Abstract
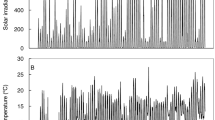
We compared the interactive effects of temperature and light intensity on growth, photosynthetic performance, and antioxidant enzyme activity in Zizania latifolia Turcz. plants in this study. Plants were grown under field (average air temperature 9.6–25°C and average light intensity 177–375 W m−2) or greenhouse (20–32°C and 106–225 W m−2) conditions from the spring to the early summer. The results indicated that greenhouse-grown plants (GGP) had significantly higher plant height, leaf length, and leaf width, but lower leaf thickness and total shoot mass per cluster compared with field-grown plants (FGP). Tiller emergence was almost completely suppressed in GGP. Significantly higher chlorophyll (Chl) content and lower Chl a/b ratio were observed in GGP than in FGP. From 4 to 8 weeks after treatment (WAT), net photosynthetic rate (P N) was significantly lower in FGP than in GGP. However, from 9 to 12 WAT, P N was lower in GGP, accompanied by a decrease in stomatal conductance (g s) and electron transport rate (ETR) compared with FGP. Suppressed P N in GGP under high temperature combined with low light was also indicated by photosynthetic photon flux density (PPFD) response curve and its diurnal fluctuation 10 WAT. Meanwhile, ETR in GGP was also lower than in FGP according to the ETR — photosynthetically active radiation (PAR) curve. The results also revealed that GGP had a lower light saturation point (LSP) and a higher light compensation point (LCP). From 4 to 8 WAT, effective quantum yield of PSII photochemistry (ΦPSII), photochemical quenching (qP), and ETR were slightly lower in FGP than in GGP. The activities of ascorbate peroxidase (APX), guaiacol peroxidase (POD), glutathione reductase (GR), superoxide dismutase (SOD), and malondialdehyde (MDA) content were significantly higher from 4 to 8 WAT, but lower from 10 to 12 WAT in FGP. However, catalase (CAT) activity was significantly lower in FGP from 4 to 8 WAT. Our results indicated that the growth and photosynthetic performance of Z. latifolia plants were substantially influenced by temperature, as well as light intensity. This is helpful to understand the physiological basis for a protected cultivation of this crop.
Similar content being viewed by others
Abbreviations
- APX:
-
ascorbate peroxidase
- AsA:
-
ascorbic acid
- C i :
-
intercellular CO2 concentration
- CAT:
-
catalase
- Chl:
-
chlorophyll
- DM:
-
dry mass
- E :
-
transpiration rate
- ETR:
-
electron transport rate
- FGP:
-
field-grown plants
- FM:
-
fresh mass
- F0 :
-
minimal fluorescence yield of the dark-adapted state
- F0′:
-
minimal fluorescence yield of the light-adapted state
- Fm :
-
maximal fluorescence yield of the dark-adapted state
- Fm′:
-
maximal fluorescence yield of the light-adapted state
- Fs :
-
steady-state fluorescence yield
- Fv :
-
variable fluorescence
- Fv/Fm :
-
maximal quantum yield of PSII photochemistry
- GGP:
-
greenhouse-grown plants
- GR:
-
glutathione reductase
- g s :
-
stomatal conductance
- LCP:
-
light-compensation point
- LSP:
-
light-saturation point
- MDA:
-
malondialdehyde
- NPQ:
-
nonphotochemical quenching
- PAR:
-
photosynthetically active radiation
- P Nmax :
-
light-saturated net photosynthetic rate
- P N :
-
net photosynthetic rate
- POD:
-
guaiacol peroxidase
- PPFD:
-
photosynthetic photon flux density
- PSII:
-
photosystem II
- qP :
-
photochemical quenching
- ROS:
-
reactive oxygen species
- SOD:
-
superoxide dismutase
- WAT:
-
weeks after treatment
- ΦPSII :
-
effective quantum yield of PSII photochemistry
References
Allen, D.J., Ort, D.R.: Impacts of chilling temperatures on photosynthesis in warm-climate plants. — Trends Plant Sci. 6: 36–42, 2001.
Almeselmani, M., Deshmukh, P.S., Sairam, R.K., Kushwaha, S.R., Singh, T.P.: Protective role of antioxidant enzymes under high temperature stress. — Plant Sci. 171: 382–388, 2006.
Ballantine, J.E.M., Forde, B.J.: The effect of light intensity and temperature on plant growth and chloroplast ultrastructure in soybean. — Amer. J. Bot. 57: 1150–1159, 1970.
Berry, J., Björkman, O.: Photosynthetic response and adaptation to temperature in higher plants. — Annu. Rev. Plant Physiol. 31: 491–543, 1980.
Bos, H.J., Neuteboom, J.H.: Morphological analysis and tiller number dynamics of wheat (Triticum aestivum L.): responses to temperature and light intensity. — Ann. Bot. 81: 131–139, 1998.
Chou, C.C., Chen, W.S., Huang, K.L. et al.: Changes in cytokinin levels of Phalaenopsis leaves at high temperature. — Plant Physiol. Biochem. 38: 309–314, 2000.
Demmig-Adams, B., Adams, W.W., III: Photoprotection and other responses of plants to high light stress. — Annu. Rev. Plant Physiol. Plant Mol. Biol. 43: 599–626, 1992.
Ding, X., Shi, G., Chen, W., Xu, X.: [Morphological studies on the development and cold resistance of the winter buds of Zizania caduciflora Hand.-Mazz.] — J. Wuhan Bot. Res. 11: 104–110, 1993. [In Chin.]
Dizengremel, P., Le Thiec, D., Bagard, M., Jolivet, Y.: Ozone risk assessment for plants: central role of metabolismdependent changes in reducing power. — Environ. Pollut. 156: 11–15, 2008.
Farage, P.K., Long, S.P.: The occurrence of photoinhibition in an over-wintering crop of oil-seed rape (Brassica napus L.) and its correlation with changes in crop growth. — Planta 185: 279–286, 1991.
Foyer, C.H., Halliwell, B.: The presence of glutathione and glutathione reductase in chloroplasts: a proposed role in ascorbic acid metabolism. — Planta 133: 21–25, 1976.
Friend, D.J.C.: Tillering and leaf production in wheat as affected by temperature and light intensity. — Can. J. Bot. 43: 1063–1076, 1965.
Genty, B., Briantais, J.M., Baker, N.R.: The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. — Biochim. Biophys. Acta 990: 87–92, 1989.
Gilmore, A.M.: Mechanistic aspects of xanthophylls cycledependent photoprotection in higher plant chloroplasts and leaves. — Physiol. Plant. 99: 197–209, 1997.
Govindachary, S., Bukhov, N.G., Joly, D., Carpentier, R.: Photosystem II inhibition by moderate light under low temperature in intact leaves of chilling-sensitive and -tolerant plants. — Physiol. Plant. 121: 322–333, 2004.
Groom, Q.J., Baker, N.R.: Analysis of light-induced depressions of photosynthesis in leaves of a wheat crop during the winter. — Plant Physiol. 100: 1217–1223, 1992.
Guo, D.P., Jiang, Y.T., Zeng, G.W., Shah, G.A.: Stem swelling of stem mustard, as affected by temperature and growth regulators. — Sci. Hort. 60: 153–160, 1994.
Guo, Y.P., Zhou, H.F., Zhang, L.C.: Photosynthetic characteristics and protective mechanisms against photooxidation during high temperature stress in two citrus species. — Sci. Hort. 108: 260–267, 2006.
Hunt, W., Thomas, V.: Growth and developmental responses of perennial ryegrass grown at constant temperature. II. Influence of light and temperature on leaf, tiller and root appearance. — Funct. Plant Biol. 12: 69–76, 1985.
Ishihara, K., Saitoh, K.: Diurnal courses of photosynthesis, transpiration, and diffusive conductance in the single-leaf of the rice plants grown in the paddy field under submerged condition. — Jpn. J. Crop Sci. 56: 8–17, 1987.
Islam, S., Izekor, E., Garner, J.O.: Effect of chilling stress on the chlorophyll fluorescence, peroxidase activity and other physiological activities in Ipomoea batatas L. genotypes. — Am. J. Plant Physiol. 6: 72–82, 2011.
Korner, C., Diemer, M.: In situ photosynthetic responses to light, temperature and carbon dioxide in herbaceous plants from low and high altitude. — Funct. Ecol. 1: 179–194, 1987.
Krause, G.H., Weis, E.: Chlorophyll fluorescence and photosynthesis-the basics. — Annu. Rev. Plant Physiol. Plant Mol. Biol. 42: 313–349, 1991.
Laloi, C., Apel, K., Danon, A.: Reactive oxygen signaling: the latest news. — Curr. Opin. Plant Biol. 7: 323–328, 2004.
Lee, D.H., Lee, C.B.: Chilling stress-induced changes of antioxidant enzymes in the leaves of cucumber: in gel enzyme activity assays. — Plant Sci. 159: 75–85, 2000.
Lichtenthaler, H.K.: Chlorophylls and carotenoids: Pigments of photosynthetic membranes. — Methods Enzymol. 148: 350–382, 1987.
Lidon, F.C., Henriques, F.S.: Oxygen metabolism in higher plant chloroplasts. — Photosynthetica 29: 249–279, 1993.
Martin, B., Ort, D.R., Boyer, J.S.: Impairment of photosynthesis by chilling-temperatures in tomato. — Plant Physiol. 68: 329–334, 1981.
McMaster, G.S., Wilhelm, W.W., Palic, D.B. et al.: Spring wheat leaf appearance and temperature: Extending the paradigm? — Ann. Bot. 91: 697–705, 2003.
Mooney, H.A., Björkman, O., Collatz, G.J.: Photosynthetic acclimation to temperature in desert shrub, Larrea divaricata. 1. Carbon-dioxide exchange characteristics of intact leaves. — Plant Physiol. 61: 406–410, 1978.
Nakano, Y., Asada, K.: Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. — Plant Cell Physiol. 22: 867–880, 1981.
Pastenes, C., Horton, P.: Effect of high temperature on photosynthesis in beans II. CO2 assimilation and metabolite contents. — Plant Physiol. 112: 1253–1260, 1996.
Patra, H.K., Kar, M., Mishra, D.: Catalase activity in leaves and cotyledons during plant development and senescence. — Biochem. Physiol. Pflanz. 172: 385–390, 1978.
Percival, G.C.: The use of chlorophyll fluorescence to identify chemical and environmental stress in leaf tissue of three oak (Quercus) species. — J. Arbor. 31: 215–227, 2005.
Perelman, A., Dubinsky, Z., Martínez, R.: Temperature dependence of superoxide dismutase activity in plankton. — J. Exp. Mar. Bio. Ecol. 334: 229–235, 2006.
Prado, C.H.B.A., De Moraes, J.A.P.V.: Photosynthetic capacity and specific leaf mass in twenty woody species of Cerrado vegetation under field conditions. — Photosynthetica 33: 103–112, 1997.
Qaderi, M.M., Reid, D.M.: Crop responses to elevated carbon dioxide and temperature. In: Singh, S.N. (ed.), Climate Change and Crops. — Springer-Verlag, Berlin Heidelberg, 1–18, 2009.
Ruelland, E., Zachowski, A.: How plants sense temperature. — Environ. Exp. Bot. 69: 225–232, 2010.
Ryle, G.J.A.: A comparison of leaf and tiller growth in seven perennial grasses as influenced by nitrogen and temperature. — Grass Forage Sci. 19: 281–290, 1964.
Salin, M.L.: Toxic oxygen species and protective systems of the chloroplast. — Physiol. Plant. 72: 681–689, 1988.
Salvucci, M.E., Osteryoung, K.W., Crafts-Brandner, S.J., Vierling, E.: Exceptional sensitivity of Rubisco activase to thermal denaturation in vitro and in vivo. — Plant Physiol. 127: 1053–1064, 2001.
Silim, S.N., Ryan, N., Kubien, D.S.: Temperature responses of photosynthesis and respiration in Populus balsamifera L.: acclimation versus adaptation. — Photosynth. Res. 104: 19–30, 2010.
Špundová, M., Slouková, K., Hunková, M., Nauš, J.: Plant shading increases lipid peroxidation and intensifies senescence-induced changes in photosynthesis and activities of ascorbate peroxidase and glutathione reductase in wheat. — Photosynthetica 43: 403–409, 2005.
Stewart, R.R.C., Bewley, J.D.: Lipid peroxidation associated with accelerated aging of soybean axes. — Plant Physiol. 65: 245–248, 1980.
Tenhunen, J.D., Lange, O.L., Gebel, J., Beyschlag, W., Weber, J.A.: Changes in photosynthetic capacity, carboxylation efficiency, and CO2 compensation point associated with midday stomatal closure and midday depression of net CO2 exchange of leaves of Quercus suber. — Planta 162: 193–203, 1984.
Turan, Ö., Ekmekçi, Y.: Activities of photosystem II and antioxidant enzymes in chickpea (Cicer arietinum L.) cultivars exposed to chilling temperatures. — Acta Physiol. Plant. 33: 67–68, 2011.
Van Kooten, O., Snel, J.: The use of chlorophyll fluorescence nomenclature in plant stress physiology. — Photosynth. Res. 25: 147–150, 1990.
Wahid, A., Gelani, S., Ashraf, M., Foolad, M.R.: Heat tolerance in plants: an overview. — Environ. Exp. Bot. 61: 199–223, 2007.
Weis, E.: Reversible heat-inactivation of the Calvin cycle: A possible mechanism of the temperature regulation of photosynthesis. — Planta 151: 33–39, 1981.
Wise, R.R., Olson, A.J., Schrader, S.M., Sharkey, T.D.: Electron transport is the functional limitation of photosynthesis in field-grown Pima cotton plants at high temperature. — Plant Cell Environ. 27: 717–724, 2004.
Yamasaki, T., Yamakawa, T., Yamane, Y. et al.: Temperature acclimation of photosynthesis and related changes in photosystem II electron transport in winter wheat. — Plant Physiol. 128: 1087–1097, 2002.
Yamori, W., Noguchi, K., Kashino, Y., Terashima, I.: The role of electron transport in determining the temperature dependence of the photosynthetic rate in spinach leaves grown at contrasting temperatures. — Plant Cell Physiol. 49: 583–591, 2008.
Zhang, F.L.: [Preliminary study on growth dynamics of doubleharvest Zizania latifolia: I. Autumn-harvest variety.] — Chin. Veg. 2: 31–33, 1991a. [In Chin.]
Zhang, F.L.: [Preliminary study on growth dynamics of doubleharvest Zizania latifolia: I. Summer-harvest variety.] — Chin. Veg. 3: 28–30, 1991b. [In Chin.]
Zhang, J.Z., Chu, F.Q., Guo, D.P. et al.: Cytology and ultrastructure of interactions between Ustilago esculenta and Zizania latifolia. — Mycol. Prog. 11: 499–508, 2012.
Zheng, Y., Mai, B., Wu, R. et al.: Acclimation of winter wheat (Triticum aestivum, cv. Yangmai 13) to low levels of solar irradiance. — Photosynthetica 49: 426–434, 2011.
Zinn, K.E., Tunc-Ozdemir, M., Harper, J.F.: Temperature stress and plant sexual reproduction: uncovering the weakest links. — J. Exp. Bot. 61: 1959–1968, 2010.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgements: This research was supported by the Special Fund for Agro-scientific Research in the Public Interest of China (No: 200903017-03).
Rights and permissions
About this article
Cite this article
Yan, N., Xu, X.F., Wang, Z.D. et al. Interactive effects of temperature and light intensity on photosynthesis and antioxidant enzyme activity in Zizania latifolia Turcz. plants. Photosynthetica 51, 127–138 (2013). https://doi.org/10.1007/s11099-013-0009-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-013-0009-2