots, D. T., Vosloo, A., & Lindeque, J. Z. (2017). From untargeted LC–QTOF analysis to characterisation of opines in abalone adductor muscle: Theory meets practice. Journal of Chromatography B, 1071, 44–48.
Vosloo, D., van Rensburg, L., & Vosloo, A. (2013). Oxidative stress in abalone: The role of temperature, oxygen and L-proline supplementation. Aquaculture, 416, 265–271.
Vosloo, D., & Vosloo, A. (2010). Response of cold-acclimated, farmed South African abalone (Haliotis midae) to short-term and long-term changes in temperature. Journal of Thermal Biology, 35(7), 317–323.
Warrack, B. M., Hnatyshyn, S., Ott, K.-H., Reily, M. D., Sanders, M., Zhang, H., et al. (2009). Normalization strategies for metabonomic analysis of urine samples. Journal of Chromatography B, 877(5), 547–552.
Wasko, A. P., Martins, C., Oliveira, C., & Foresti, F. (2003). Non-destructive genetic sampling in fish. An improved method for DNA extraction from fish fins and scales. Hereditas, 138(3), 161–165.
Wells, R. M., Baldwin, J., Speed, S. R., & Weber, R. E. (1998). Haemocyanin function in the New Zealand abalones Haliotis iris and H. australis: Relationships between oxygen-binding properties, muscle metabolism and habitat. Marine and Freshwater Research, 49(2), 143–149.
Whitney, E., & Rolfes, S. R. (2008). Understanding nutrition. Belmont: Cengage Learning.
Wilbur, K. M., & Hochachka, P. W. (2016). The Mollusca: Environmental biochemistry and physiology (Vol. 2). New York: Academic Press.
Willers, C., van Rensburg, P. J. J., & Claassens, S. (2016). Can a metabolomics-based approach be used as alternative to analyse fatty acid methyl esters from soil microbial communities? Soil Biology and Biochemistry, 103, 417–428.
Wishart, D. S., Jewison, T., Guo, A. C., Wilson, M., Knox, C., Liu, Y., et al. (2012). HMDB 3.0—the human metabolome database in 2013. Nucleic Acids Research, 41(D1), D801–D807.
Xia, J., Sinelnikov, I. V., Han, B., & Wishart, D. S. (2015). MetaboAnalyst 3.0—making metabolomics more meaningful. Nucleic Acids Research, 43(W1), 251–257.
Acknowledgements
We gratefully acknowledge the North-West University and the Technology Innovation Agency of the Department of Science and Technology of South Africa for financial support. We also thank HIK Abalone Farm (Pty) Ltd, South Africa for supplying experimental animals and welcoming us to your farm.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All of the research pertaining to the perturbations and abalone collection was done in accordance with institutional guidelines of the relevant institutional committees and granted Aquaculture Research Permit.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Venter, L., Loots, D.T., Mienie, L.J. et al. Uncovering the metabolic response of abalone (Haliotis midae) to environmental hypoxia through metabolomics. Metabolomics 14, 49 (2018). https://doi.org/10.1007/s11306-018-1346-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-018-1346-8