Abstract
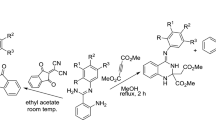
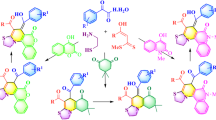
A convenient synthesis of functionalized 2-iminothiazolidin-4-ones and thiadiazoloquinazolinones has been achieved by the reaction of dialkyl acetylenedicarboxylates (DAAD) with 1-pheny-3-pyridin-2-yl-thiourea (1) and 3-amino-2-mercapto-3H-quinazolin-4-one (8), respectively. The starting materials for these processes were prepared from phenyl isothiocyanate, 2-aminopyridine for (1) and anthranilic acid, carbon disulfide, hydrazine for (8). Treatment of these compounds with DAAD resulted in the formation of an intermediate that readily underwent intramolecular cyclization to afford functionalized five membered heterocyclic rings was formed.
Similar content being viewed by others
References
Singh SP, Parmar SS, Raman K, Stenberg VI (1981) Chemistry and biological activity of thiazolidinones. Chem Rev 81: 175–203. doi:10.1021/cr00042a003
Brown FC (1961) 4-Thiazolidinones. Chem Rev 61: 463–521. doi:10.1021/cr60213a002
Vogeli U, Von Philipsborn W, Nagarajan K, Nair MD (1978) Structures of addition products of acetylenedicarboxylic acid esters with various dinucleophiles. An application of C, H-spin-coupling constants. Helv Chim Acta 61: 607–617. doi:10.1002/hlca.19780610207
Acheson RM, Wallis JD (1981) Addition reactions of heterocyclic compounds. Part 74. Products from dimethyl acetylenedicarboxylate with thiourea, thioamide and guanidine derivatives. J Chem Soc Perkin Trans 1: 415–422. doi:10.1039/P19810000415
Giammona G, Neri M, Carlisi B, Pazzo A, La Rosa C (1991) Reactions of azoesters and dimethyl acetylenedicarboxylate with 3- methyl-1,2,4-triazole-5-thione. J Heterocycl Chem 28: 325–327
Coen S, Ragonnet B, Vieillescazes C, Roggero JP (1985) Reactivity of conjugated enaminothioamides toward activated acetylenic compounds. Heterocycles 23: 1225–1228
Pandeya SN, Sriram D, Nath G, De Clercq E (1999) Synthesis, antibacterial, antifungal and anti-HIV activities of norfloxacin Mannich bases. Pharm Acta Helv 74: 11–17. doi:10.1016/S0031-6865(99)00010-2 Medline
Valdes-Martinez J, Hernandez-Ortega S, Espinosa-peres G, Presto CA, Hermetet AK, Haslow KD, Ackerman LJ, Szczepura LF, Goldberg KI, Kaminsky W, West DX (2002) Structural, spectral and thermal studies of substituted N-(2-pyridyl)-N’-phenylthioureas. J Mol Struct 608: 77–87. doi:10.1016/S0022-2860(01)00934-6
Ahmadi A, Saidi K, Khabazzadeh H, Sheibani H, Molahoseini A (2007) Synthesis of phosphorus ylides using 2-(1,3-Dioxo-1,3- dihydro isoindol-2-yl)-N-phenylacetamide derivatives. Phosphorus Sulfur 182: 1225–1231. doi:10.1080/10426500601160520
Berseneva VS, Morzherin YY, Dehaen W, Luyten I, Bakulev VA (2001) Reaction of heterocyclic thioamides with dimethyl acetylenedicarboxylate. Synthesis of novel 2-azolyl-5-methoxycarbonylmethylene thiazolin-4-ones. Tetrahedron 57: 2179–2184. doi:10.1016/S0040-4020(01)00049-7
Hendrickson JB, Rees R, Templeton JF (1964) A new general heterocycle synthesis, use of acetylenedicarboxylic esters. J Am Chem Soc 86: 107–111. doi:10.1021/ja01055a024
Esmaili AA, Ghereghloo M, Islami MR, Bijanzadeh HR (2003) One-pot synthesis of stable phosphonium ylides using 2-aminothiopheno. Tetrahedron 59: 4785–4788. doi:10.1016/S0040-4020(03)00693-8
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahmadi, A., Saidi, K. & Khabazzadeh, H. An efficient synthesis of substituted 2-iminothiazolidin-4-one and thiadiazoloquinazolinone derivatives. Mol Divers 13, 353–356 (2009). https://doi.org/10.1007/s11030-009-9124-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-009-9124-1