Abstract
Aim: To identify and review studies which have sought to define the pharmacokinetics of imipenem and cilastatin in patients receiving continuous renal replacement therapy (CRRT).
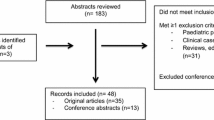
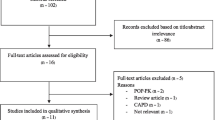
Method: Literature was primarily identified using Pharmline, Embase and Medline databases using the search terms ‘imipenem,’ ‘haemofiltration,’ ‘haemodialysis’ and ‘pharmacokinetics.’ Papers that discussed only intermittent haemodialysis were excluded.
Results: Seven papers were identified which described the pharmacokinetics of imipenem in patients receiving CRRT. Four different modes of CRRT were used. The methods of sampling, dosages used and assumptions made during pharmacokinetic calculations varied widely between the studies. Total body clearance of imipenem during CRRT in patients suffering from acute renal failure was found to range between 89 and 149 ml/min. Total body clearance of cilastatin ranged between 9 and 32 ml/min. Total body clearance of both imipenem and cilastatin was reduced in patients with chronic renal failure. Total body clearance of cilastatin was also reduced by impaired liver function. Dose recommendations made ranged between 500 mg 6-hourly and 500 mg 12-hourly.
Conclusions: The heterogeneity of the studies identified prevents them being analysed as a single group. For meaningful dosage recommendations to be made, further studies are required using larger populations and with more detail regarding liver dysfunction and duration of renal failure.
Similar content being viewed by others
References
SR Norrby K Alestig F Ferber JL Huber KH Jones FM Kahan et al. (1983a) ArticleTitlePharmacokinetics and tolerance of N-formimidoyl thienamycin (MK0787) in humans Antimicrob Agents Chemother 23 293–9 Occurrence Handle1:CAS:528:DyaL3sXhtlOitLw%3D
SR Norrby K Alestig B Björnegård LA Burman F Ferber JL Huber et al. (1983b) ArticleTitleUrinary recovery of N-formimidoyl thienamycin (MK0787) as affected by co-administration of of N-formimidoyl thienamycin dihydropeptidase inhibitor Antimicrob Agents Chemother 23 300–7 Occurrence Handle1:CAS:528:DyaL3sXhslant7Y%3D
AHFS Drug Information. Bethesda: American Society of Health-System Pharmacists Inc., 2003. ISBN 1-58528-039-9
JD Rogers MAL Meisinger F Ferber GB Calendra JL Demetriades JA Bland (1985) ArticleTitlePharmacokinetics of imipenem/ cilastatin in volunteers Rev Infec Dis 7 IssueIDSuppl 3 S435–46 Occurrence Handle1:CAS:528:DyaL2MXltFGnu7o%3D
GL Drussano HC Standiford (1985) ArticleTitlePharmacokinetic profile of imipenem/cilastatin in normal volunteers Am J Med 78 IssueIDSuppl 6a 47–53
British National Formulary 46. London: British Medical Association & Royal Pharmaceutical Society Great Britain, Sept 2003. ISBN 0-85369-556-3
JA Kellum JP Johnson D Kramer et al. (1998) ArticleTitleDiffusive versus convective treatment: effects on mediators of inflammation in patients with severe systemic inflammatory response syndrome Crit Care Med 26 1995–2000 Occurrence Handle9875910 Occurrence Handle1:STN:280:DyaK1M%2FptFWgug%3D%3D
I Tegeder F Bremer R Oelkers H Schobel J Schüttler K Brune et al. (1997) ArticleTitlePharmacokinetics of imipenem–cilastatin in critically ill patients undergoing continuous venovenous hemofiltration Antimicrob Agents Chemother 41 IssueID12 2640–5 Occurrence Handle9420033 Occurrence Handle1:CAS:528:DyaK2sXnvFWjs78%3D
B Mueller SK Scarim WL Macias (1993) ArticleTitleComparison of imipenem pharmacokinetics in patients with acute or chronic renal failure treated with continuous hemofiltration Am J Kidney Dis 21 IssueID2 172–9 Occurrence Handle8430678 Occurrence Handle1:STN:280:ByyC2MflvVc%3D
Hashimoto S, Honda M, Yamaguchi M, Sekimoto M, Tanaka Y. Pharmacokinetics of imipenem and cilastatin during continuous venovenous hemodialysis in patients who are critically iII. ASAIO J 1997; 84–8.
MC Vos HH Vincent EPF Yzerman (1992) ArticleTitleClearance of imipenem/cilastatin in acute renal failure patients treated by continuous hemodiafiltration (CAVHD) Intensive Care Med 18 282–5 Occurrence Handle10.1007/BF01706474 Occurrence Handle1527259 Occurrence Handle1:STN:280:By2H3cjhsVE%3D
E Keller H Fecht J Böhler P Schollmeyer (1989) ArticleTitleSingle-dose kinetics of imipenem/cilastatin during continuous arteriovenous haemofiltration in intensive care patients Nephrol Dial Transplant 4 640–5 Occurrence Handle2510062 Occurrence Handle1:STN:280:By%2BD2cvpslQ%3D
M Przechera D Bengel T Rider (1991) ArticleTitlePharmacokinetics of imipenem/cilastatin during continuous arteriovenous hemofiltration Contrib Nephrol 93 131–4 Occurrence Handle1802563 Occurrence Handle1:STN:280:By2B3c%2FptFA%3D
MG Dehne UR Kroh (1995) ArticleTitlelmipenem/cilastin dosage during acute renal fialure and hemofiltration Intensive Care Med 21 863 Occurrence Handle10.1007/BF01700974 Occurrence Handle8557879 Occurrence Handle1:STN:280:BymC38fmsVc%3D
M Kihara Y Ikeda K Shibata S Masumori H Ebira K Shiratori et al. (1994) Clin Nephrol 42 IssueID3 193–97 Occurrence Handle7994939 Occurrence Handle1:STN:280:ByqD1cjisFE%3D
Reeves DS. Human pharmacokinetics of N-formimidoylthienamycin alone and in combination with a dehydropeptidase inhibitor (MK-701) and probenecid. Proc 13th Int Congress Chemother 30–33
TP Gibson (1986) ArticleTitleRenal disease and drug metabolism: an overview Am J Kidney Dis 8 7–17 Occurrence Handle3524205 Occurrence Handle1:CAS:528:DyaL2sXkvV2isLY%3D
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cotton, A., Franklin, B.D., Brett, S. et al. Using Imipenem and Cilastatin During Continuous Renal Replacement Therapy. Pharm World Sci 27, 371–375 (2005). https://doi.org/10.1007/s11096-005-1636-x
Issue Date:
DOI: https://doi.org/10.1007/s11096-005-1636-x