Abstract
Background and aims
We sought to describe the species and functional composition of Brazilian campos rupestres plant communities on severely nutrient-impoverished white sands, to test hypotheses relating plant communities and physiological adaptations to infertile soils. Based on recently-published information on a south-western Australian dune chronosequence, we hypothesised that campos rupestres plant communities would similarly contain a relatively large proportion of non-mycorrhizal species, because of the phosphorus-(P) impoverished nature of the soils. We also sought to test the hypothesis that many of these non-mycorrhizal species have high leaf manganese (Mn) concentrations as a consequence of carboxylate exudation to mobilise soil P.
Methods
We conducted flora surveys and quantified mycorrhizal status and foliar Mn concentrations in field sites with strongly-weathered sandy soils. Rhizosphere carboxylates were collected from glasshouse-grown plants to assess a potential correlation of carboxylates and leaf Mn concentrations.
Results
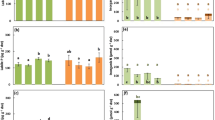
Soils were depleted of all major plant nutrients. Non-mycorrhizal plants were abundant in most field sites (mean relative cover = 48%). Vellozia species were dominant aboveground; belowground, roots were colonised more by dark septate endophytic fungi than by mycorrhizal fungi. From the field sites, foliar Mn concentrations in non-mycorrhizal species increased with decreasing soil P concentrations, but only when soil Mn concentrations were above a minimum threshold (exchangeable [Mn] above detection limit). Across all species, however, there was no relationship of foliar Mn concentrations with soil P concentrations.
Conclusions
Our hypothesis that white-sand campos rupestres communities contain a relatively large proportion of non-mycorrhizal plants was supported. Comparison with similar ecosystems in south-western Australia suggests that plant communities on severely P-impoverished sandy soils, despite differing evolutionary histories and little overlap in plant families, follow convergent evolutionary paths towards increasing abundance of non-mycorrhizal species.







Similar content being viewed by others
References
Abrahão A, Lambers H, Sawaya A, Mazzafera P, Oliveira R (2014) Convergence of a specialized root trait in plants from nutrient-impoverished soils: phosphorus-acquisition strategy in a nonmycorrhizal cactus. Oecologia 176:345–355
Ahmed MA, Kroener E, Holz M, Zarebanadkouki M, Carminati A (2014) Mucilage exudation facilitates root water uptake in dry soils. Funct Plant Biol 41:1129–1137
Albornoz FE, Burgess TI, Lambers H, Etchells H, Laliberté E (2016) Native soilborne pathogens equalize differences in competitive ability between plants of contrasting nutrient-acquisition strategies. J Ecol 105:549–557
Alcantara S, Mello-Silva R, Teodoro GS, Drequeceler K, Ackerly DD, Oliveira RS (2015) Carbon assimilation and habitat segregation in resurrection plants: a comparison between desiccation- and non-desiccation-tolerant species of Neotropical Velloziaceae (Pandanales). Funct Ecol 29:1499–1512
Alves RJV, Kolbek J (1994) Plant species endemism in savanna vegetation on table mountains (campo Rupestre) in Brazil. Vegetatio 113:125–139
Alves RJV, Kolbek J (2010) Can campo rupestre vegetation be floristically delimited based on vascular plant genera? Plant Ecol 207:67–79
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48
Brundrett MC (2009) Mycorrhizal associations and other means of nutrition of vascular plants: understanding the global diversity of host plants by resolving conflicting information and developing reliable means of diagnosis. Plant Soil 320:37–77
Conceição AA, Pirani JR, Meirelles ST (2007) Floristics, structure and soil of insular vegetation in four quartzite-sandstone outcrops of "Chapada Diamantina", Northeast Brazil. Rev Bras Bot 30:641–656
de Campos MCR (2012) Phosphorus-acquisition and phosphorus-conservation mechanisms of plants native to south-western Australia or to Brazilian rupestrian fields. PhD Thesis. School of Plant Biology. The University of Western Australia, Perth
de Carvalho F, Godoy EL, Lisboa FJ, de Souza Moreira FM, de Souza FA, Berbara RLL, Fernandes GW (2014) Relationship between physical and chemical soil attributes and plant species diversity in tropical mountain ecosystems from Brazil. J Mt Sci 11:875–883
Eiten G (1978) Delimitation of the cerrado concept. Vegetatio 36:169–178
Fox J (2003) Effect displays in R for generalised linear models. J Stat Softw 8:1–27
Giovannetti M, Mosse B (1980) An evaluation of techniques for measuring vesicular arbuscular mycorrhizal infection in roots. New Phytol 84:489–500
Goldblatt P (1997) Floristic diversity in the cape flora of South Africa. Biodivers Conserv 6:359–377
Google Inc (2012) Google earth (version 6.2.1). Google Inc., mountain view
Holdaway RJ, Richardson SJ, Dickie IA, Peltzer DA, Coomes DA (2011) Species- and community-level patterns in fine root traits along a 120 000-year soil chronosequence in temperate rain forest. J Ecol 99:954–963
Hopper SD (2009) OCBIL theory: towards an integrated understanding of the evolution, ecology and conservation of biodiversity on old, climatically buffered, infertile landscapes. Plant Soil 322:49–86
Hopper SD, Silveira FA, Fiedler PL (2016) Biodiversity hotspots and Ocbil theory. Plant Soil 403:167–216
INMET (2015) Historical Data, station 83538 (1961–2015). Instituto Nacional de Meteorologia, Brasília. http://www.inmet.gov.br/portal/index.php?r=bdmep/bdmep
Jumpponen A (2001) Dark septate endophytes–are they mycorrhizal? Mycorrhiza 11:207–211
Laliberté E, Turner BL, Costes T, Pearse SJ, Wyrwoll KH, Zemunik G, Lambers H (2012) Experimental assessment of nutrient limitation along a 2-million-year dune chronosequence in the south-western Australia biodiversity hotspot. J Ecol 100:631–642
Laliberté E, Zemunik G, Turner BL (2014) Environmental filtering explains variation in plant diversity along resource gradients. Science 345:1602–1605
Lambers H, Shane MW, Cramer MD, Pearse SJ, Veneklaas EJ (2006) Root structure and functioning for efficient acquisition of phosphorus: matching morphological and physiological traits. Ann Bot 98:693–713
Lambers H, Raven JA, Shaver GR, Smith SE (2008) Plant nutrient-acquisition strategies change with soil age. Trends Ecol Evol 23:95–103
Lambers H, Bishop JG, Hopper SD, Laliberté E, Zúñiga-Feest A (2012) Phosphorus-mobilization ecosystem engineering: the roles of cluster roots and carboxylate exudation in young P-limited ecosystems. Ann Bot 110:329–348
Lambers H, Clements JC, Nelson MN (2013) How a phosphorus-acquisition strategy based on carboxylate exudation powers the success and agronomic potential of lupines (Lupinus, Fabaceae). Am J Bot 100:263–288
Lambers H, Shane MW, Laliberté E, Swarts ND, Teste F, Zemunik G (2014) Plant mineral nutrition. In: Lambers H (ed) Plant life on the sandplains in Southwest Australia, a global biodiversity hotspot. UWA Publishing, Crawley
Lambers H, Hayes PE, Laliberté E, Oliveira RS, Turner BL (2015) Leaf manganese accumulation and phosphorus-acquisition efficiency. Trends Plant Sci 20:83–90
Lamont B (2003) Structure, ecology and physiology of root clusters – a review. Plant Soil 248:1–19
Lima TA, Pinto JRR, Lenza E, Pinto AS (2010) Florística e estrutura da vegetação arbustivo-arbórea em uma área de cerrado rupestre no Parque Estadual da Serra de Caldas Novas, Goiás. Biota. Neotropica 10:159–166
Lipton DS, Blanchar RW, Blevins DG (1987) Citrate, malate, and succinate concentration in exudates from P-sufficient and P-stressed Medicago Sativa L. seedlings. Plant Physiol 85:315–317
Mandyam K, Jumpponen A (2005) Seeking the elusive function of the root-colonising dark septate endophytic fungi. Stud Mycol 53:173–189
McCully ME (1999) Roots in soil: unearthing the complexities of roots and their rhizospheres. Annu Rev Plant Biol 50:695–718
Myers N, Mittermeier RA, Mittermeier CG, Da Fonseca GA, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Nagajyoti P, Lee K, Sreekanth T (2010) Heavy metals, occurrence and toxicity for plants: a review. Environ Chem Lett 8:199–216
Newsham KK (2011) A meta-analysis of plant responses to dark septate root endophytes. New Phytol 190:783–793
Oliveira RS, Galvão HC, Campos MC, Eller CB, Pearse SJ, Lambers H (2015) Mineral nutrition of campos rupestres plant species on contrasting nutrient-impoverished soil types. New Phytol 205:1183–1194
Orchard S, Standish R, Nicol D, Gupta V, Ryan M (2016) The response of fine root endophyte (Glomus Tenue) to waterlogging is dependent on host plant species and soil type. Plant Soil 403:305–315
Phillips J, Hayman D (1970) Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. T Brit Mycol Soc 55:158–161
Pinheiro J, Bates D, DebRoy S, Sarkar D, Team RDC (2015) Nlme: linear and nonlinear mixed effects models. R package version 3.1-120 edn
Porder S, Ramachandran S (2013) The phosphorus concentration of common rocks—a potential driver of ecosystem P status. Plant Soil 367:41–55
Price SR (1911) The roots of some north African desert-grasses. New Phytol 10:328–340
Quesada C, Lloyd J, Schwarz M, Patiño S, Baker T, Czimczik C, Fyllas N, Martinelli L, Nardoto G, Schmerler J (2010) Variations in chemical and physical properties of Amazon forest soils in relation to their genesis. Biogeosciences 7:1515–1541
R Development Core Team (2013) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Richardson SJ, Peltzer DA, Allen RB, McGlone MS, Parfitt RL (2004) Rapid development of phosphorus limitation in temperate rainforest along the Franz Josef soil chronosequence. Oecologia 139:267–276
Rossel RAV, Bui EN (2016) A new detailed map of total phosphorus stocks in Australian soil. Sci Total Environ 542:1040–1049
Ryan M, Tibbett M, Edmonds-Tibbett T, Suriyagoda L, Lambers H, Cawthray G, Pang J (2012) Carbon trading for phosphorus gain: the balance between rhizosphere carboxylates and arbuscular mycorrhizal symbiosis in plant phosphorus acquisition. Plant Cell Environ 35:2170–2180
Saadi A (1995) A geomorfologia da Serra do Espinhaço em Minas Gerais e de suas margens. Geonomos 3:41–63
Shane MW, Lambers H (2005a) Cluster roots: a curiosity in context. Plant Soil 274:101–125
Shane MW, Lambers H (2005b) Manganese accumulation in leaves of Hakea prostrata (Proteaceae) and the significance of cluster roots for micronutrient uptake as dependent on phosphorus supply. Physiol Plant 124:441–450
Shane MW, Cramer MD, Funayama-Noguchi S, Cawthray GR, Millar AH, Day DA, Lambers H (2004) Developmental physiology of cluster-root carboxylate synthesis and exudation in harsh hakea. Expression of phosphoenolpyruvate carboxylase and the alternative oxidase. Plant Physiol 135:549–560
Shane MW, Cawthray GR, Cramer MD, Kuo J, Lambers H (2006) Specialized ‘dauciform’roots of Cyperaceae are structurally distinct, but functionally analogous with ‘cluster’roots. Plant Cell Environ 29:1989–1999
Shane MW, McCully ME, Canny MJ, Pate JS, Lambers H (2011) Development and persistence of sandsheaths of Lyginia barbata (Restionaceae): relation to root structural development and longevity. Ann Bot 108:1307–1322
Silveira FAO, Negreiros D, Barbosa NP, Buisson E, Carmo FF, Carstensen DW, Conceição AA, Cornelissen TG, Echternacht L, Fernandes GW, Garcia QS, Guerra TJ, Jacobi CM, Lemos-Filho JP, Le Stradic S, Morellato LPC, Neves FS, Oliveira RS, Schaefer CE, Viana PL, Lambers H (2016) Ecology and evolution of plant diversity in the endangered campo rupestre: a neglected conservation priority. Plant Soil 403:129–152
Smith SE, Read DJ (2008) Mycorrhizal Symbiosis. Elsevier, New York
Smith SE, Facelli E, Pope S, Smith FA (2010) Plant performance in stressful environments: interpreting new and established knowledge of the roles of arbuscular mycorrhizas. Plant Soil 326:3–20
Smith RJ, Hopper SD, Shane MW (2011) Sand-binding roots in Haemodoraceae: global survey and morphology in a phylogenetic context. Plant Soil 348:453–470
Trouvelot A, Kough JL, Gianinazzi-Pearson V (1986) Mesure du taux de mycorhization VA d’un système radiculaire. Recherche de méthodes d’estimation ayant une signification fonctionnelle. In: Gianinazzi-Pearson V, Gianinazzi S (eds) Physiological and genetical aspects of mycorrhizae proceedings of the 1st European symposium on mycorrhizae. INRA, Paris
Turner BL, Condron LM (2013) Pedogenesis, nutrient dynamics, and ecosystem development: the legacy of TW Walker and JK Syers. Plant Soil 367:1–10
Turner BL, Haygarth PM (2001) Biogeochemistry: phosphorus solubilization in rewetted soils. Nature 411:258–258
Turner BL, Romero TE (2009) Short-term changes in extractable inorganic nutrients during storage of tropical rain forest soils. Soil Sci Soc Am J 73:1972–1979
Walker TW, Syers JK (1976) The fate of phosphorus during pedogenesis. Geoderma 15:1–19
Wardle DA, Walker LR, Bardgett RD (2004) Ecosystem properties and forest decline in contrasting long-term chronosequences. Science 305:509–513
Xu R (2003) Measuring explained variation in linear mixed effects models. Stat Med 22:3527–3541
Zemunik G, Turner BL, Lambers H, Laliberté E (2015) Diversity of plant nutrient-acquisition strategies increases during long-term ecosystem development. Nature plants 1: article number: 15050, 15051–15054
Zemunik G, Turner BL, Lambers H, Laliberté E (2016) Increasing plant species diversity and extreme species turnover accompany declining soil fertility along a long-term chronosequence in a biodiversity hotspot. J Ecol 104:792–805
Acknowledgements
GZ was supported by a scholarship from the Paul Hasluck Bequest administered by the Kwongan Foundation and was the recipient of an Endeavour Research Award provided by Austraining International. GZ also acknowledges the Plant Ecology Program at Unicamp, for financial support for the field work. HL acknowledges support from a CNPq grant, CAPES PVE 88887.108541/2015-00. We thank the Minas Gerais State Park authority for permission to conduct research in the state parks under their administration. UPLC laboratory analyses were done by Alexandra Sarawaya. Sara Adrian, Paulo Mazzafera and Sandra Maria Carmello-Guerreiro, provided laboratory assistance and facilities for the mycorrhizal analysis of roots; Sara Adrian kindly analysed the roots of Vellozia stenocarpa. Livia Echternacht, Graça Wanderley, Juliana dos Santos, Renato de Mello-Silva, Taciana Barbosa Calvacanti, João Martins do Carmo, Edson Dias da Silva, Suzana Costa, Angela Martins and Gustavo Shimizu provided valuable assistance in species identification, and many people from Unicamp assisted with field work. Topographic data for Fig. 1 are based on data services provided by the OpenTopography Facility with support from the National Science Foundation under NSF Award Numbers 1226353 & 1225810.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Philippe Hinsinger.
Electronic supplementary material
ESM 1
(DOCX 2667 kb)
Rights and permissions
About this article
Cite this article
Zemunik, G., Lambers, H., Turner, B.L. et al. High abundance of non-mycorrhizal plant species in severely phosphorus-impoverished Brazilian campos rupestres . Plant Soil 424, 255–271 (2018). https://doi.org/10.1007/s11104-017-3503-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-017-3503-7