Abstract
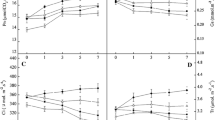
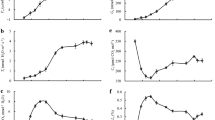
The effects of calcium chloride solution (10 mmol L–1) on mesophyll cell ultrastructure, gas exchange, chlorophyll and carotenoid content, and PSII in tobacco leaf were studied by simulating water deficit conditions via treatment with 25% PEG-6000 for 24 h. The results showed that under drought stress, the mesophyll cell structure and morphology were destroyed, photosynthesis and gas-exchange processes changed, photosynthetic pigment content decreased, and the electron transfer efficiency in PSII reduced. However, compared with the control treatment, under drought conditions, the addition of exogenous calcium could stabilize the structure and function of the chloroplasts, mitochondria, and endomembrane system in the mesophyll cells, maintain normal leaf net photosynthetic rate and gas exchange, alleviate the degree of photosynthetic pigment degradation, and increase the electron transfer energy in the leaves in PSII. As a means of ensuring normal photosynthesis under drought stress, we discovered that the application of exogenous calcium was more important for stabilization of the structure of the organelles, regulation of the osmotic balance, and increase of the photosynthetic pigment content, and proved to be less important for regulation of stomatal opening and closing.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- Car:
-
carotenoids
- C i :
-
intercellular CO2 concentration
- Chl:
-
chlorophyll
- E :
-
transpiration rate
- FM:
-
fresh mass
- gs:
-
stomatal conductance
- NPQ:
-
nonphotochemical quenching
- ΦPSII :
-
effective quantum yield of PSII photochemistry
- PEG:
-
polyethylene glycol
- P N :
-
net photosynthetic rate
- qp:
-
photochemical quenching coefficient
- Ψw :
-
water potential
References
Anjum S.A., Wang L.C., Farooq M. et al.: Brassinolide application improves the drought tolerance in maize through modulation of enzymatic antioxidants and leaf gas exchange.–J. Agron. Crop Sci. 197: 177–185, 2011.
Betterle N., Ballottari M., Baginsky S. et al.: High lightdependent phosphorylation of photosystem II inner antenna CP29 in Monocots is STN7 independent and enhances nonphotochemical quenching.–Plant Physiol. 167: 457–471, 2015.
Betterle N., Poudyal R.S., Rosa A. et al.: The STN8 kinase-PBCP phosphatase system is responsible for high-light-induced reversible phosphorylation of the PSII inner antenna subunit CP29 in rice.–Plant J. 89: 681–691, 2017.
Bilger W., Björkman O.: Temperature-dependence of violaxanthin deepoxidation and nonphotochemical fluorescence quenching in intact leaves of Gossypium hirsutum L and Malva parviflora L.–Planta 184: 226–234, 1991.
Chen D.Q., Wang S.W., Cao B.B. et al.: Genotypic variation in growth and physiological response to drought stress and rewatering reveals the critical role of recovery in drought adaptation in maize seedlings.–Front Plant Sci. 6: 1241, 2016.
Cho E.K., Hong C.B.: Over-expression of tobacco NtHSP70-1 contributes to drought-stress tolerance in plants.–Plant Cell Rep. 25: 349–358, 2006.
Dolatabadian A., Sanavy S.A.M.M., Gholamhoseini M. et al.: The role of calcium in improving photosynthesis and related physiological and biochemical attributes of spring wheat subjected to simulated acid rain.–Physiol. Mol. Biol. Plant. 19: 189–198, 2013.
Elgersma A., Soegaard K., Jensen S.K.: Interrelations between herbage yield, alpha-tocopherol, beta-carotene, lutein, protein, and fiber in non-leguminous forbs, forage legumes, and a grassclover mixture as affected by harvest date.–J. Agr. Food Chem. 63: 406–414, 2015.
Farquhar G.D., Sharkey T.D.: Stomatal conductance and photosynthesis.–Annu. Rev. Plant Phys. 33: 317–345, 1982.
Gong M., Chen S.N., Song Y.Q. et al.: Effect of calcium and calmodulin on intrinsic heat tolerance in relation to antioxidant systems in maize seedlings.–Aust. J. Plant Physiol. 24: 371–379, 1997.
Hochmal A.K., Schulze S., Trompelt K. et al.: Calciumdependent regulation of photosynthesis.–BBA-Bioenergetics 1847: 993–1003, 2015.
Hu Y.B., Sun G.Y., Wang X.C.: Induction characteristics and response of photosynthetic quantum conversion to changes in irradiance in mulberry plants.–J. Plant Physiol. 164: 959–968, 2007.
Huo Y.J., Wang M.P., Wei Y.Y. et al.: Overexpression of the maize psbA gene enhances drought tolerance through regulating antioxidant system, photosynthetic capability, and stress defense gene expression in tobacco.–Front Plant Sci. 6: 1223, 2016.
Ingram J., Bartels D.: The molecular basis of dehydration tolerance in plants.–Annu. Rev. Plant Phys. 47: 377–403, 1996.
Liu J., Shi D.C.: Photosynthesis, chlorophyll fluorescence, inorganic ion and organic acid accumulations of sunflower in responses to salt and salt-alkaline mixed stress.–Photosynthetica 48: 127–134, 2010.
Lu C., Vonshak A.: Effects of salinity stress on photosystem II function in cyanobacterial Spirulina platensis cells.–Physiol. Plantarum 114: 405–413, 2002.
Reif C., Arrigoni E., Berger F. et al.: Lutein and beta-carotene content of green leafy Brassica species grown under different conditions.–Lebensm.-Wiss. Technol. 53: 378–381, 2013.
Rickauer M., Tanner W.: Effects of Ca2+ on amino-acid-transport and accumulation in roots of phaseolus-vulgaris.–Plant Physiol. 82: 41–46, 1986.
Tan C.S., Buttery B.R.: Response of stomatal conductance, transpiration, photosynthesis, and leaf water potential in peach seedlings to different watering regimes.–HortScience 107: 222–223, 1982.
Tan W., Meng Q.W., Brestic M. et al.: Photosynthesis is improved by exogenous calcium in heat-stressed tobacco plants.–J. Plant Physiol. 168: 2063–2071, 2011.
Verslues P.E., Agarwal M., Katiyar-Agarwal S. et al.: Methods and concepts in quantifying resistance to drought, salt and freezing, abiotic stresses that affect plant water status.–Plant J. 45: 523–539, 2006.
Wang W.B., Kim Y.H., Lee H.S. et al.: Analysis of antioxidant enzyme activity during germination of alfalfa under salt and drought stresses.–Plant Physiol. Bioch. 47: 570–577, 2009.
White P.J., Broadley M.R.: Calcium in plants.–Ann. Bot.-London 92: 487–511, 2003.
Wise R.R., Ortiz-Lopez A., Ort D.R.: Spatial-distribution of photosynthesis during drought in field-grown and acclimated and nonacclimated growth chamber-grown cotton.–Plant Physiol. 100: 26–32, 1992.
Xu C.B., Li X.M., Zhang L.H.: The effect of calcium chloride on growth, photosynthesis, and antioxidant responses of Zoysia japonica under drought conditions.–PLoS ONE 8: e68214, 2013.
Xu H.X., Heath M.C.: Role of calcium in signal transduction during the hypersensitive response caused by basidiosporederived infection of the cowpea rust fungus.–Plant Cell 10: 585–598, 1998.
Xu P.L., Guo Y.K., Bai J.G. et al.: Effects of long-term chilling on ultrastructure and antioxidant activity in leaves of two cucumber cultivars under low light.–Physiol. Plantarum 132: 467–478, 2008.
Xu Z.Z., Zhou G.S., Shimizu H.: Effects of soil drought with nocturnal warming on leaf stomatal traits and mesophyll cell ultrastructure of a perennial grass.–Crop Sci. 49: 1843–1851, 2009.
Zhou W.J., Lin X.Q.: Effects of waterlogging at different growth stages on physiological characteristics and seed yield of winter rape (Brassica napus L.).–Field Crop Res. 44: 103–110, 1995.
Zhu S., Zhou X.P., Wu X.M. et al.: Structure and function of the CBL-CIPK Ca2+-decoding system in plant calcium signaling.–Plant Mol. Biol. Rep. 31: 1193–1202, 2013.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgements: This study was supported by the Young Elite Scientists Sponsorship Program by the China Association for Science and Technology (2016QNRC001).
This article is published with open access at link.springer.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Hu, W., Tian, S.B., Di, Q. et al. Effects of exogenous calcium on mesophyll cell ultrastructure, gas exchange, and photosystem II in tobacco (Nicotiana tabacum Linn.) under drought stress. Photosynthetica 56, 1204–1211 (2018). https://doi.org/10.1007/s11099-018-0822-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-018-0822-8