Abstract
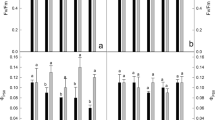
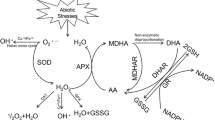
Nicosulfuron is a post-emergence herbicide used for weed control in fields of maize (Zea mays L.). We used a pair of nearly isogenic inbred lines, SN509-R (nicosulfuron-resistant) and SN509-S (nicosulfuron-sensitive), to study the effect of nicosulfuron on waxy maize seedling. After the nicosulfuron treatment, net photosynthetic rate, stomatal conductance, transpiration rate, leaf maximum photochemical efficiency of PSII, photochemical quenching of chlorophyll fluorescence, and the actual photochemical efficiency of PSII were significantly lower in SN509-S than those of SN509-R, contrary to intercellular CO2 concentration, stomatal limitation, and nonphotochemical quenching. Compared to SN509-R, antioxidant enzyme activities in SN509-S decreased significantly in response to the nicosulfuron treatment, while SN509-S exhibited an increased malondialdehyde content, which was associated with lower antioxidant enzyme activities. These results collectively suggest that the nicosulfuron-resistance mechanism was associated with photosynthetic rate, reactive oxygen species metabolism, and protective mechanisms.
Similar content being viewed by others
Abbreviations
- APX:
-
ascorbate peroxidase
- CAT:
-
catalase
- Chl:
-
chlorophyll
- C i :
-
intercellular CO2 concentration
- DAT:
-
days after herbicide treatment
- ETR:
-
electron transport rate
- F0 :
-
minimal fluorescence yield of the dark-adapted state
- Fm :
-
maximal fluorescence yield of the dark-adapted state
- F0' :
-
minimal fluorescence yield of the light-adapted state
- Fm' :
-
maximal fluorescence yield of the lightadapted state
- Fv :
-
variable fluorescence
- Fv/Fm :
-
maximal quantum yield of PSII photochemistry
- gs:
-
stomatal conductance
- Ls :
-
stomatal limitation
- MDA:
-
malondialdehyde
- NPQ:
-
nonphotochemical quenching
- NILs:
-
nearly-isogenic lines
- NBT:
-
nitroblue tetrazolium chloride
- P N :
-
net photosynthetic rate
- POD:
-
peroxidase
- qP:
-
photochemical quenching coefficient
- ROS:
-
reactive oxygen species
- SOD:
-
superoxide dismutase
- ΦPSII :
-
effective quantum yield of PSII photochemistry
References
Aebi H.: Catalase in vitro.–Methods Enzymol. 105: 121–126, 1984.
Aroca R., Irigoyen J.J., Sánchez-Díaz M.: Photosynthetic characteristics and protective mechanisms against oxidative stress during chilling and subsequent recovery in two maize varieties differing in chilling sensitivity.–Plant Sci. 161: 719–726, 2001.
Belgers J.D.M., van Lieverloo R.J., van der Pas L.J.T. et al.: Effects of the herbicide 2,4-D on the growth of nine aquatic macrophytes.–Aquat. Bot. 86: 260–268, 2007.
Bigot A., Fontaine F., Clément C., Vaillant-Gaveau N.: Effect of the herbicide flumioxazin on photosynthetic performance of grapevine (Vitis vinifera L.).–Chemosphere 67: 1243–1251, 2007.
Cakmak I., Marschner H.: Magnesium deficiency and high light intensity enhance activities of superoxide dismutase, ascorbate peroxidase, and glutathione reductase in bean leaves.–Plant Physiol. 98: 1222–1227, 1992.
Debona D., Nascimento K.J.T., Gomes J.G.O. et al.: Physiological changes promoted by a strobilurin fungicide in the rice-Bipolaris oryzae interaction.–Pestic. Biochem. Phys. 130: 8–16, 2016.
Demmig B., Winter K., Krüger A., Czygan F.C.: Photoinhibition and zeaxanthin formation in intact leaves: A possible role of the xanthophyll cycle in the dissipation of excess light energy.–J. Plant Physiol. 84: 218–224, 1987.
Dobbels A.F., Kapusta G.: Postemergence weeds control in corn (Zea mays) with nicosulfuron combinations.–Weed Technol. 7: 844–850, 1993.
Eberline C.V., Rosow K.M., Geadelmann J.L., Openshaw S.J.: Differential tolerance of corn genotypes to DPX-M6316.–Weed Sci. 37: 651–657, 1989.
Frankart C., Eullaffroy P., Vernet G.: Comparative effects of four herbicides on non-photochemical fluorescence quenching in Lemna minor.–Environ. Exp. Bot. 49: 159–168, 2003.
Freemark K., Boutin C.: Nontarget-plant risk assessment for pesticide registration.–Environ. Manage. 18: 841–854, 1994.
Genty B., Briantais J.M., Baker N.R.: The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence.–Biochim. Biophys. Acta 990: 87–92, 1989.
Geoffroy L., Frankart C., Eullaffroy P.: Comparison of different physiological parameter responses in Lemna minor and Scenedesmus obliquus exposed to herbicide flumioxazin.–Environ. Pollut. 131: 233–241, 2004.
Giannopolitis C.N., Ries S.K.: Superoxide dismutases: I. occurrence in higher plants.–Plant Physiol. 59: 309–314, 1977.
Govindjee: A role for a light-harvesting antenna complex of photosystem II in photoprotection.–Plant Cell 14: 1663–1668, 2002.
Hassan N.M., Nemat Alla M.M.: Oxidative stress in herbicidetreated broad bean and maize plants.–Acta Physiol. Plant. 27: 429–438, 2005.
Heath R.L., Packer L.: Photoperoxidation in isolated chloroplasts. I. kinetics and stoichiometry of fatty acid peroxidation.–Arch. Biochem. Biophys. 125: 189–198, 1968.
Hu H.J., Shi Z.S., Lv X.L. et al.: [Effect of herbicide nicosulfuron on photosynthesis traits and chlorophyll fluorescence parameters of wax maize.]–J. Maize Sci. 22: 77–80, 2014. [In Chinese]
Hussain M.I., González L., Reigosa M.: Phytotoxic effects of allelochemicals and herbicides on photosynthesis, growth and carbon isotope discrimination in Lactuca sativa.–Allelopathy J. 26: 157–174, 2010.
Hwang H.J., Kim J.H., Eu Y.J. et al.: Photoinhibition of photosystem I is accelerated by dimethyldithiocarbamate, an inhibitor of superoxide dismutase, during light-chilling of spinach leaves.–J. Photoch. Photobio. B. 73: 79–85, 2004.
Jakob B., Heber U.: Photoproduction and detoxification of hydroxyl radicals in chloroplasts and leaves and relation to photoinactivation of photosystems I and II.–Plant Cell Physiol. 37: 629–635, 1996.
Jiang L., Yang H.: Prometryne-induced oxidative stress and impact on antioxidant enzymes in wheat.–Ecotox. Environ. Safe. 72: 1687–1693, 2009.
Jiang Z., Ma B.B., Erinle K.O. et al.: Enzymatic antioxidant defense in resistant plant: Pennisetum americanum (L.) K. Schum during long-term atrazine exposure.–Pestic. Biochem. Phys. 133: 59–66, 2016.
Kana R., Špundová M., Ili´k P. et al.: Effect of herbicide clomazone on photosynthetic processes in primary barley (Hordeum vulgare L.) leaves.–Pestic. Biochem. Phys. 78: 161–170, 2004.
Kapusta G., Krausz R.F., Khan M., Matthews J.L.: Effect of nicosulfuron rate, adjuvant and weed size on annual weed control in corn (Zea mays).–Weed Technol. 8: 696–702, 1995.
Liu Z., Shi L., Bai L., Zhao K.: [Effects of salt stress on the contents of chlorophyll and organic solutes in Aeluropus littoralis var. sinensis Debeaux.]–J. Plant Physiol. Mol. Biol. 33: 165–172, 2007. [In Chinese]
Macedo R.S., Lombardi A.T., Omachi C.Y., Rörig L.R.: Effects of herbicide bentazon on growth and maximum quantum yield of photosystem II in the marine diatom skeletonema costatum.–Toxicol. in Vitro 22: 716–722, 2008.
Meyer M.D., Pataky J.K., Williams M.M.: Genetic factors influencing adverse effects of mesotrione and nicosulfuron on sweet corn yield.–Agron. J. 102: 1138–1145, 2010.
Murata N., Takahashi S., Nishiyama Y., Allakhverdiev S.I.: Photoinhibition of photosystem II under environmental stress.–BBA-Biochemistry 1767: 414–421, 2007.
Nakano Y., Asada K.: Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts.–Plant Cell Physiol. 22: 867–880, 1981.
Pataky J.K., Meyer J.D., Bollman J.D. et al.: Genetic basis for varied levels of injury to sweet corn hybrids from three cytochrome P450-metabolized herbicides.–J. Am. Soc. Hortic. Sci. 133: 438–447, 2008.
Platiša J., Veljovic-Jovanovic S., Kukavica B. et al.: Induction of peroxidases and superoxide dismutases in transformed embryogenic calli of alfalfa (Medicago sativa L.).–J. Plant Physiol. 165: 895–900, 2008.
Qian H., Chen W., Sheng G.D. et al.: Effects of glufosinate on antioxidant enzymes, subcellular structure, and gene expression in the unicellular green alga Chlorella vulgaris.–Aquat. Toxicol. 88: 301–307, 2008.
Ramel F., Sulmon C., Bogard M. et al.: Differential patterns of reactive oxygen species and antioxidative mechanisms during atrazine injury and sucrose-induced tolerance in Arabidopsis thaliana plantlets.–BMC Plant Biol. 9: 28, 2009.
Ray T.B.: Site of action of chlorsulfuron.–Plant Physiol. 75: 827–831, 1984.
Saladin G., Magné C., Clément C.: Impact of flumioxazin herbicide on growth and carbohydrate physiology in Vitis vinifera L.–Plant Cell Rep. 21: 821–827, 2003.
Sonoike K.: Degradation of PsaB gene product, the reaction center subunit of photosystem I, is caused during photo inhibition of photosystem I: possible involvement of active oxygen species.–Plant Sci. 115: 157–164, 1996.
Stall W.M., Bewick T.A.: Sweet corn cultivars respond differentially to the herbicide nicosulfuron.–HortScience 27: 131–133, 1992.
Tan W., Li Q.L., Zhai H.: Photosynthesis and growth responses of grapevine to acetochlor and fluoroglycofen.–Pestic. Biochem. Phys. 103: 210–218, 2012.
Tarchoune I., Sgherri C., Izzo R. et al.: Antioxidative responses of Ocimum basilicum to sodium chloride or sodium sulphate salinization.–Plant Physiol. Bioch. 48: 772–777, 2010.
Tjus S.E., Scheller H.V., Andersson B., Møller B.L.: Active oxygen produced during selective excitation of photosystem I is damaging not only to photosystem I, but also to photosystem II.–Plant Physiol. 125: 2007–2015, 2001.
van Kooten O., Snel J.F.H.: The use of chlorophyll fluorescence nomenclature in plant stress physiology.–Photosynth. Res. 25: 147–150, 1990.
Vass I., Styring S., Hundal T. et al.: Reversible and irreversible intermediates during photoinhibition of photosystem II: stable reduced QA species promotes chlorophyll triplet formation.–P. Natl. Acad. Sci. USA 89: 1408–1412, 1992.
Wang J., Zhong X.M., Lv X.L. et al.: [Advances in research on tolerance of differential corn cultivars to the nicosulfuron.]–Chinese J. Pestic. Sci. 18: 282–290, 2016. [In Chinese]
Wang X.F., Guan C.H., Fan Z.W., Zhen H.J.: Application and development of sulfonylurea herbicides.–Agrochemicals 50: 9–15, 2011.
Williams B.J., Harvey R.G.: Effect of nicosulfuron timing on wild-proso millet (Panicum miliaceum) control in sweet corn (Zea mays).–Weed Technol. 14: 377–382, 2000.
Wu G.L., Cui J., Tao L., Yang H.: Fluroxypyr triggers oxidative damage by producing superoxide and hydrogen peroxide in rice (Oryza sativa).–Ecotoxicology 19: 124–132, 2010.
Wu X.X., Wu J.C., Jin G.Y. et al.: [Impact of herbicides on physiology and ecology of hydrophytes.]–Acta Ecol. Sin. 24: 2037–2042, 2004. [In Chinese]
Xue Y.J., Tao L., Yang Z.M.: Aluminum-induced cell wall peroxidase activity and lignin synthesis are differentially regulated by jasmonate and nitric oxide.–J. Agr. Food Chem. 56: 9676–9684, 2008.
Yoon J.Y., Shin J.S., Shin D.Y. et al.: Tolerance to paraquatmediated oxidative and environmental stresses in squash (Cucurbita spp.) leaves of various ages.–Pestic. Biochem. Physiol. 99: 65–76, 2011.
Yuan X.Y, Zhang L.G., Ning N. et al.: Photosynthetic physiological response of radix isatidis (Isatis indigotica Fort.) seedlings to nicosulfuron.–PLoS ONE 9: e105310, 2014.
Zheleva D., Tsonev T., Sergiev I., Karanov E.: Protective effect of exogenous polyamines against atrazine in pea plants.–J. Plant Growth Regul. 13: 203–211,1994.
Zhong X.M., Shi Z.S., Li F.H., Huang H.J.: Photosynthesis and chlorophyll fluorescence of infertile and fertile stalks of paired near-isogenic lines in maize (Zea mays L.) under shade conditions.–Photosynthetica 52: 597–603, 2014.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgements: This work was supported by the National Science and Technology Support Project (2014215031) and Cultivation Plan for Youth Agricultural Science and Technology Innovative Talents of Liaoning Province (2015041).
Rights and permissions
About this article
Cite this article
Wang, J., Zhong, X.M., Lv, X.L. et al. Photosynthesis and physiology responses of paired near-isogenic lines in waxy maize (Zea mays L.) to nicosulfuron. Photosynthetica 56, 1059–1068 (2018). https://doi.org/10.1007/s11099-018-0816-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-018-0816-6