Abstract
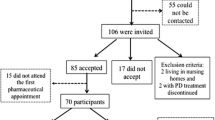
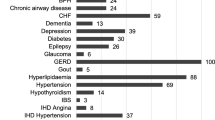
Background Since the new German Apothekenbetriebsordnung was released, medication therapy management (MTM) has increased in importance. MTM is intended to improve the quality of life of patients. Objectives The aim of this study was to improve the quality of life of patients with Parkinson’s disease through an MTM by a community pharmacist. Setting The patients were recruited in cooperation with the Deutsche Parkinson Vereinigung e.V. (dPV) in Germany. Methods All patients were evaluated at baseline (t0) and after a follow-up of 4 months (t1). During the intervention period, the pharmacists implemented an MTM with standardized pharmaceutical care. Main outcome measure The effects of the interventions were measured by the Unified Parkinson Disease Rating Scale (UPDRS) and the Movement Disorder Society Unified Parkinson Disease Rating Scale (MDS-UPDRS). Results In this study, 90 patients with Parkinson’s disease were included. The most common intervention was to find a therapy for untreated comorbidities. The UPDRS or MDS-UPDRS improved significantly after the intervention period by a median change rate of 1 (p < 0.05) or rather 2 (p < 0.05) compared to the baseline. Conclusion The study shows that the quality of life in Parkinson’s disease patients improved significantly through MTM.


Similar content being viewed by others
References
Deutsche Gesellschaft für Neurologie. 2Sk-Leitlinie Parkinson-Syndrome Diagnostik und Therapie [Internet]. 2012, last update 07/2015. http://www.awmf.org/uploads/tx_szleitlinien/030-010l_S2k_Parkinson-syndrome_Diagnostik_Therapie_2012_verlaengert.pdf Last accessed date 28 July 2015.
Visser M, Verbaan D, van Rooden S, Marinus J, van Hilten J, Stiggelbout A. A longitudinal evaluation of health-related quality of life of patients with Parkinson’s disease. Value Health. 2009;12:392–6.
Schröder S, Zöllner YF, Schaefer M. Drug related problems with Antiparkinsonian agents: consumer Internet reports versus published data. Pharmacoepidemiol Drug Saf. 2007;16:1161–6.
Rose O, Jaehde U, Leuner K, Ritter C, Müller K, Maschke T, et al. Statement der DPhG und der DPhG-FG Klinische Pharmazie: Implementierung des Medikationsmanagements als neue pharmazeutische Dienstleistung [Internet]. 2013. http://www.dphg.de/news-folder/detailansicht/implementierung-des-medikationsmanagements-als-neue-pharmazeutische-dienstleistung/f196c965646ee2fac82c21c165e1f939/#.VbexuPntlBc Last accessed date 28 July 2015.
World Medical Association. World Medical Association Declaration of Helsinki. Ethical principles for medical research involving human subjects. Bull World Health Organ. 2001;79:373.
Gola P, Schomerus R, Klug C. BDSG Bundesdatenschutzgesetz: Kommentar. Beck; 2010. ISBN 978-3-406-67176-0.
Richards M, Marder K, Cote L, Mayeux R. Interrater reliability of the Unified Parkinson’s Disease Rating Scale motor examination. Mov Disord. 1994;9:89–91.
Goetz CG, Tilley, BC, Shaftman SR, Stebbins GT, Fahn S, Martinez-Martin P, et al. Movement disorder society UPDRS revision task force movement disorder society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord. 2008;260:2129–2170.
Fricke U, Günther J, Zawinell A. Methodik der ATC-Klassifikation und der DDD-Festlegung. Wissenschaftliches Institut der AOK, Bonn. 2006. [ISBN 3-922093-40-X].
World Health Organization. The international classification of diseases (ICD) [Internet]. 2015. http://apps.who.int/classifications/icd10/browse/2015/en. Last Accessed date 28 July 2015.
Hoehn MM, Yahr MD. Parkinsonism: onset, progression and mortality. Neurology. 1967;17:427–42.
Schaefer M. Discussing basic principles for a coding system of drug-related problems: the case of PI-Doc. Pharm World Sci. 2002;24:120–7.
Holt S, Schmiedl S, Thürmann P. Potentially inappropriate medications in the elderly: the PRISCUS list. Deutsches Ärzteblatt Int. 2010;107:543.
Gallegher P, Ryan C, Byrne S, Kennedy J, O’Mahony D. STOPP (screening tool of older person’s prescriptions) and START (screening tool to alert doctors to right treatment). Consensus validation. Int J Clin Pharmacol Ther. 2008;2:72–83.
Thompson K, Kulkarni J, Sergejew AA. Reliability and validity of a new Medication Adherence Rating Scale (MARS) for the psychoses. Schizophr Res. 2000;3:241–7.
Samsa GP, Hanlon JT, Schmader KE, Weinberger M, Clipp EC, Uttech KM, et al. A summated score for the medication appropriateness index: development and assessment of clinimetric properties including content validity. J Clin Epidemiol. 1994;8:891–6.
Grosset KA, Bone I, Grosset DG. Suboptimal medication adherence in Parkinson’s disease. Mov Disord. 2005;11:1502–7.
Schröder S, Martus P, Odin P, Schaefer M. Drug related problems in Parkinson’s disease: the role of community pharmacists in primary care. Int J Clin Pharm. 2011;33:674–82.
Schröder S, Martus P, Odin P, Schaefer M. Impact of community pharmaceutical care on patient health and quality of drug treatment in Parkinson’s disease. Int J Clin Pharm. 2012;34:746–56.
Roughead EE, Semple SJ, Vitry AI. Pharmaceutical care services: a systematic review of published studies, 1990 to 2003, examining effectiveness in improving patient outcomes. Int J Pharm Pract. 2005;13:53–70.
Machado M, Bajcar J, Guzzo GC, Einarson TR. Sensitivity of patient outcomes to pharmacist interventions. Part I: systematic review and meta-analysis in diabetes management. Ann Pharmacother. 2007;41:1569–78.
Machado M, Nassor N, Bajcar JM, Guzzo GC, Einarson TR. Sensitivity of patient outcomes to pharmacist interventions. Part III: systematic review and meta-analysis in hyperlipidemia management. Ann Pharmacother. 2008;42:1195–207.
Funding
The author would like to thank the Apothekerstiftung Westfalen-Lippe for financial support of this study.
Conflicts of interest
None of the authors has any relevant conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Henrichsmann, M., Hempel, G. Impact of medication therapy management in patients with Parkinson’s disease. Int J Clin Pharm 38, 54–60 (2016). https://doi.org/10.1007/s11096-015-0206-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11096-015-0206-0