Abstract
Identification of cytotoxic T lymphocyte (CTL) epitopes from additional tumor antigens is essential for the development of specific immunotherapy of malignant tumors. Neuritin, a recently discovered antigen overexpressed in astrocytoma, is considered to be a promising target for biological therapy. In the present study, we predicted and identified HLA-A2-restricted CTL epitopes from neuritin by using the following four-step procedure: (1) computer-based epitope prediction from the amino acid sequence of neuritin; (2) peptide-binding assay to determine the affinity of the predicted peptide with HLA-A2.1 molecule; (3) stimulation of primary T cell response against the predicted peptides in vitro; and (4) testing of the induced CTLs toward target cells expressing neuritin and HLA-A2.1. The results demonstrated that effectors induced by peptides of neuritin containing residues 13–21, 121–129 and 4–12 could specifically-secrete interferon-γ and lyse target cells. Our results indicate that these peptides are new HLA-A2.1-restricted CTL epitopes, and may serve as valuable tools for astrocytoma immunotherapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Astrocytoma is a common malignant primary brain tumor with a poor clinical prognosis in both adults and children [1–3]. The therapeutic options for the treatment of patients with astrocytoma are limited to three fundamental modalities: surgical resection, chemotherapy, and radiation therapy [4–6]. Cellular adoptive immunotherapy with specific CTLs has been recently used to treat malignant tumors [7–9].
Neuritin has been cloned and characterized as an important neurotrophin [10]. Recently, neuritin expression was found in astrocytomas and the expression level increased with astrocytoma pathologic grade, indicating that neuritin has an important role both in the promotion and in the progression of astrocytomas [11].
Anti-tumor vaccination is based on the existence of antigens, selectively or preferentially expressed by tumors, called tumor-associated antigens (TAAs) [12–14]. Vaccination with peptides derived from TAAs designed to stimulate specific T-cells is being a practicable approach evaluated in clinical trials [15–17]. To investigate the immune response elicited by neuritin CTL epitopes, the present study was undertaken to identify candidate CTL epitopes derived from neuritin.
Materials and methods
Cell lines and animals
The human TAP-deficient T2 cell line, BB7.2 cell line producing mAb against HLA-A2, human glioma cell line U251 and U87 (HLA-A2+), human breast cancer cell line MCF-7 (HLA-A2+) were purchased from the American Type Culture Collection (Manassas, VA). Cells were cultured in RPMI-1640 medium containing 10 % FBS (Gibco, with endotoxin level ≤ 10 EU/ml), penicillin (200 U/ml), and streptomycin (100 μg/ml). All cell lines mentioned previously were kept at 37 °C in a humidified atmosphere containing 5 % CO2. HLA-A*0201/Kb transgenic (Tg) mice, 8–12 weeks-old, were purchased from The Jackson Laboratory (USA). Mice were bred and maintained in specific pathogen-free (SPF) facilities. Animal experiments were performed in accordance with the guidelines of the Animal Care and Use Committee of Third Military Medical University.
Peptide synthesis
In the present study, two programs BIMAS (http://www-bimas.cit.nih.gov/molbio/hla_bind/) and SYFPEITH (http://www.syfpeithi.de/Scripts/MHCServer.dll/EpitopePrediction.htm) were used to identify the candidate HLA-A2 restricted CTL epitopes from the neuritin antigen. The candidate peptides validated by epitope prediction were then synthesized by Fmoc chemistry (Sangon, China), and purified by HPLC to a purity of >95 %. Lyophilized peptides were dissolved in DMSO at a concentration of 20 mg/ml and stored at −70 °C. The control peptide HBcAg (18–27) (FLPSDFFPSV) was synthesized and purified using the same methodology.
Peptide-binding assay
To determine whether the candidate epitopes can bind to HLA-A*0201 molecules, up-regulation of peptide-induced HLA-A*0201 molecules on T2 cells was examined. Briefly, according to previous reference, 1 × 106 T2 cells were incubated with 50 μM of the synthesized peptides in serum-free RPMI 1640 medium supplemented with β2-microglobulin (Sigma) at a concentration of 3 μg/ml for 16 h at 37 °C, 5 % CO2 [18]. Expression of HLA-A*0201 on T2 cells was then determined with the FACS Calibur flow cytometer (Becton–Dickinson, USA), by staining with primary anti-HLA-A2 Ab derived from BB7.2 and FITC-labeled goat-antimouse IgG (BD Biosciences Pharmingen, USA) secondary antibody. The data were analyzed using Cell Quest software (Becton–Dickinson, USA). The Fluorescence index (FI) was calculated as follows: FI = (mean FITC fluorescence with the given peptide − mean FITC fluorescence without peptide)/(mean FITC fluorescence without peptide). Samples were measured in triplicate and then mean FI was calculated. An octapeptide mHpa (519–526) (FSYGFFVI) derived from mouse Hpa was served negative control.
Measurement of the peptide/HLA-A*0201 complex stability
Briefly, T2 cells (106/ml) were incubated overnight with the candidate peptides, respectively, at a concentration of 20 μg/ml in serum-free medium supplemented with β2-microglobulin at a concentration of 3 μg/ml at 37 °C.
Thereafter, they were washed four times to remove free peptides, incubated with Brefeldin A (10 lg/ml) for 1 h to block cell surface expression of newly synthesized HLA-A2.1 molecules, washed and incubated at 37 °C for 0, 2, 4, 6, or 8 h. Subsequently, cells were stained with anti-HLA-A2 antibody from BB7.2 cells to evaluate the HLA-A2.1 molecule expression. For each time point, peptide induced HLA-A*0201 expression was evaluated by the formula mentioned above. Dissociation complex50 (DC50) was defined as the time required for the loss of 50 % of the HLA-A*0201/peptide complexes stabilized at time = 0.
RT-PCR analysis of neuritin expression
RT-PCR was used to analyze the expression of neuritin mRNA in cell lines. Total RNA was isolated from tumor cell lines using Tripure Isolation Regent Kit (Progema). Synthesis of cDNA was performed with 2 μg of total RNA with the aid of a reverse transcriptase Kit (Progema) and oligo(dT) primers. Two microliters RT product was amplified with PCR by using TaqDNA polymerase (Sangon, Shanghai) using standard procedures. The forward and reverse primer sequences were as follows: neuritin: sense primer, 5′-GTG CGA TGC AGT CTT TAA GTT-3′; anti-sense primer, 5′-GGG CTT TTC AGA CTG TTT GTT-3′; GAPDH: sense primer, 5′-GCA CCG TCA AGG CTG AGA AC-3′; antisense primer, 5′-ATG GTG GTG AAG ACG CCA GT-3′. Thirty amplification cycles were run: 1 min at 94 °C; 1 min at 60 °C; and 1 min at 72 °C. Cycling was ceased with a final extension of 10 min at 72 °C. RT-PCR products were then run on a gel and visualized with ethidium bromide.
Western blot analysis of neuritin expression
For Western blot analysis, proteins in the cell extracts were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) through an 8 % polyacrylamide gel and were then transferred onto a nitrocellulose membrane. The membrane was incubated with 5 % non-fat milk in PBS and later with anti-neuritin MAb for 2 h at room temperature. After washing, the membranes were incubated with an alkaline phosphatase-conjugated goat anti-mouse IgG antibody (Amersham Biosciences, Buckinghamshire, England) for 1 h at room temperature. Immunoreactive bands were detected using the ECL Western blot analysis system (Amersham Biosciences, Buckinghamshire, England).
Dendritic cell generation from human peripheral blood precursors
PBMCs were isolated from healthy HLA-A2+ donors by Ficoll–Hypaque density gradient centrifugation (TBC company, Tianjing, China) and then seeded into culture flasks in RPMI-1640 medium supplemented with penicillin (100 U/ml), streptomycin (100 μg/ml), and 10 % FBS. After monocytes adhered (incubation for 2 h), the nonadherent cells were collected and frozen in freeze medium (60 % RPMI-1640 and 30 % FBS, 10 % DMSO) for later use in CTL assays. The adherent cells were cultured for 5 days in RPMI-1640 containing 1,000 U/ml of granulocyte–macrophage colony-stimulating factor (R&D Systems, Inc., Minneapolis, MN) and interleukin-4 (IL-4; R&D Systems, Inc.) and were the cultured for an additional 2 days in the presence of 1,000 U/ml of tumor necrosis factor α (R&D Systems, Inc.) to induce final maturation. After 7 days of culture, the mature DCs were harvested and analyzed for DC typical phenotypes by FACS analysis.
Induction of peptide-specific CTL with synthetic peptides
Briefly, DCs were loaded with different peptides at a final concentration of 100 μg/ml for 4 h and were then irradiated with 20 Gy, which prevented all outgrowths in the control cultures. Autologous T cells were restimulated every 7 days with the previously mentioned peptide-pulsed DCs to generate peptide-specific CTLs. Recombinant interleukin 2 (IL-2) at a concentration of 20 U/ml was added to the culture medium on day 3 after every stimulation. Cytotoxic T lymphocyte activity was then assessed on day 23 by a 4 h 51Cr release assay. Effectors generated from negative peptide-pulsed DCs were used as controls.
ELISPOT assay
IFN-γ secretion of effectors was assayed by enzyme-linked immunospot (ELISPOT). Multiscreen 96-well assay plates (Dakewe, Shenzhen, China) were precoated overnight at 4 °C with anti-IFN-γ antibody according to the manufacturer’s instruction. After washing with PBST (PBS-0.05 % Tween 20), plates were blocked for 1 h at 37 °C with PBS/1 % BSA. Cytotoxic T lymphocyte effectors from human HLA-A2+ donors were plated in triplicate wells at a density of 1 × 105/100 μl in RPMI-1640 medium. Plates were cultured overnight, washed extensively with PBST, and incubated with anti-IFN-γ mAb for 1 h at 37 °C. After washing, goat anti-biotin antibodies (Dakewe) were added, and the plates were incubated for 1 h at 37 °C. Thirty microliters of activator solution (Dakewe) was added to develop spots, and after 10–30 min, the plates were washed with distilled water to stop the reaction. After being air-dried, the number of spots in each well was counted using the Bioreader 4000 PRO-X (Bio-Sys; Germany).
Cytotoxicity assay
To evaluate the levels of CTL activity, a standard 4-h 51Cr release assay was used. Briefly, target cells were incubated with 51Cr (100 μCi per 1 × 106 cells) for 2 h in a 37 °C water bath. After incubation with 51Cr, target cells were washed three times with PBS, resuspended in RPMI-1640 medium, and mixed with effector cells at a 25:1, 50:1 or 100:1 of effector to target (E/T) ratio. Assays were performed in triplicate for each sample at each ratio in a 96-well round-bottomed plate. After a 4-h incubation, the supernatants were harvested, and the amount of released 51Cr was measured with a gamma counter. The percent specific lysis was calculated according to the following formula:
Specific lysis = (experimental release − spontaneous release)/(maximal release − spontaneous release) × 100 %
Analysis of in vivo immunogenicity
HLA-A*0201/Kb mice were immunized with 100 μg of various peptides prepared in incomplete Freund’s adjuvant (IFA) and boosted once a week for three times. As a control, mice were injected with an IFA emulsion without peptide. 7 days after immunization, splenocytes from injected animals were cultured and used as effector cells.
Statistical analysis
Results were expressed as mean ± SEM. Analysis of Student’s t test were performed to determine effects of the treatments. A difference was considered significance level of P < 0.05.
Results
Prediction of putative CTL epitopes restricted with HLA-A*0201
To predict the HLA-A*0201-restricted CTL epitopes of neuritin, two programs (BIMAS and SYFPEITHI), were used to scan the complete amino acid sequence of this antigen. Four highest-scored 9-amino-acid peptides were chosen as candidates for further identification (Table 1). These peptides were chemically synthesized, purified, and identified. The molecular weight of each peptide determined by mass spectrometry assay was similar to its theoretical molecular weight, and the purities of these peptides were all >95 % (data not shown).
MHC peptide-binding and stability assay
The binding affinity and stability of these peptides to HLA-A2.1 was determined by using antigen processing-deficient T2 cells because their enhanced HLA-A2.1. As shown in Table 2, all of the peptides synthesized were bound to HLA-A2.1 molecules but with different affinity and stability. Of four peptides selected, neuritin13–21 up-regulated the HLA-A2.1 molecular expression and showed high affinity and stability to HLA-A2.1, whereas neuritin121–129 and neuritin4–12 showed moderate affinity and neuritin127–135 only had low affinity and stability to the molecule.
Expression of neuritin in target cells
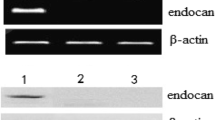
The expression of neuritin mRNA and protein in cell lines in this study was analyzed by RT-PCR and Western blot. As shown in Fig. 1, neuritin mRNA and protein were detected in U251 and U87 cell lines. However, neuritin mRNA and protein could not be detected in MCF-7 and autologous lymphocytes.
Expression of neuritin in different target cells. Total RNA was isolated from tumor cell lines using Tripure Isolation Regent Kit. Two microliters RT product was amplified with PCR by using TaqDNA polymerase (using standard procedures). RT-PCR products were then run on a gel and visualized with ethidium bromide. For Western blot analysis, proteins in the cell extracts were separated by SDS-PAGE and were then analyzed with anti-neuritin MAb (antibodies-online company). 1 U251 cells; 2 U87 cells; 3 MCF-7 cells; 4 autologous lymphocytes
Enzyme-linked immunospot (ELISPOT) assay for IFN-γ
Since CTLs are known to produce the Th1 cytokine IFN-γ, peptide-specific T cells were enumerated by measuring IFN-γ-producing cells by ELISPOT assay. As shown in Fig. 2, neuritin13–21, neuritin121–129 and neuritin4–12 peptides were found to generate a strong peptide-specific T cell response by virtue of their ability to induce increased frequencies of IFN-γ-producing T cells, as compared to the negative peptide control. These results suggest that neuritin peptide vaccines can increase IFN-γ secretion by effectors and enhance the Th1 immune response.
Specific IFN-γ by ELISPOT assay. The PBMCs of human HLA-A2+ donors were obtained and then cultured in RPMI 1640 supplemented with 10 % FCS, 100 U/ml penicillin, and 100 μg/ml streptomycin. Dendritic cell were generated, and loaded with different peptides at a final concentration of 100 μg/ml for 4 h and were then irradiated with 20 Gy, which prevented all outgrowths in the control cultures. Autologous T cells were restimulated every 7 days with the peptide-pulsed DCs to generate peptide-specific CTLs. The IFN-γ secretion was then assessed on day 23. Experiments performed in triplicate showed consistent results. Compared with controls, P < 0.05
Induction of CTLs efficiently in vitro
PBMCs from four HLA-A2.1+ donors were stimulated with synthetic peptides with the previously published method for CTL induction [19]. Of four tested, neuritin13–21, neuritin121–129 and neuritin4–12 peptides were able to elicit neuritin-specific CTLs, which could lyse target cells expressing neuritin and HLA-A2.1 (Fig. 3).
Specific lysis of CTLs against target cells. Target cells were incubated with 51Cr (100 μCi per 1 × 106 cells) for 2 h in a 37 °C water bath. After incubation with 51Cr, target cells were washed three times with PBS, resuspended in RPMI-1640 medium, and mixed with effector cells at a 25:1, 50:1 or 100:1 of effector to target (E/T) ratio. After a 4-h incubation, the supernatants were harvested, and the amount of released 51Cr was measured with a gamma counter. Compared with controls, P < 0.05
Inhibition of the recognition of effectors by anti-HLA2 antibody
To determine whether the peptides induced effectors recognized target cells in an HLA-A2-restricted manner, the mAbs against HLA-A2 were used to block recognition by effectors. Our results showed that the anti-HLA-A2 antibody could significantly eliminate the cytotoxicity of the effectors against neuritin and HLA-A2 positive cells (Fig. 4), which implied that the induced effectors lysed target cells in an HLA-A2-restricted manner.
Inhibited recognition of induced cells by anti-HLA-A2 antibody. Target cells were incubated with 100 μl anti-HLA-A2 antibody (functionally blocking mAb, from BB7.2 cell hybridoma supernate) for 1 h at 4 °C. The cytotoxic activities of CTLs were determined against target cells at various E/T ratios using 51Cr release assay. Experiments performed in triplicate showed consistent results. Compared with controls, P < 0.05
In vivo induction of epitope-specific CTLs in vivo
We investigated whether peptides could induce immunity in vivo. HLA-A*0201/Kb mice were immunized with 100 μg of various peptides prepared in incomplete Freund’s adjuvant (IFA). The cytolytic assay showed that CTLs primed from neuritin13–21, neuritin121–129 and neuritin4–12 immunized mice could lyse neuritin and HLA-A2.1 positive cells with high efficiency (Fig. 5). These results suggested that the peptides could also achieve higher immunogenicity in vivo.
In vivo induction of epitope-specific CTLs in vivo. HLA-A*0201/Kb mice were immunized with 100 μg of various peptides prepared in incomplete Freund’s adjuvant (IFA) and boosted once a week for three times. As a control, mice were injected with an IFA emulsion without peptide. 7 days after immunization, splenocytes from injected animals were cultured and used as effector cells. The cytotoxic activities of CTLs were determined against target cells at various E/T ratios using 51Cr release assay. Experiments performed in triplicate showed consistent results. Compared with controls, P < 0.05
Discussion
Survival in the majority of high-grade astrocytoma (HGA) patients is very poor, with only a rare population of long-term survivors [20–22]. During the last decades, great progress has been made in the treatment of patients with astrocytoma [23–25]. Many astrocytoma can be cured or eliminated using the therapeutic options such as surgical resection, chemotherapy, and (or) radiation therapy. However, as for advanced astrocytoma, the modalities mentioned above do not yield good results [26–28].
Over the past few years, the analysis of spontaneous immune responses to autologous tumors in cancer patients has allowed the identification of several kinds of tumor-associated antigens that can be the targets for tumor specific immune responses based on the recognition of tumor antigen by CTLs in an MHC-class I/peptide complex-restricted manner [29–31]. Therefore, cancer-specific immunotherapy has become an attractive fourth-therapeutic approach against carcinomas. Among them, one of the most relevant for the development of tumor immunotherapy is peptide-based, cancer-specific immunotherapy using the group of the tumor antigens [32, 33].
Neuritin has been cloned and characterized as an important neurotrophin. The expression of neuritin is closely associated with the growth of afferent nerves and the development of dendrites, axons, and synapses [34]. Recently, neuritin expression was found in tissues besides nervous system and tumor tissues [35]. Moreover, the neuritin protein was highly expressed in astrocytomas and increased with pathologic grade, indicating that neuritin has an important role both in the promotion and in the progression of astrocytomas.
In this study, we first predicted four candidate epitopes from neuritin antigen by using HLA-A2.1-restricted epitope prediction algorithms based on long distance prediction systems SYFPEITHI and BIMAS. Secondly, peptide-binding assay was used to determine the affinity of every epitope with HLA-A2.1 and the results showed that neuritin13–21 had high affinity to HLA-A2.1, whereas neuritin121–129 and neuritin4–12 showed moderate affinity to the molecule. Thirdly, cytotoxic activity of CTLs was measured by ELISPOT and 51Cr release assay. The results demonstrated that neuritin13–21, neuritin121–129 and neuritin4–12 could elicit CTLs to lyse target cells in an HLA-A2.1-restricted manner. Lastly, we immunized the HLA-A*0201/Kb mice with various peptides and found neuritin13–21, neuritin121–129 and neuritin4–12 could also elicit CTLs to lyse neuritin and HLA-A2.1 positive target cells. These results suggested that the peptides had the potential of immunogenicity in vivo.
In conclusion, our results suggest that neuritin13–21, neuritin121–129 and neuritin4–12 might be capable of inducing HLA-A2.1-restricted CD8+ CTL, which would be lethal for neuritin and HLA-A2.1 positive cells. Therefore, identification of neuritin peptide would contribute to the design of epitope-based vaccine for astrocytomas immunotherapy.
References
Tchoghandjian A, Fernandez C, Colin C, El Ayachi I, Voutsinos-Porche B, Fina F et al (2009) Pilocytic astrocytoma of the optic pathway: a tumour deriving from radial glia cells with a specific gene signature. Brain 132(Pt 6):1523–1535
Sathornsumetee S, Cao Y, Marcello JE, Herndon JE 2nd, McLendon RE, Desjardins A et al (2008) Tumor angiogenic and hypoxic profiles predict radiographic response and survival in malignant astrocytoma patients treated with bevacizumab and irinotecan. J Clin Oncol 26(2):271–278
Tehrani M, Friedman TM, Olson JJ, Brat DJ (2008) Intravascular thrombosis in central nervous system malignancies: a potential role in astrocytoma progression to glioblastoma. Brain Pathol 18(2):164–171
Hu W, Shen F, Chen G, Shen G, Liu W, Zhou J (2009) Possible involvement of brain tumour stem cells in the emergence of a fast-growing malignant meningioma after surgical resection and radiotherapy of high-grade astrocytoma: case report and preliminary laboratory investigation. J Int Med Res 37(1):240–246
Chamberlain MC, Johnston S (2009) Salvage chemotherapy with bevacizumab for recurrent alkylator-refractory anaplastic astrocytoma. J Neurooncol 91(3):359–367
Ooba H, Abe T, Kamida T, Anan M, Momii Y, Tokuuye K et al (2009) Malignant fibrous histiocytosis after high-dose proton radiation therapy for anaplastic astrocytoma. J Clin Neurosci 16(12):1641–1643
Yang I, Han S, Parsa AT (2009) Heat-shock protein vaccines as active immunotherapy against human gliomas. Expert Rev Anticancer Ther 9(11):1577–1582
De Vleeschouwer S, Fieuws S, Rutkowski S, Van Calenbergh F, Van Loon J, Goffin J et al (2008) Postoperative adjuvant dendritic cell-based immunotherapy in patients with relapsed glioblastoma multiforme. Clin Cancer Res 14(10):3098–3104
Zhang JG, Kruse CA, Driggers L, Hoa N, Wisoff J, Allen JC et al (2008) Tumor antigen precursor protein profiles of adult and pediatric brain tumors identify potential targets for immunotherapy. J Neurooncol 88(1):65–76
Krishnamoorthy RR, Agarwal P, Prasanna G, Vopat K, Lambert W, Sheedlo HJ et al (2001) Characterization of a transformed rat retinal ganglion cell line. Brain Res 86(1–2):1–12
Zhang L, Zhao Y, Wang CG, Fei Z, Wang Y, Li L et al (2011) Neuritin expression and its relation with proliferation, apoptosis, and angiogenesis in human astrocytoma. Med Oncol 28(3):907–912
Yamaguchi S, Tatsumi T, Takehara T, Sasakawa A, Hikita H, Kohga K et al (2008) Dendritic cell-based vaccines suppress metastatic liver tumor via activation of local innate and acquired immunity. Cancer Immunol Immunother 57(12):1861–1869
Wang B, He J, Liu C, Chang LJ (2006) An effective cancer vaccine modality: lentiviral modification of dendritic cells expressing multiple cancer-specific antigens. Vaccine 24(17):3477–3489
Neeley YC, Arredouani MS, Hollenbeck B, Eng MH, Rubin MA, Sanda MG (2008) Partially circumventing peripheral tolerance for oncogene-specific prostate cancer immunotherapy. Prostate 68(7):715–727
Cao DY, Yang JY, Yue SQ, Tao KS, Song ZS, Wang DS et al (2009) Comparative analysis of DC fused with allogeneic hepatocellular carcinoma cell line HepG2 and autologous tumor cells as potential cancer vaccines against hepatocellular carcinoma. Cell Immunol 259(1):13–20
Rinaldi M, Iurescia S, Fioretti D, Ponzetto A, Carloni G (2009) Strategies for successful vaccination against hepatocellular carcinoma. Int J Immunopathol Pharmacol 22(2):269–277
Hirohashi Y, Torigoe T, Inoda S, Kobayasi J, Nakatsugawa M, Mori T et al (2009) The functioning antigens: beyond just as the immunological targets. Cancer Sci 100(5):798–806
Han JF, Zhao TT, Liu HL, Lin ZH, Wang HM, Ruan ZH et al (2006) Identification of a new HLA-A*0201-restricted cytotoxic T lymphocyte epitope from CML28. Cancer Immunol Immunother 55(12):1575–1583
Chen T, Tang XD, Wan Y, Chen L, Yu ST, Xiong Z et al (2008) HLA-A2-restricted cytotoxic T lymphocyte epitopes from human heparanase as novel targets for broad-spectrum tumor immunotherapy. Neoplasia 10(9):977–986
Ritz R, Muller M, Dietz K, Duffner F, Bornemann A, Roser F et al (2008) Hypericin uptake: a prognostic marker for survival in high-grade glioma. J Clin Neurosci 15(7):778–783
Bay JO, Linassier C, Biron P, Durando X, Verrelle P, Kwiatkowski F et al (2007) Does high-dose carmustine increase overall survival in supratentorial high-grade malignant glioma? An EBMT retrospective study. Int J Cancer 120(8):1782–1786
Datta NR, Pasricha R, Gambhir S, Phadke RV, Prasad SN (2004) Postoperative residual tumour imaged by contrast-enhanced computed tomography and 201Tl single photon emission tomography: can they predict progression-free survival in high-grade gliomas? Clin Oncol 16(7):494–500
Koul D (2008) PTEN signaling pathways in glioblastoma. Cancer Biol Ther 7(9):1321–1325
Kargiotis O, Rao JS, Kyritsis AP (2006) Mechanisms of angiogenesis in gliomas. J Neurooncol 78(3):281–293
Gunny RS, Hayward RD, Phipps KP, Harding BN, Saunders DE (2005) Spontaneous regression of residual low-grade cerebellar pilocytic astrocytomas in children. Pediatr Radiol 35(11):1086–1091
Fukushima T, Takeshima H, Kataoka H (2009) Anti-glioma therapy with temozolomide and status of the DNA-repair gene MGMT. Anticancer Res 29(11):4845–4854
Wirth T, Samaranayake H, Pikkarainen J, Maatta AM, Yla-Herttuala S (2009) Clinical trials for glioblastoma multiforme using adenoviral vectors. Curr Opin Mol Ther 11(5):485–492
de Groot J, Milano V (2009) Improving the prognosis for patients with glioblastoma: the rationale for targeting Src. J Neurooncol 95(2):151–163
Homma S, Harada M, Yano H, Ogasawara S, Shichijo S, Matsueda S et al (2006) Identification of squamous cell carcinoma antigen-derived peptides having the capacity of inducing cancer-reactive CTLs in HLA-A24+ cancer patients. Int J Oncol 29(3):577–587
Elkord E, Williams PE, Kynaston H, Rowbottom AW (2005) Differential CTLs specific for prostate-specific antigen in healthy donors and patients with prostate cancer. Int Immunol 17(10):1315–1325
Azuma K, Sasada T, Takedatsu H, Shomura H, Koga M, Maeda Y et al (2004) Ran, a small GTPase gene, encodes cytotoxic T lymphocyte (CTL) epitopes capable of inducing HLA-A33-restricted and tumor-reactive CTLs in cancer patients. Clin Cancer Res 10(19):6695–6702
Lazoura E, Apostolopoulos V (2005) Insights into peptide-based vaccine design for cancer immunotherapy. Curr Med Chem 12(13):1481–1494
Belnoue E, Guettier C, Kayibanda M, Le Rond S, Crain-Denoyelle AM, Marchiol C et al (2004) Regression of established liver tumor induced by monoepitopic peptide-based immunotherapy. J Immunol 173(8):4882–4888
Cappelletti G, Galbiati M, Ronchi C, Maggioni MG, Onesto E, Poletti A (2007) Neuritin (cpg15) enhances the differentiating effect of NGF on neuronal PC12 cells. J Neurosci Res 85(12):2702–2713
Kojima N, Shiojiri N, Sakai Y, Miyajima A (2005) Expression of neuritin during liver maturation and regeneration. FEBS Lett 579(21):4562–4566
Acknowledgments
This work was supported by National Natural Science Foundation of China (NSFC No.81200908).
Conflict of interest
None of the authors has any potential financial conflict of interest related to this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Zhao Yang and Tianzhi Zhao have contributed equally to this study.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Yang, Z., Zhao, T., Liu, Y. et al. Identification of new HLA-A*0201-restricted cytotoxic T lymphocyte epitopes from neuritin. J Neurooncol 114, 51–58 (2013). https://doi.org/10.1007/s11060-013-1167-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-013-1167-6