Abstract
A new synthesis of ethynyldimethoxytriazine 1, an important platform-compound for developing new chemical entities for anticancer research and for other biological applications, is described. Compound 1 was further reacted with azides 5a–i to provide triazin–triazoles 2a–i, which were tested on human farnesyltransferase and on the NCI-60 human tumor cell lines. Synthesis of other dimethoxytriazine derivatives 15 and 16, linked to a sp 2 or a sp 3 carbon atom were also studied.
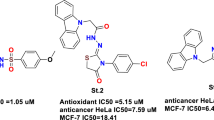
Graphical Abstract







Similar content being viewed by others
Notes
CCDC-1045377 (for zinc derivative 3b) and CCDC-1045376 (for phenothiazine derivative 16) contain the crystallographic data for this contribution. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
Acids 7a–d were obtained by saponification of the corresponding known esters.
For additional proof on the electronic behavior of acetylene group of compound 1, see [65].
For activating groups in accelerating reactivity of compounds bearing a triple bond group, see [66].
References
R. Kumar, L. Gupta, P. Pal, S. Khan, N. Singh, S.B. Katiyar, S. Meena, J. Sarkar, S. Sinha, J.K. Kanaujiya, S. Lochab, A.K. Trivedi, P.M.S. Chauhan, Eur. J. Med. Chem. 45, 2265 (2010)
B.B. Baldaniya, P.K. Patel, Eur. J. Chem. 6, 673 (2009)
L. Almeida, B. Aquila, C.E. Chuaqui, H.P. Guan, S. Huang, S. Ioannidis, M. Lamb, B. Peng, J. Shi, M. Su, Q.B. Su, WO2009016410 (2009)
L. Almeida, B. Aquila, C.E. Chuaqui, H.P. Guan, S. Huang, S. Ioannidis, M. Lamb, B. Peng, J. Shi, M. Su, Q.B. Su, Chem. Abstr. 153, 456713 (2010)
N.M. Niyaz, K.A. Guenthenspberger, R. Hunter, A.V. Brown, J.S. Nugent, US 20090093481 (2009)
N.M. Niyaz, K.A. Guenthenspberger, R. Hunter, A.V. Brown, J.S. Nugent, Chem. Abstr. 150, 398582 (2009)
B. Liu, Y. Lee, J.M. Zou, H.M. Petrassi, R.W. Joseph, W.C. Chao, E.L. Michelotti, M. Bukhtiyarova, E.B. Springman, B.D. Dorsey, Bioorg. Med. Chem. Lett. 20, 6592 (2010)
A. Solankee, K. Kapadia, A. Ciric, M. Sokovic, I. Doytchinova, A. Geronikaki, Eur. J. Med. Chem. 45, 510 (2010)
P.K. Pareek, P.D. Mithlesh, M. Chaudhary, A. Pareek, R. Kant, K.G. Ojha, Main Group Chem. 10, 63 (2011)
H. Ojha, P. Gahlot, A.K. Tiwari, M. Pathak, R. Kakkar, Chem. Biol. Drug Des. 77, 57 (2011)
C. Fanelli, A.A. Fabbri, S. Monti, R. Menicagli, D. Pini, S.M. Rapaccini, S. Samaritani, P. Salvadori, 7th International Congress of Plant Pathology (Edinburgh, 1998)
S. Samaritani, A.A. Fabbri, C. Fanelli, R. Menicagli, P. Salvadori, XXV Convegno Nazionale della Divisione di Chimica Organica (Folgaria, 1998)
A. Ricelli, A.A. Fabbri, C. Fanelli, R. Menicagli, S. Samaritani, D. Pini, S.M. Rapaccini, P. Salvadori, Restaurator 20, 97 (1999)
G.H. Singhal, H. Roebke, DE 2115318 A (1971)
G.H. Singhal, H. Roebke, Chem. Abstr. 76, 72560 (1972)
M. Just, I. Glase, DD 248590 (1987)
M. Just, Chem. Abstr. 108, 131864 (1988)
K. Koizumi, O. Yamashita, K. Wakabayashi, K. Tomono, H. Sasayama, WO 9720825 (1997)
K. Koizumi, O. Yamashita, K. Wakabayashi, K. Tomono, H. Sasayama, Chem. Abstr. 127, 95296 (1997)
A.S. Gajare, S.B. Bhawsar, D.B. Shinde, M.S. Shingare, Indian J. Chem. Sect. B 37B, 510 (1998)
H. Zhao, Y. Liu, Z. Cui, D. Beattie, Y. Gu, Q. Wang, J. Agric. Food Chem. 59, 11711 (2011)
L.M. Abell, J.V. Schloss, A.R. Rendina, in Target-side Directed Herbicide Design; ACS Symposium Series, vol 524 (1993), p. 16
L. Ballantine, J. McFarland, D.Hackett, in Triazine Herbicides: Risk Assessments; ACS Symposium Series, vol 683 (1998), p. xi
G. Rembarz, E. Fischer, K.Ch. Roeber, R. Ohff, H. Crahmer, J. Prakt. Chem. 311, 889 (1969)
G.C. Chiang, US Patent 4933450 A (1990)
G.C. Chiang, Chem. Abstr. 113, 231414 (1990)
P.S. Gates, G.P. Jones, D. E. Saunders, PCT Int. Appl. WO 9309099 A2 (1993)
P.S. Gates, G.P. Jones, D.E. Saunders, Chem. Abstr. 119, 180810 (1993)
O. Voss, P.J. Dudfield, K. Bauer, H. Bieringer, C. Rosinger, M. J. Ford, D. Green, Ger. Offen. 19521355 (1996)
O. Voss, P.J. Dudfield, K. Bauer, H. Bieringer, C. Rosinger, M.J. Ford, D. Green, Chem. Abstr. 126, 144293 (1997)
U. Burckhardt, M. Zimmermann, DE 2209470 (1972)
U. Burckhardt, M. Zimmermann, Chem. Abstr. 77, 164744 (1972)
S. Weiss, H. Krommer, US Patent 6342600 (2002)
S. Weiss, H. Krommer, Chem. Abstr. 136, 134795 (2002)
R. Menicagli, S. Samaritani, G. Signore, F. Vaglini, L.J. Dalla Via, Med. Chem. 47, 4649 (2004)
C. Courme, S. Gillon, N. Gresh, M. Vidal, C. Garbay, J.-C. Florent, E. Bertounesque, Tetrahedron Lett. 49, 4542 (2008)
R. Menicagli, S. Samaritani, S. Gori, Tetrahedron Lett. 40, 8419 (1999)
S. Samaritani, R. Menicagli, Tetrahedron 58, 1381 (2002)
S. Oudir, B. Rigo, J.P. Hénichart, P. Gautret, Synthesis 17, 2845 (2006)
L. Lucescu, P. Gautret, S. Oudir, B. Rigo, D. Belei, E. Bîcu, A. Ghinet, Synthesis 45, 1333 (2013)
J. Sun, M.A. Blaskovich, D. Knowles, Y. Qian, J. Ohkanda, R.D. Bailey, A.D. Hamilton, S.M. Sebti, Cancer Res. 59, 4919 (1999)
C.-M. Abuhaie, A. Ghinet, A. Farce, J. Dubois, P. Gautret, B. Rigo, D. Belei, E. Bîcu, Eur. J. Med. Chem. 59, 101 (2013)
C.-M. Abuhaie, A. Ghinet, A. Farce, J. Dubois, B. Rigo, E. Bîcu, Bioorg. Med. Chem. Lett. 23, 5887 (2013)
G.-M. Dumitriu, A. Ghinet, E. Bîcu, B. Rigo, J. Dubois, A. Farce, D. Belei, Bioorg. Med. Chem. Lett. 24, 3180 (2014)
D. Belei, C. Dumea, A. Samson, A. Farce, J. Dubois, E. Bîcu, A. Ghinet, Bioorg. Med. Chem. Lett. 22, 4517 (2012)
F.L. Zhang, P.J. Casey, Ann. Rev. Biochem. 65, 241 (1996)
K. Kónya, S. Fekete, A. Ábrahám, T. Patonay, Mol. Divers. 16, 91 (2012)
M. Boominathan, N. Pugazhenthiran, M. Nagaraj, S. Muthusubramanian, S. Murugesan, N. Bhuvanesh, ACS Sustainable Chem. Eng. 1, 1405 (2013)
B. Quan, S.X. Zhuang, C.C. Li, H.S. Dong, Indian J. Chem. 44B, 1717 (2005)
G. Biagi, I. Giorgi, O. Livi, A. Nardi, F. Pacchini, V. Scartoni, A. Lucacchini, Eur. J. Med. Chem. 38, 983 (2003)
D.R. Buckle, J. Heterocycl. Chem. 22, 77 (1985)
H. Schirok, C. Alonso-Alija, M. Michels, Synthesis 18, 3085 (2005)
W.J. Balfour, C.C. Greig, S. Visaisouk, J. Org. Chem. 39, 725 (1974)
K. von Auwers, E. Frese, Justus Liebigs Anna. Chem. 450, 273 (1926)
K. von Auwers, H.G. Allardt, Justus Liebigs Anna. Chem. 438, 20 (1924)
D. Belei, E. Bicu, P.G. Jones, M.L. Birsa, J. Heterocycl. Chem. 48, 129 (2011)
D. Belei, E. Bicu, M.L. Birsa, Acta Chem. Iasi 17, 197 (2009)
A.N. Gritsenko, S.V. Zhuravlev, V.A. Skorodumov, Uchenye Zapiski Inst. Farmakol. I 90 Kimioterapii Akad. Med. Nauk. USSR 1, 13 (1958)
J.B. Christensen, Molecules 6, 47 (2001)
G. Lang, G. Plos, PCT Int. Appl. (2002) WO 2002007689 A1 20020131
G. Lang, G. Plos, Chem. Abstr. 136, 139633 (2002)
L. Coudray, R.M. De Figueiredo, S. Duez, S. Cortial, J.J. Dubois, Enz. Inhib. Med. Chem. 24, 972 (2009)
R.B. Boyd, The NCI in vitro anticancer drug discovery screen, in Anticancer Drug Development Guide: Preclinical Screening, Clinical Trials, and Approval, ed. by B. Teicher (Humana Press Inc., Totowa, NJ, 1997), pp. 23–42
P. Skehan, R. Storeng, D. Scudiero, A. Monks, J. McMahon, D. Vistica, J.T. Warren, H. Bokesh, S. Kenney, M.R. Boyd, J. Natl. Cancer Inst. 82, 1107 (1990)
S. Diring, P. Retailleau, R. Ziessel, Synlett 19, 3027 (2007)
R. Frei, D. Wodrich, D.P. Hari, P.-A. Borin, C. Chauvier, J. Waser, J. Am. Chem. Soc. 136, 16563 (2014)
F. Diederich, P.J. Stang, R.R. Tykwinski, in Acetylene Chemistry: Chemistry, Biology and Material Science (Wiley-VCH: Weinheim, 2005)
Acknowledgments
This work was supported by the strategic Grant POSDRU/159/1.5/S/137750, Project “Doctoral and Postdoctoral programs support for increased competitiveness in Exact Sciences research,” co-financed by the European Social Fund within the Sectorial Operational Program Human Resources Development 2007–2013 (Ph. D. scholarship L. L.). The authors acknowledge the National Cancer Institute (NCI) for biological evaluation of compounds on their 60-cell panel: the testing was performed by the Developmental Therapeutics Program, Division of Cancer Treatment and Diagnosis (the URL to the Program’s website: http://dtp.cancer.gov).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lucescu, L., Bîcu, E., Belei, D. et al. Synthesis and biological evaluation of a new class of triazin–triazoles as potential inhibitors of human farnesyltransferase. Res Chem Intermed 42, 1999–2021 (2016). https://doi.org/10.1007/s11164-015-2131-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-015-2131-1