Abstract
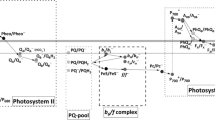
This is an article on the peroxydicarbonic acid (PODCA) hypothesis of photosynthetic water oxidation, which follows our first article in this general area (Castelfranco et al., Photosynth Res 94:235–246, 2007). In this article I have expanded on the idea of a protein-bound intermediate containing inorganic carbon in some chemically bound form. PODCA is conceived in this article as constituting a bridge between two proteins of the oxygen-evolving complex (OEC) that are essential for the evolution of O2. Presumably, these are two proteins which have been shown to possess Mn-dependent carbonic anhydrase activity (Lu et al., Plant Cell Physiol 46:1944–1953, 2005; Shitov et al., Biochemistry (Moscow) 74:509–517, 2009). One of these proteins may be the DI of the OEC core and the other may be the PsbO extrinsic protein. I attempt to relate briefly the PODCA hypothesis to the role of two cofactors for O2 evolution: Ca2+ and inorganic carbon. In this scheme, inorganic carbon (HCO3 −) mediates the oxidation of peroxide to dioxygen, thus avoiding the homolytic cleavage of the peroxide into two free radicals. I visualize the role of Ca2+ in the binding of PODCA to two essential photosystem II proteins. I propose that PODCA alternates between two Phases. In Phase 1, PODCA is broken down with the production of O2. In Phase 2, PODCA is regenerated.




Similar content being viewed by others
References
Castelfranco PA, Lu Y-K, Stemler AJ (2007) Hypothesis: the peroxydicarbonic acid cycle in photosynthetic water oxidation. Photosynth Res 94:235–246
Ignatova LK, Rudenko NN, Khristin MS, Ivanov BN (2006) Heterogeneous origin of carbonic anhydrase activity of thylakoid membranes. Biochemistry (Moscow) 71:525–532 (English translation)
Khristin MS, Ignatova LK, Rudenko NN, Ivanov BN (2004) Photosystem II associated carbonic anhydrase in higher plants is situated in core complex. FEBS Lett 577:305–308
Kreutz W (1974) Considerations on water-splitting in photosynthesis. In: Colhov K (ed) On the physics of biological membranes. Department of Physics, Simon Fraser University, Vancouver, pp 419–429
Lu Y-K, Stemler AJ (2002) Extrinsic photosystem II carbonic anhydrase in maize mesophyll chloroplasts. Plant Physiol 128:643–649
Lu Y-K, Stemler AJ (2007) Differing responses of two forms of carbonic anhydrases to chloride. BBA 1767:633–638
Lu Y-K, Theg SM, Stemler AJ (2005) Carbonic anhydrase activity in the photosystem II OEC33 protein from pea. Plant Cell Physiol 46:1944–1953
Metzner H (1978) Oxygen evolution as energetic problem. In: Metzner H (ed) Photosynthetic oxygen evolution. Academic Press, London, pp 59–76
Murray JW, Barber J (2006) Identification of a calcium-binding site in the PsbO protein of photosystem II. Biochemistry 45:4128–4130
Shitov V, Pobeguts OV, Smolova TN, Allakhverdiev SI, Klimov VV (2009) Manganese dependent carboanhydrase activity of photosystem II proteins. Biochemistry (Moscow) 74:509–517
Stemler AJ (1977) The binding of bicarbonate ions to washed chloroplast grana. Biochem Biophys Acta 460:511–522
Acknowledgments
The author wishes to thank Dr. William Lucas, chair of the Department of Plant Biology and many colleagues therein for their encouragement, departmental support, and for putting at my disposal space, equipment, etc. Friends in the Chemistry Department, particularly Dr. Mark Kurth, helped me to visualize the chemical properties of the compounds of interest and to foresee how they might react under different conditions. My friend and colleague, Dr. Alan Stemler, lent his expertise and full support to the development of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Castelfranco, P.A. A proposed role for inorganic carbon in water oxidation. Photosynth Res 116, 231–234 (2013). https://doi.org/10.1007/s11120-013-9864-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-013-9864-8