Abstract
A challenge of phenological research is to integrate remotely sensed observations obtained at different spatial and temporal scales to provide information that contains both a high temporal density and fine spatial scale observations. This study aims to improve the level of spatial detail and the temporal density required for efficient monitoring of vegetation phenology by applying two remote sensing techniques, MODIS and digital camera images. Based on the vegetation indices extracted from each measurement, we analyzed phenological changes of vegetation and deduced phenophases transition dates, such as start dates of green-up and senescence by applying different two methods, the rate of change of curvature, K and HMV. The start and end of the growing season of Mongolian oak expressed in ExG–DC, which were extracted from digital camera image, were agreed well with that of visual assessment. EVI among three vegetation indices (EVI, NDVI and ExG–MI) showed the high correlation with ExG–DC and visual assessment. Based on RMSE, the transition dates assessed by visual observation were agreed better with the dates, which were estimated based on curvature K than with the dates estimated from HMV in all vegetation indices. Sap flow time-series estimates for the phenological transition dates were closely accorded with the estimates derived from near-surface time-series, and coincided better with the dates estimated based on curvature K than that based on HMV. In conclusion, based on the result of this study, we suggest that it is effective to use EXG–DC obtained from digital camera and EVI from MODIS when these two instrument are integrated as the vegetation indices and that curvature K is an effective method for extracting the phenological event dates of vegetation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Long-term phenological observations from North America, Europe, and Asia provide indisputable evidence that climate change (particularly recent warming trends) is affecting the timing of life-cycle events of species from a diverse range of taxonomic groups (Peñuelas et al. 2002; Parmesan 2006; Schwartz et al. 2006; Cleland et al. 2007; Richardson et al. 2009). At the same time, experimental studies show how other global change factors (e.g. elevated CO2 and N deposition) can also influence phenology (Cleland et al. 2006). The recent establishment of the USA National Phenology Network (USA-NPN; https://www.usanpn.org/), ‘citizen science’ efforts such as ProjectBudBurst (http://budburst.org/), and the GLOBE phenology project (Gazal et al. 2008) all suggest a need for better documentation of biological responses to a changing world and highlight the importance of phenological monitoring to achieve this goal (Morisette et al. 2009).
Many factors can affect vegetation phenology, which involves budburst, flowering, and leaf senescence. These factors include seasonal variations, such as climate, day-length, and nutrients, as well as plant species and age. Recent climate change has strongly affected the trends of phenological events (Peñuelas and Filella 2001; Walther et al. 2002, Ovaskainen et al. 2013).
Traditionally, assessment of changes in the phenology has relied on field measurements, often by volunteers and amateur naturalists, who record discrete events, such as flowering, leaf emergence, and other characteristics, that depend on observation and site location (Richardson et al. 2007). Traditional plant phenology observations have involved recording the date of occurrence for key events, including first leaf opening, first flowering, full bloom, and end of bloom, by repeated visual observations. Capturing the exact date of key events requires daily observations during the growing season. Therefore, phenology observations are relatively labor intensive and costly (Sparks et al. 2006) to collect data. Although these observations are a valuable source of information, they are limited in spatial coverage and use a range of methodologies conducted by a large number of personnel with variable training and skill levels, resulting in highly variable data consistency and quality (Coops et al. 2012).
As an alternative to traditional field-based measurements, a number of remote-sensing techniques have been developed to observe vegetation cycles throughout the year. New information is being gained through increased use of inexpensive visible spectrum digital cameras at the stand level, as phenological events can be monitored on the local scale (Woebbecke et al. 1995; Graham et al. 2010; Ide and Oguma 2010). Time-lapse photography at daily or hourly intervals provides very accurate temporal sampling for assessing vegetation phenology. Furthermore, additional data from an intermediate scale of observation can be obtained by mounting these systems on higher objects, such as towers or platforms. These techniques provide a spectrum of data ranging from field-based observations to satellite-derived estimates (Coops et al. 2012).
At the satellite level, field-based assessments have been successfully linked to multi-spectral images of different spatial resolutions. For instance, estimates of vegetation phenology have been compared with data acquired from the National Oceanic and Atmospheric Administration (NOAA) AVHRR (Advanced Very High Resolution Radiometer) (Schwartz and Reiter 2000; Studer et al. 2007), and more recently the MODIS (MODerate-resolution Imaging Spectroradiometer) instrument onboard the Terra and Aqua platforms (Fisher and Mustard 2007; Soudani et al. 2008).
A fundamental challenge faced when using remote-sensing techniques to monitor vegetation phenology is the trade-off required between the level of spatial detail and the revisit time provided by the sensor. For instance, although the data from the Landsat series of satellites have been successfully used to map vegetation with a 30-m spatial resolution, its 16-day temporal resolution is often not sufficient for timely observation of changes in vegetation and landscape characteristics throughout the year (Gao et al. 2006). This deficiency is even greater in areas with persistent cloud cover as there are longer intervals between clear imagery suitable for analysis. In contrast, satellites with higher revisit rates often do not provide a sufficient level of spatial detail, making species-specific phenological events difficult to ascertain and extract (Botta et al. 2000). In order to solve those problems, studies, which create new product by integrating more than two satellite images are carried out such as MODIS-Landsat fusion algorithm (Walker et al. 2012, 2014; Viña et al. 2016) via STARFM (spatial and temporal adaptive reflectance fusion mode).
One way to address this challenge is to integrate remotely sensed observations obtained at different spatial and temporal scales to provide information that contains both a high temporal density and fine spatial scale observations. At the stand level, images obtained from transects of phenological cameras (Bater et al. 2011) can be directly linked to satellite observations to upscale fine-scale information on vegetation green-up and senescence to larger areas. As a result, these observations can reflect seasonal changes in vegetation phenology at a spatial resolution sufficient to provide detailed information on different vegetation zones and land-cover types (Hilker et al. 2009) and at time intervals with relevance to vegetation phenology. But as Hufkens et al. (2012) and Klosterman et al. (2014) point out, first of all, we have to test whether each method as a tool to monitor phenological trajectory of vegetation for observation is appropriate or not to integrate each method. If their appropriateness is proved, standard protocol to find out phenological event date in field from data obtained by each method has to be established in the next stage. In addition, we have to select the most appropriate vegetation index comparing difference and uncertainty among existing various vegetation indices.
This study aims to answer to the following questions: (1) Is it possible to integrate both methods with different spatiotemporal scales as a tool for phenological study? (2) What is a vegetation index appropriate for monitoring phenological event date significant ecologically? (3) Which method can infer start dates of green-up and senescence correctly from vegetation index?
In order to arrive at the goals, we compared measurements of vegetation phenology compiled from a network of ground-based cameras and satellite-derived measurements of vegetation conditions derived from a fused broad MODIS with field-based visual observation as a reference information. In addition, we measured sap flow as a tool to confirm leaf unfolding and senescence dates in a physiological level. Two key indicators of phenological activity were compared across a spectrum of elevations: (1) start date of green-up, (2) start date of senescence.
Materials and methods
Study area
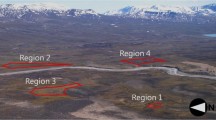
The phenological signal of canopy reflectance from deciduous broadleaved forests can be clearly defined, ensuring accurate measurement of the change in growing season. Therefore, we selected Mongolian oak (Quercus mongolica), a dominant species in the temperate deciduous broadleaved forest zone on the Korean Peninsula, as the target species of this study. The species belonging to the Quercus genus in Korea (Republic of), Q. mongolica grows at the highest elevation and thus, Q. mongolica would likely be a species sensitive to global warming. Four sites of Mt. Nam, Mt. Jeombong, Mt. Jiri, and Gwangneung, were selected for analysis. The sites, where the ecological tower was installed, has a large volume of basic ecological information available, and thus, was selected as the study sites (Fig. 1, Table 1). Those sites are LTER (long term ecological research) sites of Korea and this study was carried out as a LTER program. Because we captured crown layer of Q. mongolica stand, our research activity did not any damages for endangered species or protective species.
The climate of areas where this study was carried out, is the temperate of continental type. Mean annual temperature and precipitation for 30 years (1981–2010) in Seoul, Inje, Sancheong, Dongducheon where Mt. Nam, Mt. Jeombong, Mt. Jiri, and Gwangneung are located, represented 12.5 °C and 1450.5 mm, 10.1 °C and 1210.5 mm, 12.8 °C and 1556.6 mm, and 11.2 °C and 1502.9 mm, respectively. Among study sites, Mt. Nam and Gwangneung sites where are located on the urban center and suburban area, respectively, are under influences of urban heat island, whereas Mt. Jeombong and Mt. Jiri where are located on mountainous area with high elevation, are influenced by mountainous climate.
Digital camera and satellite image acquisition
We mounted commercial digital cameras (Model Ltl–6210 M, Little Acorn Outdoors, Denmark, WI, USA) near the top of each tower (30 m above ground), looking north and angled slightly downward, providing a view across the top of the canopy (Richardson et al. 2007, 2009). Each camera was set to record high-quality JPEG images three times per day (09:00, 12:30, and 16:00) using intervalometers. However, only the 12:30 images were used for the analysis to maintain consistency (Table 2).
Due to the shorter collection cycle and greater serial time clarity in the phenological study using satellite imagery, we utilized MODIS (MODerate–Resolution Imaging Spectroradiometer) 500-m resolution land surface imagery (MOD09A1) supplied at 8-day intervals as multi-spectral satellite images and MODIS 500-m resolution daily surface reflectance (MOD09GA). The MODIS is a payload scientific instrument placed in the Earth’s orbit by NASA in 1999 on board the Terra (EOS AM) Satellite. The MODIS functions well for monitoring environmental changes because the sensor incorporates enhanced atmospheric correction, cloud detection, improved georeferencing, and the enhanced ability to monitor vegetation (Kang et al. 2003; Kim et al. 2013). For the current study, MODIS scenes of the study area from 1/1/2014 to 12/31/2014 were downloaded and the phenological signals were extracted.
Near-surface image analysis
The annual cycle of vegetation phenology inferred from remote sensing is characterized by four key transition dates that define the key phenological phases of vegetation dynamics at annual time scales: (1) green-up, (2) maturity, (3) senescence, and (4) dormancy (Zhang et al. 2003; Richardson et al. 2007). Phenological signals maintain low values, but increase rapidly as the green-up (unfolding) phase begins. Signals no longer increase, but maintain a high value, when the leaves reach the mature phase. As the plants enter into the senescence stage, the signals decrease radically. As the dormancy phase begins, the signals return to the lowest value of the initial stage. In such a temporal phenological signal curve, transition dates correspond to the times at which the rate of change in curvature in the Vegetation Index (VI) exhibits local minima or maxima (Zhang et al. 2003) (Fig. 2). This method, proposed by Zhang et al. (2003), was used originally to model changes in VI data extracted from satellite images, but the same method can be applied for analysis of phenological signals from near-surface remote sensing (Crimmins and Crimmins 2008; Richardson et al. 2009; Bater et al. 2011; Ide and Oguma 2013; Klosterman et al. 2014). To extract phenological signals from images, the near-infrared band is used together with visible light bands, whereas only visible light bands are extracted from digital camera signals.
Regions of interest (ROI) are defined when we analyze phenological changes in vegetation using near-surface images (Walker et al. 2012). Because the images include a mixture of landscape, sky, and other features, ROI are restricted to the crown layer for extracting phenological signals from the images. Furthermore, the Q. mongolica stands are mixed with stands of Q. serrata, Q. variabills, and other species at the Mt. Jiri and Gwangneung sites. Therefore, ROI in this study were restricted to pure stands of Q. mongolica and avoided mountains and sky above the ROI and undergrowth on the forest floor (covered by snow in winter) below the ROI (Richardson et al. 2007, 2009) (Fig. 3).
To extract phenological signals, we collected images from the digital cameras periodically, and classified them into red, green, and blue bands. Using digital numbers for each band, we calculated the average excess green index (ExG; Eq. 5) for each ROI (Richardson et al. 2009; Hufkens et al. 2012). The ExG–DC (ExG in digital camera) compares the green band of the RGB image to the red and blue bands to derive excess greenness corrected for illumination differences (Nijland et al. 2013). The ExG–DC is an indicator sensitive to vegetation pigment and activity because the seasonal pattern of the ExG–DC correlates with gross primary production (GPP) (Richardson et al. 2009; Ide and Oguma 2010).
The VI was obtained using the smooth curve fitting method to remove variation and to gather trends because interpretation error can occur due to data errors and variation depending on weather conditions. In this study, the ExG–DC was smoothed to the 80th percentile using an exponentially weighted moving average (EWMA). The EWMA was defined as:
where, t is the day of year (DoY); St is the EWMA value at the DoY; Yt is the VI value at the DoY; and α is the smoothing coefficient.
In order to derive phenological event dates from phenological time-series data, we used a sigmoid-based equation (Zhang et al. 2003; Fisher and Mustard 2007; Richardson et al. 2009; Ide and Oguma 2010; Hufkens et al. 2012; Klosterman et al. 2014): In the sigmoid model, phenological transition dates were attained by obtaining local extrema in the rate of change of curvature K (hereafter, abbreviated as curvature K) (Zhang et al. 2003; Klosterman et al. 2014) and half maximum value which is identified as the steepest point on the sigmoid (hereafter, abbreviated as HMV) (Richardson et al. 2009; Ide and Oguma 2010; Nijland et al. 2013):
where, t is time (in days), c is the amplitude of increase or decrease in greenness, d is the dormant season baseline value, a and b control the lower and upper limits of the function (Richardson et al. 2009; Hufkens et al. 2012; Klosterman et al. 2014).
To validate the estimated transition dates based on the ExG, we used a visual observation method (Crimmins and Crimmins 2008; Ide and Oguma 2010; Sonnentag et al. 2012; Ide and Oguma 2013; Walker et al. 2014). For example, the green-up date was estimated as the first day when most of the ROI were tinged with light green (Ide and Oguma 2010; Walker et al. 2012).
Analysis of satellite images
The MOD09A1 and MOD09GA datasets are comprised of seven bands, including visible light bands and near-infrared bands. Three VIs (EVI: Enhanced Vegetation Index; NDVI: Normalized Difference Vegetation Index; ExG–MI: ExG in MODIS) were calculated using red (Band 1: 620–670 µm), green (Band 4: 545–565 µm), blue (Band 3: 459–479 µm) and near-infrared (Band 2: 841–876 µm) based on the equations:
where ρNIR, ρRED, ρGREEN, ρBLUE are value in near infrared, red, green, blue band, respectively. L is the canopy background adjustment (L = 1), C1 and C2 are the coefficients (C1 = 6, C2 = 7.5), G is the gain factor (G = 2.5).
The ExG–DC was calculated based on information obtained from digital cameras and the NDVI and EVI were based on information from MODIS images. The ExG–MI was calculated to directly compare data from MODIS images with those from the digital cameras (Hufkens et al. 2012). For MODIS data analysis, we followed the same procedures used for analysis of the digital camera data.
Because the collected data used a sinusoidal projection, we reprojected the data onto Transverse Mercator (TM) coordinates and saved them in Geotif format. The South Korea zone was clipped from the Geotif data by masking and rearranged to be distributed in the range of − 1 to 1 by a multiplied scale factor.
The VI for each site based on the respective MODIS image was obtained from the mean VIs extracted from ROI clipped by masking relevant areas in the VI map prepared from the MODIS image. The annual cycle of vegetation phenology inferred from MODIS images showed the same pattern as that from the near-surface images.
To compare the phenological transition dates different in observation methods (near-surface and satellite remote sensing time series) for phenology and treatment tools of phenological data (curvature K and HMV), the root mean square error (RMSE) was calculated.
Sap flow measurement
To elucidate phenological event dates significantly more, we collected phenological data in another level from sap flow measurement (model SFM1 Sap Flow Meter, ICT international, Armidale, Australia) installed in the Mt. Jeombong site from April to November, 2015. Mongolian oak individual for measuring sap flow were selected randomly from individuals included in ROI of digital camera (DBH, average bark width, sapwood width, heartwood diameter of individuals selected were 20, 3, 1, and 6, respectively). Sap flow velocity (cm3 h−1) was calculated from temperature and heat pulse measured by thermistor inserted in 7.5 and 22.5 mm within stock removed bark (Burgess et al. 2001).
where, Vsi is the sap velocity (cm h−1) at the position i, k the thermal diffusivity (cm2·s−1), 0.0025 the reference thermal diffusivity (cm2 s−1), B the wound correction factor (–), ρb the basic density of wood (sapwood dry weight/sapwood fresh volume, kg m−3), cw the specific heat capacity of the wood matrix (1200 J kg−1 °C−1), ρs the density of water (1000 kg m−3), mc the water content of sapwood (sapwood fresh weight-sapwood dry weight/sapwood dry weight, kg kg−1) and Vhi the measured heat pulse velocity (cm h−1) at position i (Burgess et al. 2001).
Transition date of sap flow was determined from curvature K (Formula 3) of seasonal trajectory of sap flow obtained based on daily sap velocity. Transition dates of sap flow were compared with phenological transition dates obtained from digital camera installed in the same site.
Results
Performance of digital camera as a monitoring tool of phenology
The ExG–DC calculated from data obtained from digital cameras set in the study sites clearly indicated seasonal changes reflecting phenological trajectory of the Mongolian oak stand at each site (Fig. 4). Start dates of green-up determined from curvature K were earlier in the order of Mt. Nam, Gwangneung, Mt. Jiri, Mt. Jeombong and start dates of senescence were vice versa (Table 3). Phenological event dates determined from HMV showed the same trend (Table 3). Phenological event dates determined from curvature K were usually earlier than those dates observed by eye, meanwhile, the results determined from HMV were later compared with dates observed by eye (Fig. 5 and Table 3).
Logistic models of spring green-up (a) and autumn senescence (b) in study sites. Closed circles indicate the times at which the rate of change in curvature exhibits local maxima (a) or minima (b). Open circles indicate the times at which ExG–DC value exhibits HMV in each site (ExG–DC ExG in digital camera, DoY day of year, HMV half maximum value)
Performance of different indices based on MODIS
Seasonal changes in EVI, NDVI and ExG–MI obtained from MODIS09A1 and MOD09GA were shown in Figs. 6, 7, and 8. Phenological event dates were different depending on vegetation indices, but green-up start dates from all VIs and methods were earlier in the order of Mt. Nam, Gwangneung, Mt. Jiri, and Mt. Jeombong and senescence start date showed the antagonistic trend (Table 4).
Deviation among sites compared with visual observation in green-up start was the smallest in EVI and ExG–MI equally and that in senescence start was the smallest in EVI based on curvature K (Table 4). Deviation among sites compared with visual observation in green-up start was the smallest in NDVI and that in senescence start was the smallest in EVI based on HMV (Table 4). Compared between curvature K and HMV, deviation in green-up start was smaller in the latter than that in the former, whereas the result in the senescence start was the vice versa (Table 4).
Comparison of the performance between digital camera and MODIS, and curvature K and HMV
Transition dates assessed by visual observation and ExG–DC showed the highest consistency as RMSEs between both dates are 2.60 and 2.74 for green up and senescence dates, respectively. Among the vegetation indices derived from MODIS, EVI had the highest consistency with visual assessment (3.71 and 4.85) and ExG–DC (3.87 and 2.55). Transition dates assessed by ExG–DC showed a little higher consistency (2.60 and 2.74) than them by EVI (3.71 and 4.85) compared with visual assessment.
The results based on HMV showed the same trend compared with visual observation but showed a little difference compared with ExG–DC. Compared between curvature K and HMV based on root mean squared error (RMSE), the transition dates estimated based on the former were agreed better with the dates by visual observation than with the dates estimated from the latter (Table 5).
Relationship between leaf phenology and sap flow
Seasonal change of sap flow was shown in Figs. 9 and 10. Incline start date of sap flow velocity was shown in the 119th day of year in spring. The date was 2 days later than green-up start date based on curvature K and earlier 4 days compared with that assessed by HMV (Fig. 9). Decline start date was the 282nd day of year. The date was earlier 2 and 9 days than senescence start dates based on curvature K and HMV, respectively (Fig. 10).
Logistic model of spring green-up (a) and sap flow velocity during the same period (b) obtained from Mongolian oak stand of Mt. Jeombong in 2015. Closed circle and square in (a) are green–up start dates derived from curvature K and HMV and open circle indicates incline start date of sap velocity. c Indicates changes of curvature in logistic models derived from sap velocity [dotted line in (b)]. Error bars in (b) indicate standard error of daily sap flow velocity (ExG–DC ExG in digital camera, DoY Day of year, HMV half maximum value)
Logistic model of autumn senescence (a) and sap flow velocity during the same period (b) obtained from Mongolian oak stand of Mt. Jeombong in 2015. Closed circle and square in (a) are senescence start dates derived from curvature K and HMV and open circle indicates decline start date of sap velocity. c Indicates changes of curvature in logistic models derived from sap velocity [dotted line in (b)]. Error bars in (b) indicate standard error of daily sap flow velocity (ExG–DC ExG in digital camera, DoY day of year, HMV half maximum value)
Discussion
Phenological studies as a diagnostic tool for climate change
In this study, green-up of Q. mongolica stands initiated on different days of the year at the different sites studied, in the order of Mt. Nam, Gwangneung, Mt. Jiri, and Mt. Jeombong, whereas the order in which senescence occurred was reversed (Tables 3 and 4). Because there are differences in longitude, latitude, and altitude, the dates were corrected by applying the linear regression equations, which were about the relationship between phenology dates and geographic location (Klosterman et al. 2014). In addition, to clarify the differences among sites due to artificial interference, the dates were compared based on the dates of the Mt. Jeombong site, where maintains a pure stand of Q. mongolica and is escaped from artificial interferences and it is designated as a reserve by Korea Forest Service (Table 6). The observed green-up start date was earlier than the expected date in the Mt. Nam and Gwangneung sites, whereas the opposite was observed for the Mt. Jiri site. The earlier date of green-up initiation at the Mt. Nam and Gwangneung sites could be due to the intensive land use near those sites. Mt. Nam is located in the Seoul metropolitan area, which is the most densely developed area in Korea, and Gwangneung is also near this site. Those sites had higher air temperatures than the surrounding rural areas due to urban heat island effect (Zhang et al. 2004; Lee et al. 2007; Cho et al. 2009). On the other hand, the Mt. Jiri site that showed late unfolding is topographically located in a ravine. Valleys, such as where the Mt. Jiri site is located, have lower air temperatures than other topographical locations due to micrometeorological factors, such as cold air drainage flow (Pypker et al. 2007).
Utility of digital cameras in phenology observation
In this study, phenological variation in the greenness of dominant natural vegetation was derived from digital image analysis (Fig. 4). The phenological trajectory of ExG–DC reflected the seasonal course of vegetation. The estimated green-up dates agreed closely with the dates when leaf emergence became visible in photos. In addition, the findings of this study were similar to phenological timings identified in previous studies based on field observations reported at Long Term Ecological Research (LTER) sites in Korea (http://www.knlter.net). Therefore, the start and end of the growing season of Mongolian oak expressed as ExG–DC accorded well with that of field observations. In fact, the seasonal pattern of ExG–DC correlated well with that of GPP (Richardson et al. 2009), and thus, annual GPP could be estimated using digital image analysis (Ide and Oguma 2010).
In the present study, the annual phenological trajectories of Mongolian oak stands were discriminated as well. The ExG–DC was sensitive to vegetation change, producing high correlations that were superior to those achieved using individual normalized bands or other indices (Walker et al. 2012). In this application, we focused our initial analysis on green-up and senescence events, which are essentially the development and loss of green vegetation, respectively, within the field of view. Digital camera technology has become a common methodology for monitoring vegetation, and other researchers have correlated responses with fluxes related to carbon and water, such as CO2 exchange (Richardson et al. 2009), leaf area index, and the fraction of absorbed photosynthetically active radiation (Ide and Oguma 2010).
The development of inexpensive, commercially available systems, such as the one described in this study, will be critical to improving our understanding of seasonal variations in vegetation phenology, the detection of changing vegetation dynamics for biodiversity and wildlife management, and the calibration of datasets obtained from other remote-sensing technologies (Walker et al. 2012). Additional instrumentation co-located with the camera network would be required to investigate these additional aspects of forest function. However, information on the timing and location of vegetation phenological phases is critical to understanding the behavior and habitat usage of animals, such as grizzly bears (Walker et al. 2012). We believe that the application of these systems for biodiversity-related issues is a logical and important extension of ongoing work.
Integration of near-surface and satellite-based approaches for phenological study
Greenness is often calculated as a broadband NDVI (Huemmrich et al. 1999; Jenkins et al. 2007). However, an NDVI calculation requires the measurement of near-infrared light, a band that is not collected by RGB cameras (Crimmins and Crimmins 2008). In this study, phenological variations in the greenness of natural vegetation were derived from digital cameras and satellite images (MODIS). The start and end of the growing season of Mongolian oak stands, expressed as ExG, are correlated well with those of visual observations at the same site (Table 3). Among the VIs derived from MODIS, EVI had the highest correlation with ExG–DC in temporal dynamics and was also consistent with the findings of Hufkens et al. (2012) and Klosterman et al. (2014). In fact, Turner et al. (2003) suggested that they might predict the onset of green-up earlier than visual observations of the field, because many MODIS 8-day composites are derived from maximum values. However, the temporal resolution of the MODIS 8-day composites, which is dictated by the frequency of missing data caused by clouds in many parts of the world, do not support precise characterization by rapid leaf emergence and development during spring (Kang et al. 2003). On the other hand, although use of the MODIS daily surface reflectance could provide the enhanced temporal detection of MODIS-based analysis at a daily time step, this advantage can be constrained by daily cloud contamination (Kang et al. 2003). In this study, we resolved both spatial and temporal drawbacks by applying a derivation method compared with spline curves of VIs that were derived from both MODIS 8-day composites and MODIS daily surface reflectance. In this study, RMSE between ExG–DC and EVI was shown in about 3 days. The difference was smaller about 9 days than that derived from PhenoCam network installed in USA (Klosterman et al. 2014). The difference would be due to the fact that we selected pure stand of Mongolian oak for photographing differently from PhenoCam Network, which selected mixed forest (Tierney et al. 2013). In addition, Mongolian oak begins leaf unfolding and senescence the earliest among deciduous oaks growing in Korea. Therefore, it is estimated that Mongolian oak could be the best species to derive phenological transition date from MODIS image with low spatial resolution. In fact, deviation between greenness derived near-surface phenology and remote-sensing derived phenology was the biggest in Gwangneung site where Q. serrata is intermingled in the Mongolian oak stand (Tables 3 and 4).
Selection of method to estimate phenological event date
In the study on vegetation phenology, fluctuation of transition dates is significant phenomenologically, but more important in the physiological response of plant and biogeochemical cycle. In particular, phenological event dates such as unfolding or senescence of leaf determine the periods of transpiration and photosynthesis, and consequently influence on the yearly variation of energy, water, and carbon fluxes (Yoshifuji et al. 2011). In this respect, selecting criteria that reflects transition events well occupy the central position in the study for monitoring phenology using vegetation index (Hufkens et al. 2012). We determined phenological transition dates by using two methods in this study. The method using changes of curvature has usually used as the main algorithm to determine phenological transition dates from satellite image interpretation like MODIS Global Vegetation Phenology Product (MOD12Q2) (Roger et al. 2015), whereas has rarely used in greenness derived near-surface phenology research (e.g. Klosterman et al. 2014). HMV has usually used in near-surface technique (Fisher and Mustard 2007; Richardson et al. 2009; Ide and Oguma 2010; Coops et al. 2012; Hufkens et al. 2012; Walker et al. 2012; Nijland et al. 2013). In this method, the date of leaf onset is defined as the date at which the sigmoid curve reaches its HMV (Richardson et al. 2009). In other words, the HMV is the steepest point on the symmetric sigmoid or the peak of the first derivative and can be envisioned as representing the date when most leaves are likely to emerge (Fisher and Mustard 2007). Transition dates are determined as the local extrema in curvature K method, while, in HMV method, the dates are determined as the point, in which curvature close to 0 (Fig. 11).
The changes of curvature in logistic models derived from ExG–DC in study sites (a green–up stage, b senescence stage). Closed circles indicate the times at which the rate of change in curvature exhibits local maxima (a) or minima (b). Open circles indicate the times at which ExG–DC value HMV in each site (ExG–DC ExG in digital camera, DoY day of year, HMV half maximum value)
In this study, we supposed that green-up stage was begun when Mongolian oak stands started leafing out, and senescence stage was done when the canopy began to change color at first in the fall. Therefore, in visual assessment, the transition dates of green-up and senescence were determined as times at which those phenological phenomena can be verified visibly in the photography. Although the difference between the expected dates derived from each index and the dates based on observation by eyes not consistent (Table 4), variation of transition dates from curvature K was less than that from HMV (Table 5). From this result, we estimated that curvature K index is more sensitive to the phenological phenomenon that we plan to monitor rather than HMV index.
According to numerous studies, which investigated water relation along the annual changes of phenology (Jackson and Bliss 1984; Seghieri et al. 1995; Rojas-Jiménez et al. 2007), sap flow is closely related to change of leaf area (Urban et al. 2013). Sap flow is tightly connected to the phenological stage of the tree (Urban et al. 2013) due to the fact that tree has to progress transpiration to participate in forming the leaves and follow-up radial growth with leaves development (Střelcová et al. 2006). The transition dates in sap flow seasonality accorded better with the dates extracted by using curvature K method (2 days in mean deviation) than the dates extracted by using HMV method (about a week in mean deviation). In this respect, the phenological event dates extracted from curvature K method in this study, show higher reliability than those from HMV method. Moreover, Hufkens et al. (2012) pointed out that the ecological meaning of the HMV is ambiguous.
Conclusion
In this study, phenological responses of plants reflected the climate of the local areas where they grow. The results suggest that the continuous monitoring of vegetation phenology could be used as an effective diagnostic tool for the ongoing climate change. In this respect, it was proposed in this study that an effective study for vegetation phenology could be carried out by selecting appropriate monitoring instrument and integrating appropriate vegetation indices. In the results of this study, transition date of phenology extracted from digital camera image interpretation were accorded with that of visual assessment but it has limitations for wide–area applications as it is a point based data source. Conversely, information obtained from MODIS image are useful for monitoring the extensive area but it is inappropriate for monitoring variation among species of vegetation phenology as an area based data source. It is deemed that integrating two monitoring tools could resolve this problem. Further, based on the result of this study, we suggest that it is effective to use ExG–DC obtained from digital camera and EVI from MODIS when these two instrument are integrated as the vegetation indices and that curvature K is an effective method for extracting the phenological event dates of vegetation. This result is novel and advanced one as it is also proved from physiological response such as sap flow differently from existing phenological research, which is usually focused on external response.
References
Bater CW, Coops NC, Wulder MA, Nielsen SE, McDermid G, Stenhouse GB (2011) Design and installation of a camera network across an elevation gradient for habitat assessment. Instrum Sci Technol 39:231–247
Botta A, Viovy N, Ciais P, Friedlingstein P, Monfray P (2000) A global prognostic scheme of leaf onset using satellite data. Glob Chang Biol 6:709–725
Burgess SS, Adams MA, Turner NC, Beverly CR, Ong CK, Khan AA, Bleby TM (2001) An improved heat pulse method to measure low and reverse rates of sap flow in woody plants. Tree Physiol 21:589–598
Cho YC, Cho HJ, Lee CS (2009) Greenbelt systems play an important role in the prevention of landscape degradation due to urbanization. J Ecol Environ Sci 32:207–217
Cleland EE, Chiariello NR, Loarie SR, Mooney HA, Field CB (2006) Diverse responses of phenology to global changes in a grassland ecosystem. Proc Natl Acd Sci USA 103:13740–13744
Cleland EE, Chuine I, Menzel A, Mooney HA, Schwartz MD (2007) Shifting plant phenology in response to global change. Trends Ecol Evol 22:357–365
Coops NC, Hilker T, Bater CW, Wulder MA, Nielsen SE, McDermid G, Stenhouse G (2012) Linking ground-based to satellite-derived phenological metrics in support of habitat assessment. Remote Sens Lett 3:191–200
Crimmins MA, Crimmins TM (2008) Monitoring plant phenology using digital repeat photography. Environ Manag 41:949–958
Fisher JI, Mustard JF (2007) Cross–scalar satellite phenology from ground, Landsat, and MODIS data. Remote Sens Environ 109:261–273
Gao F, Masek J, Schwaller M, Hall F (2006) On the blending of the Landsat and MODIS surface reflectance: predicting daily Landsat surface reflectance. IEEE Trans Geosci Remote Sens 44:2207–2218
Gazal R, White MA, Gillies R, Rodemaker E, Sparrow E, Gordon L (2008) GLOBE students, teachers, and scientists demonstrate variable differences between urban and rural leaf phenology. Glob Chang Biol 14:1568–1580
Graham EA, Riordan EC, Yuen EM, Estrin D, Rundel PW (2010) Public Internet-connected cameras used as a cross-continental ground-based plant phenology monitoring system. Glob Chang Biol 16:3014–3023
Hilker T, Wulder MA, Coops NC, Seitz N, White JC, Gao F, Masek JG, Stenhouse G (2009) Generation of dense time series synthetic Landsat data through data blending with MODIS using a spatial and temporal adaptive reflectance fusion model. Remote Sens Environ 113:1988–1999
Huemmrich KF, Black TA, Jarvis PG, McCaughey J, Hall FG (1999) High temporal resolution NDVI phenology from micrometeorological radiation sensors. J Geophys Res Atmos Atmos 104:27935–27944
Hufkens K, Friedl M, Sonnentag O, Braswell BH, Milliman T, Richardson AD (2012) Linking near-surface and satellite remote sensing measurements of deciduous broadleaf forest phenology. Remote Sens Environ 117:307–321
Ide R, Oguma H (2010) Use of digital cameras for phenological observations. Ecol Infom 5:339–347
Ide R, Oguma H (2013) A cost–effective monitoring method using digital time–lapse cameras for detecting temporal and spatial variations of snowmelt and vegetation phenology in alpine ecosystems. Ecol Infom 16:25–34
Jackson LE, Bliss L (1984) Phenology and water relations of three plant life forms in a dry tree-line meadow. Ecology 65:1302–1314
Jenkins J, Richardson AD, Braswell B, Ollinger SV, Hollinger DY, Smith M-L (2007) Refining light–use efficiency calculations for a deciduous forest canopy using simultaneous tower–based carbon flux and radiometric measurements. Agric For Meteorol 143:64–79
Kang S, Runniing SW, Lim JH, Zhao M, Park CR, Loehman R (2003) A regional phenology model for detecting onset of greenness in temperate mixed forests, Korea: an application of MODIS leaf area index. Remote Sens Environ 86:232–242
Kim N-S, Lee H-C, Cha J-Y (2013) A study on changes of phenology and characteristics of spatial distribution using MODIS images. J Korea Soc Environ Restor Technol 16:59–69
Klosterman S, Hufkens K, Gray J, Melaas E, Sonnentag O, Lavine I, Mitchell L, Norman R, Friedl M, Richardson A (2014) Evaluating remote sensing of deciduous forest phenology at multiple spatial scales using PhenoCam imagery. Biogeosciences Discuss 11:2305–2342
Lee C-S, Song H-G, Kim H-S, Lee B, Pi J-H, Cho Y-C, Seol E-S, Oh W-S, Park S-A, Lee S-M (2007) short communication: which environmental factors caused lammas shoot srowth of Korean red pine? J Ecol Environ Sci 30:101–105
Morisette JT, Richardson AD, Knapp AK, Fisher JI, Graham EA, Abatzoglou J, Wilson BE, Breshears DD, Henebry GM, Hanes JM (2009) Tracking the rhythm of the seasons in the face of global change: phenological research in the 21st century. Front Ecol Environ 7:253–260
Nijland W, Coops N, Coogan S, Bater C, Wulder M, Nielsen S, McDermid G, Stenhouse G (2013) Vegetation phenology can be captured with digital repeat photography and linked to variability of root nutrition in Hedysarum alpinum. Appl Veg Sci 16:317–324
Ovaskainen O, Skorokhodova S, Yakovleva M, Sukhov A, Kutenkov A, Kutenkova N, Shcherbakov A, Meyke E, del Mar Delgado M (2013) Community-level phenological response to climate change. Proc Natl Acd Sci USA 110:13434–13439
Parmesan C (2006) Ecological and evolutionary responses to recent climate change. Annu Rev Ecol Evol Syst 37:637–669
Peñuelas J, Filella I (2001) Responses to a warming world. Science 294:793–795
Peñuelas J, Filella I, Comas P (2002) Changed plant and animal life cycles from 1952 to 2000 in the Mediterranean region. Glob Chang Biol 8:531–544
Pypker T, Unsworth MH, Lamb B, Allwine E, Edburg S, Sulzman E, Mix A, Bond B (2007) Cold air drainage in a forested valley: investigating the feasibility of monitoring ecosystem metabolism. Agric For Meteorol 145:149–166
Richardson AD, Jenkins JP, Braswell BH, Hollinger DY, Ollinger SV, Smith M-L (2007) Use of digital webcam images to track spring green-up in a deciduous broadleaf forest. Oecologia 152:323–334
Richardson AD, Braswell BH, Hollinger DY, Jenkins JP, Ollinger SV (2009) Near-surface remote sensing of spatial and temporal variation in canopy phenology. Ecol Appl 19:1417–1428
Roger JC, Vermote EF, Ray J (2015) MODIS surface reflectance user's guide. Collection 6. MODIS Land Surface Reflectance Science Computing Facility. http://modisland.gsfc.nasa.gov/pdf/MOD09_UserGuide_v1.4.pdf. Accessed 15 Dec 2015
Rojas-Jiménez K, Holbrook NM, Gutiérrez-Soto MV (2007) Dry-season leaf flushing of Enterolobium cyclocarpum (ear-pod tree): above–and belowground phenology and water relations. Tree Physiol 27:1561–1568
Schwartz MD, Reiter BE (2000) Changes in north American spring. Int J Climatol 20:929–932
Schwartz MD, Ahas R, Aasa A (2006) Onset of spring starting earlier across the Northern Hemisphere. Glob Change Biol 12:343–351
Seghieri J, Floret C, Pontanier R (1995) Plant phenology in relation to water availability: herbaceous and woody species in the savannas of northern Cameroon. J Trop Ecol 11:237–254
Sonnentag O, Hufkens K, Teshera-Sterne C, Young AM, Friedl M, Braswell BH, Milliman T, O’Keefe J, Richardson AD (2012) Digital repeat photography for phenological research in forest ecosystems. Agric For Meteorol 152:159–177
Soudani K, le Maire G, Dufrêne E, François C, Delpierre N, Ulrich E, Cecchini S (2008) Evaluation of the onset of green-up in temperate deciduous broadleaf forests derived from Moderate Resolution Imaging Spectroradiometer (MODIS) data. Remote Sens Environ 112:2643–2655
Sparks T, Huber K, Croxton P (2006) Plant development scores from fixed–date photographs: the influence of weather variables and recorder experience. Int J Biometeorol 50:275–279
Střelcová K, Priwitzer T, Minďáš J (2006) Fenologické fázy a transpirácia buka lesného v horskom zmiešanom lese. Fenologická odezva proměnlivosti podnebí Brno, Czech Republic
Studer S, Stöckli R, Appenzeller C, Vidale PL (2007) A comparative study of satellite and ground–based phenology. Int J Biometeorol 51:405–414
Tierney G, Mitchell B, Miller–Rushing A, Katz J, Denny E, Brauer C, Donovan T, Richardson AD, Toomey M, Kozlowski A (2013) Phenology monitoring protocol: Northeast Temperate Network. National Park Service
Turner DP, Ritts WD, Cohen WB, Gower ST, Zhao M, Running SW, Wofsy SC, Urbanski S, Dunn AL, Munger J (2003) Scaling gross primary production (GPP) over boreal and deciduous forest landscapes in support of MODIS GPP product validation. Remote Sens Environ 88:256–270
Urban J, Bednářová E, Plichta R, Kučera J (2013) Linking phenological data to ecophysiology of European beech IX International Workshop on Sap Flow 991, pp.293–299
Viña A, Liu W, Zhou S, Huang J, Liu J (2016) Land surface phenology as an indicator of biodiversity patterns. Ecol Indic 64:281–288
Walker J, De Beurs K, Wynne R, Gao F (2012) Evaluation of Landsat and MODIS data fusion products for analysis of dryland forest phenology. Remote Sens Environ 117:381–393
Walker J, De Beurs K, Wynne R (2014) Dryland vegetation phenology across an elevation gradient in Arizona, USA, investigated with fused MODIS and Landsat data. Remote Sens Environ 144:85–97
Walther G-R, Post E, Convey P, Menzel A, Parmesan C, Beebee TJ, Fromentin J-M, Hoegh-Guldberg O, Bairlein F (2002) Ecological responses to recent climate change. Nature 416:389–395
Woebbecke D, Meyer G, Von Bargen K, Mortensen D (1995) Color indices for weed identification under various soil, residue, and lighting conditions. Trans ASAE 38:259–269
Yoshifuji N, Komatsu H, To Kumagai, Tanaka N, Tantasirin C, Suzuki M (2011) Interannual variation in transpiration onset and its predictive indicator for a tropical deciduous forest in northern Thailand based on 8-year sap-flow records. Ecohydrology 4:225–235
Zhang X, Friedl MA, Schaaf CB, Strahler AH, Hodges JC, Gao F, Reed BC, Huete A (2003) Monitoring vegetation phenology using MODIS. Remote Sens Environ 84:471–475
Zhang X, Friedl MA, Schaaf CB, Strahler AH, Schneider A (2004) The footprint of urban climates on vegetation phenology. Geophys Res Lett 31:L12209. https://doi.org/10.1029/2004G
Acknowledgements
The research was supported by a Grant from ‘Conservation Project of Threatened Plants for Climate Change’ of Korea National Arboretum.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Lim, C.H., An, J.H., Jung, S.H. et al. Ecological consideration for several methodologies to diagnose vegetation phenology. Ecol Res 33, 363–377 (2018). https://doi.org/10.1007/s11284-017-1551-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-017-1551-3