Abstract
The aim of this study was to determine uranium concentrations in common nettle (Urtica dioica) plants and corresponding soils samples which were collected from the area of phosphogypsum stockpile in Wiślinka (northern Poland). The uranium concentrations in roots depended on its concentrations in soils. Calculated BCF and TF values showed that soils characteristics and air deposition affect uranium absorption and that different uranium species have different affinities to U. dioica plants. The values of 234U/238U activity ratio indicate natural origin of these radioisotopes in analyzed plants. Uranium concentration in plants roots is negatively weakly correlated with distance from phosphogypsum stockpile.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Phosphogypsum is a byproduct from phosphoric acid production from phosphate rocks usually stored in specially designated areas. The phosphogypsum stockpile in Wiślinka (northern Poland) is located between the Martwa Wisła river and farm fields, close to the Gdańsk agglomeration. It is considered to be one of the major contaminators of Vistula river delta. The stockpile contains about 16 million tons of phosphogypsum. Phosphate rocks used for phosphoric acid production are characterized by high content of natural alpha radioactive elements, especially from uranium decay series (210Po, 226Ra, 234U, 238U) and beta emitter (210Pb). In the process of phosphoric acid production about 80 % of uranium is associated with the phosphoric acid fraction, while about 90 % of the 210Po and 210Pb is bound to the phosphogypsum fraction [1–3]. Phosphogypsum in Wiślinka might have serious radiological impact on the local environment. Radionuclides might be leached by wet precipitation and transported through groundwaters to plants where they are accumulated [4–9].
Natural uranium consists of three alpha radioactive isotopes: 99.2745 % of 238U, 0.7200 % of 235U, and 0.0054 % of 234U [10]. Environmental occurrence of uranium can be a result of human activities. As a major uranium sources in the environment can be considered nuclear industry, combustion of fossil fuels, production and use of phosphorous fertilizers or use of depleted uranium for military purposes [11–13]. Normally in water 234U and 238U radionuclides are not in the radioactive state of equilibrium. In groundwaters, the average values of the activity ratio between 234U and 238U are in the range from 0.51 to 9.02, in salt water from 1.11 to 5.14, in river water from 1.00 to 2.14, in river suspension from 0.80 to 1.00, in oceanic water 1.14 and in Baltic water 1.17 [14–16]. In rocks, soils and sediments the uranium isotopes 234U and 238U are in relative equilibrium (from 0.84 to 1.19 for oceanic basalts, from 0.70 to 1.16 for phosphorite concretions, from 0.83 to 1.28 for oceanic sediments and from 0.98 to 1.04 for Baltic sediments) [17, 18].
The main factors that contribute to uranium content and leaching ability in soil environment are the proximity of the water to the uranium source, the degree of hydraulic isolation of the water from dilution by fresher water, climatic effects and their seasonal variability (evapotranspiration), the pH and E h of the water (uranium is mobilized under oxidizing conditions and immobilized under reducing conditions), concentrations of important species that can form either strong complexes or precipitate insoluble uranium minerals (e.g., carbonate, phosphate, vanadate, fluoride, sulfate, silicate, calcium, potassium) and the presence of highly sorbents like organic matter or Fe/Mn/Ti oxyhydroxides [19, 20]. In case of plants large number of factors control metal accumulation and bioavailability such as soil and climatic conditions, plant genotype and agronomic management, including: active/passive transfer processes, sequestration and speciation, redox states, the type of plant root system and the response of plants to elements in relation to seasonal cycles [21, 22]. Structure of the soil is also considered as one of the major factors that contribute to extent of the metals taken up by the plants. Such factors as clay particles, metal solubility controlled by pH, amount of metals cations exchange capacity, organic carbon content and oxidation state of the system are also important in metals availability [22].
The main aim of this work was to establish a possible use of Urtica dioica (common nettle) plants as uranium contamination bioindicator in the area of phosphogypsum stockpile by analysis of 234U and 238U in plants samples and corresponding soils and to examine the impact of phosphogypsum stockpile on the surrounding environment. Additionally, the values of the 234U/238U activity ratio and BCF (biocencentration) and TF (translocation) factors are calculated in order to define both the possible uranium sources and level of their accumulation in plants.
Experimental
Collection of samples
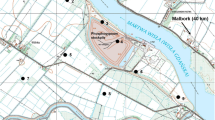
The U. dioica plants samples along with corresponding soils were collected from the area of phosphogypsum stockpile in Wiślinka (northern Poland) in October 2013. The locations of the analyzed plants and soils samples are presented in Fig. 1. Control samples were collected in Malbork (Pomeranian Voivodeship). As the aim of the study was to collect multiple samples in various distances from stockpile, U. dioica due to it commonness seemed to be the most appropriate plant.
Preparation of samples
Collected plants were divided into green part and root. Roots were washed with double deionized water in order to remove soil particles. Green parts were cleaned from soils particles but not washed in order to retain possible aerial contamination (wet and dry deposition and phosphogypsum particles). Before analysis, each sample was air dried, homogenized using mortar and dried in 60 °C. Additionally, soils samples were passed through 0.25 mm sieve. From homogenized sample three subsamples were weighted and enriched with approximately 15 mBq (plants) and 30 mBq (soils) of 232U as the yield tracer.
Analysis of samples
Radioanalytical proceducre
The analytical procedure of determination of uranium radioisotopes (234U, 238U) in analyzed samples was based on the mineralization of soils samples in concentrated acids HNO3 and HCl, mineralization of plants samples in concentrated HNO3 acid with H2O2 addition and separation on the anion exchange resins according to method established by Skwarzec and Boryło [23, 24].
Instrument for analysis
The activities of 234U and 238U were measured using alpha spectrometer (Alpha Analyst S470) equipped with a surface barrier PIPS detector with an active surface of 300 mm2 placed in a vacuum chamber connected to a 1024 multichannel analyzer (Canberra–Packard, USA). Detector yield ranged from 0.30 to 0.40. In most of the used detectors with a surface of 300 mm2, the resolution was 17–18 keV. Minimal detectable activity (MDA) was measured to be 0.2 mBq for both 238U and 234U [25]. The results of 234U and 238U concentrations in analyzed samples are given with expanded standard uncertainty calculated for a 95 % CI. The concentrations of uranium isotopes in the IAEA-330 and IAEA-375 samples were consistent with the reference values reported by the IAEA. The accuracies for 234U, 238U determinations were high as all analyzed values were within certified reference confidence intervals with precisions estimated to be less than 5 %. Bioconcentration factor (BCF) and translocation factors (TF) were calculated as [7]:
Results and discussion
238U, 234U and total uranium concentration in Urtica dioica roots, shoots and soils
The concentrations of 234U and 238U radioisotopes in analyzed plants and soils samples are given in Table 1. 238U concentrations ranged from 0.05 ± 0.03 to 2.46 ± 0.11 mBq g−1 dry wt in green parts of analyzed U. dioica, from 0.15 ± 0.06 to 2.50 ± 0.03 mBq g−1 dry wt in roots and from 6.0 ± 0.6 to 29.3 ± 1.5 mBq g−1 dry wt in corresponding soils samples. For analyzed control sample the obtained results for 238U are 1.00 ± 0.04 mBq g−1 dry wt in green part, 1.03 ± 0.15 mBq g−1 dry wt in root and 5.9 ± 0.4 mBq g−1 dry wt for soil. Total uranium values calculated for green parts, roots, whole plants of analyzed U. dioica plants and corresponding soils are presented in Table 2.
Comparison with other studies on uranium uptake by plants
Wild and cultivated plants from the immediate vicinity of uranium waste dumps in Ronneburg in Germany stored normal to eightfold uranium contents. Leafy plant species accumulated much uranium, whereas tubes, thick parts of stalks, fruits and grains stored less uranium. With increasing age of the vegetation uranium content decreased significantly [26]. The obtained results for U. dioica samples were similar or insignificantly higher in comparison to other results. Uranium in lettuce plants growing in contaminated soils (near uranium mining facilities in Portugal) ranged from 0.95 to 6 mg kg−1 dry wt in roots and from 0.32 to 2.6 mg kg−1 dry wt in leaves [27]. The concentration of uranium in shoots of plant species grown in soil contaminated with 100 mg kg−1 of uranium ranged between 3.2 and 24 mg kg−1 dry wt Sunflower and Indian mustard had the highest uranium concentrations in shoots (24.6 and 21.8 mg kg−1 dry wt, respectfully), while wheat and ryegrass had the lowest concentrations (3.2 and 3.8 mg kg−1 dry wt). On the other hand, uranium concentrations in roots of the analyzed plants were significantly higher than in shoots and varied from 89 to 810 mg kg−1 dry wt what were 30–50 times greater than its concentration in shoots [28]. Al-Kharouf et al. measured concentrations of 234U and 238U in watermelon and zucchini crops harvested on irrigated cultivated area which lies above superficial uranium deposits. The average 234U and 238U concentrations were found to be 0.017 and 0.010 mBq g−1 dry wt, respectively. 234U and 238U concentrations in watermelon green parts with roots had average of 0.81 and 0.65 mBq g−1 dry wt respectively, what was an order of magnitude higher than in pulp. Zucchini fruits had concentrations below the detection limit of 1 × 10−4 mBq g−1 dry wt for 234U and 238U. The average 234U and 238U concentrations in zucchini green parts with roots were 0.75 ± 0.04 and 0.72 ± 0.03 mBq g−1 dry wt, respectively [29]. The highest uranium concentrations were measured in soils, different crops and vegetables from the uranium mining area in Jiangxi province in southeastern China: 3159 ± 415 mBq g−1 dry wt for soil samples, while the mean specific activities of 238U in analyzed plants ranged from 15 to 118 and from 108 to 1167 mBq g−1 dry wt for the shoots and roots, respectively [30]. In 2011 series of different plants (meadow, hygrophilous, edible, ruderal plants and corn) were collected around phosphogypsum stockpile in Wiślinka and surveyed on uranium contents. Total uranium concentrations depended on plant type. The uranium content was associated with the plants age, root system (e.g. storage root system, taproot system, superior root system, and fibrous root system) and plant tomentose [7]. Other authors also show that uranium is accumulated in plants depending on their species and cultivars [31, 32]. In case of U. dioica it is clearly seen that localization has a crucial impact on uranium accumulation. Most of the authors report that higher uranium concentrations are present in root system of plants [28, 31, 33, 34]. This aspect is not so obvious in our research as there are samples with higher uranium concentrations in above-ground parts (Fig. 2).
Impact of the phosphogypsum stack
Possible impact of uranium from phosphogypsum stockpile on green parts, roots and whole plants of U. dioica in respect with distance from the stockpile was evaluated. The results revealed that total uranium concentrations in analyzed roots are weakly but negatively correlated with distance (r s = −0.43) (Fig. 3). Similar effect can be noticed for uranium concentrations in analyzed soils samples (r s = −0.83). This is not a general trend in this area. Some of the previously analyzed soils samples contained higher uranium concentrations than phosphogypsum what can be explained by the use of phosphate fertilizers in this area [35]. Opposite effect can be noticed in green parts. One of our main aims was to evaluate possible green parts contamination with phosphogypsum dust. There is no correlation between total uranium concentration in shoots and distance from phosphypsum stockpile (Fig. 4) and slight correlation between whole U. dioica plants and distance (Fig. 5). Only three samples (5, 6 and 8) contained increased uranium concentrations in shoots (Figs. 2 and 4). These three sites are located in open area that can be affected by air deposition with possible phosphogypsum particles. In 2012 wind directions in the area of Wiślinka were examined. The dominant winds were southern, northern and western [36]. The possibility of air transportation of both phosphogypsum and sewage sludge particles that cover the stockpile cannot be neglected. This fact may be connected with the northern-western wind that was observed in this area.
The values of 234U/238U activity ratios
The values of 234U/238U activity ratio in analyzed environmental samples ranged between 0.84 ± 0.05 and 1.29 ± 0.72 for green parts, 0.83 ± 0.15 and 1.31 ± 0.15 for roots as well as 0.96 ± 0.12 and 1.17 ± 0.08 in corresponding soils (Table 1). The obtained values of the 234U/238U activity ratios for U. dioica samples are typical for plants, where the variations lie between 1.02 and 1.30 [7, 14]. Higher values of this activity ratio might be connected with plants interaction with water. The plants could have higher values of 234U/238U activity ratios than sediments suggesting that main source of uranium is water [37]. The similar effect was observed in this area in previous years [6]. Typical value of 234U/238U activity ratio for soils lies between 0.5 and 1.3 and is dependent on the geological surface [38]. Activity ratios obtained for soils in our research are typical for terrestrial environment. As a confirmation we calculated Spearman correlation factors for 234U and 238U concentrations in green parts and roots of U. dioica as well as soils. We received statistically significant r s values: 1.00 for green parts, 0.98 for roots and 0.98 for soils. Spearman’s rank correlation is a non-parametrical alternative for Pearson’s correlation. It can be used to calculate the correlation between two variables that do not have normal distribution and are not linear. What is more, Spearman’s rank correlation is resistant for outlier results [39].
The values of uranium BCF and TF in Urtica dioica
In order to understand the aspects of uranium phytoaccumulation in analyzed U. dioica samples we calculated TF, TFgreen part/soil and BCF, BCFplant/soil according to Eqs. (1)–(4). The obtained factors are presented in Table 3. Depending on the sample collection site these factors ranged from 0.035 ± 0.004 to 0.249 ± 0.010 for BCF, from 0.002 ± 0.001 to 0.121 ± 0.008 for TFgreen part/soil, from 0.069 ± 0.045 to 0.329 ± 0.068 for BCFplant/soil and from 0.02 ± 0.01 to 3.22 ± 0.08 for TF. In control samples obtained BCF and TF are 0.97 ± 0.06, 0.174 ± 0.005, 0.168 ± 0.007 and 0.342 ± 0.008, respectively. The highest BCF factor observed for sample number 2 was more than seven times higher than the lowest for sample number 5.
Comparison with other uranium TF and BCF studies
The obtained TFgreen part/soil values for uranium are slightly different than reported in other studies. TFgreen part/soil for lettuce lied between 0.011 and 0.023 [27], while for zucchini and watermelon was 4.21 × 10−2 and 1.82 × 10−2 [29]. TFgreen part/soil and BCF values of vegetables grown in soils affected by uranium mining ranged from 0.005 to 0.037 and from 0.042 to 0.39, respectively [30]. Manigandan and Manikandan reported that uranium uptake by plants is low and BCFplant/soil ratios were between 0.303 and 0.354 for different plant species [40]. Al-Masri et al. observed that vegetables characterized with relatively higher TFgreen part/soil than their fruits [41]. Vera Tome et al. reported BCFplant/soil for 238U in range of 0.020–0.250 what is similar to our results [42]. Sheppard et al. studied uptake of natural radionuclides by field and garden crops and reported an overall geometric mean BCFplant/soil of 0.013 for uranium [43] while IAEA reports overall range of 10−2–10−4 [44]. In 2005 Sheppard et al. published results for uranium BCFplant/soil values in plants from the area of uranium refinery and background sites across the Canada. The average BCFplant/soil were 0.0068 and 0.0035, respectively [45]. BCF and TFs values received for control sample from Malbork suggest that these values are rather dependent on soils characteristics and uranium bioavailability. For analyzed meadow, hygrophilous, edible, ruderal and corn plants we observed different BCF and similar TF values (TF were in range of 0.05 for edible plants to 0.86 for hygrophilous plants, while BCF values ranged from 0.54 to 1.63). BCF values differences can be explained by different solum, substratum and bioavailability of uranium [7]. TF values were similar but no factors higher than 1 were noticed. The differences between TF values are probably connected with wet and dry air deposition and different tomentose that is dependent on plant type [7]. The comparison between BCF and TF values for U. dioica and other plants is presented on Table 4.
TF and BCF values variability explanation
Differences on uranium uptake by plants can be explained by coil characteristics and different soils composition [46, 47]. Soil type can influence the sorption and desorption of metals. There are certain differences in bioavailability of radionuclides among soils, which may or may not be based on just quantitative properties of the soils [48]. This fact is explained by Ramaswami et al. who observed that an organic-rich soil sequestered uranium, rendering it largely unavailable for plant uptake [49]. We find weak positive correlation between uranium concentration in soils and roots (Fig. 6) and weak negative correlation between BCFplant/soil (Fig. 7) and uranium concentration in soils. No correlation is observed in BCF and concentration in soils (Fig. 8). For the highest uranium concentrations in soils, although plant uptakes relatively more uranium, the BCF value is lower than for soils with less uranium. This could mean that plants may exhibit different affinities to the different uranium species. Similar effect was observed by Vandenhove et al. [47], where the uranyl cation, uranyl carbonate complexes together with the \( {\text{UO}}_{ 2} {\text{PO}}_{4}^{ - } \) species, were probably the uranyl forms most readily taken up by the roots and transferred to the shoots. According to other research, plants take up radionuclides that have similar chemical behavior as the essential nutrient. Radionuclides are then transported to specific tissues based on the function of the element in plant metabolism. It is reflected in its higher concentration in a particular part compared to others [29, 40]. On the other hand, there are considerable differences in the uptake and translocation of long-lived radionuclides among different plant species [30]. These facts as well as the extent and type of plant root system and the response of plants to elements in relation to seasonal cycles might explain different TF and BCF values in plants species [21, 22]. Very often difference in uranium uptake by plants may be connected with possible air deposition. The average concentration of uranium in air close to the ground is about 0.15 ng m−3 and depends on the amount of suspended particles in the air [50]. Atmospheric deposition is the main source of uranium in the above-ground parts of the plants and the incorporation of the radionuclides occurs mainly from the wet deposition [9]. Lower TFgreen part/soil might implicit that the main route of uranium accumulation in green parts is transportation via roots. Higher values suggest that air deposition is more important way. In general, roots serve as a natural barrier preventing the transport of many trace metals, including radionuclides to upper plant parts. Moreover, the radionuclide translocation from roots to shoots is probably dependent on the species [46]. The effect of soil adhesion to leaves is negligible [51]. In this study uranium concentration in soil do not affect the level of this element accumulation in U. dioica green parts (Fig. 9). There is no direct correlation between uranium concentrations in roots (Fig. 10) and soils (Fig. 11) and in green parts of analyzed plants, what is confirmed by TF values obtained for U. dioica plants. There are samples with TF values higher than one. High correlation between TF factors and TFgreen part/soil confirms possible aerial deposition route for uranium in green parts (Fig. 12).
Conclusions
Uranium concentration in analyzed plants and soils allows us to conclude that U. dioica is not a perfect bioindicator but it can be used as a bioimonitor of uranium contamination. Nevertheless, the level of uranium accumulation by common nettles is not extremely high. We noticed that uranium concentrations in roots depended on uranium concentrations in soils although BCF values are not correlated with its contents in soils. It suggests that different uranium species have different affinities to U. dioica. In case of distance from phosphogypsum stockpile, uranium concentration in roots and soil decreases while in green parts of some samples is high. The decrease of uranium concentration with distance for whole plants is not observed. This difference is probably connected with the fact that uranium can be uptaken by green parts from wet and dry air deposition. We can conclude that the problem of phosphogypsum stockpile is limited to the zone of maximum 300–400 m. Uranium concentrations in U. dioica samples which were collected from the slopes of the stockpile are three times higher than in plant from control area in Malbork. In case of analyzed soils it is up to six times higher. Even though we cannot neglect the fact that air deposition in the area of Wiślinka contains phosphogypsum particles.
References
Azouazi M, Quahidiy Y, Fakhi S, Anders Y, Abbe JCh, Benmansour M (2001) J Environ Radioact 54:231–242
Baxter MS (1996) J Environ Radioact 32:3–17
Hull CD, Burnett WC (1996) J Environ Radioact 32:213–238
Bem H (2005) Radioaktywność w środowisku naturalnym. Polska Akademia Nauk, Oddział w Łodzi, Komisja Ochrony Środowiska, Łódź, Poland. ISBN: 978-8-3864-9235-0
Boryło A, Nowicki W, Skwarzec B (2009) Int J Environ Anal Chem 89:677–685
Skwarzec B, Boryło A, Kosińska A, Radzajewska S (2010) Nukleonika 55:187–195
Boryło A, Skwarzec B, Olszewski G (2012) J Environ Sci Heal Part A 47:675–687
Boryło A, Olszewski G, Skwarzec B (2013) Environ Sci-Proc Imp 15:1622–1628
Boryło A, Skwarzec B (2011) Radiochim Acta 99:1–13
Delacroix D, Guerre JP, Leblanc P, Hickman C (1998) Radionuclide and radiation protection. Data Handbook. British Library, Nuclear Technology Publishing, Oxford. ISBN: 1 870965 87 6
Skwarzec B (1995) Polonium, uranium and plutonium in the southern Baltic ecosystem. Rozprawy i Monografie Instytutu Oceanologii PAN, Sopot
Meinrath A, Schneider P, Meinrath G (2003) J Environ Radioact 64:175–193
Carvalho FP, Oliveira JM (2010) Environ Int 36:352–360
Baturin GN (1975) Uran v sovremennom morskom osadkoo-brazovanii, Atomizdat, Moscow
Skwarzec B (1997) Ambio 26:113–117
Szefer P (1987) Stud Mater Oceanol 51:133–193
Szefer P (1987) Stud Mater Oceanol 52:225–268
Szefer P (1987) Stud Mater Oceanol 51:195–223
Elless MP, Lee SY (1998) Water Air Soil Pollut 107:147–162
Langmuir D (1978) Geochim Cosmochim Acta 42:547–569
Kabata-Pendias A, Pendias H (1984) Trace elements in soils and plants. CRC Press, Boca Raton. ISBN: 0-8493-6639-9
Malik RN, Husain SZ, Nazir I (2010) Pak J Bot 42:291–301
Boryło A (2013) J Radioanal Nucl Chem 295:621–631
Skwarzec B (1997) Chem Anal 42:107–115
Hurtgen C, Jerome S, Woods M (2000) Appl Radiat Isot 53:45–50
Anke M, Seebera O, Muller R, Schafer U, Zerull J (2009) Chem Erde-Geochem 69:75–90
Neves O, Abreu MM, Vicente EM (2008) Water Air Soil Poll 195:73–84
Shahandeh H, Hossner LR (2002) Water Air Soil Pollut 141:165–180
Al-Kharouf SJ, Al-Hamarneh IF, Dababneh M (2008) J Environ Radioact 99:1192–1199
Chen SB, Zhu YG, Hu QH (2005) J Environ Radioact 82:223–236
Ebbs SD, Brady DJ, Kochian LV (1998) J Exp Bot 49:1183–1190
Huang JW, Blaylock MJ, Kapulnik YK, Ensley BD (1998) Environ Sci Technol 32:2004–2008
Overall RA, Parry DL (2004) Environ Pollut 132:307–320
Soudek P, Petrová S, Benesová D, Dvoráková M, Vanek T (2011) J Environ Radioact 102:598–604
Olszewski G, Boryło A, Skwarzec B (2015) J Environ Radioact 146:56–66
Boryło A, Nowicki W, Olszewski G, Skwarzec B (2012) J Environ Sci Heal A 47:1831–1842
Vargas MJ, Tome FV, Sánchez AM, Vázquez MTC, Murillo JLG (1997) Appl Radiat Isot 48:1137–1143
Goldstein SJ, Rodriguez JM, Lujan N (1997) Health Phys 7:10–18
Corder GW, Foreman DI (2014) Nonparametric statistics: a step-by-step approach, 2nd edn. Wiley, New York
Manigandan PK, Manikandan NM (2008) Iran J Radiat Res 6:7–12
Al-Masri MS, Al-Akel BA, Nashawani A, Amin Y, Khalifa KH, Al-Ain F (2008) J Environ Radioact 99:322–331
Tome FV, Rodriguez MPB, Lozano JC (2003) J Environ Radioact 65:161–175
Sheppard SC, Evenden WG, Pollock RJ (1989) Can J Soil Sci 69:751–767
IAEA, International Atomic Energy Agency (1994) Handbook of parameters values for the prediction of radionuclide transfer in temperate environments. International Atomic Energy Agency, Vienna
Sheppard SC, Sheppard MI, Llin M, Thompson P (2005) Radioprot Suppl 40:253–259
Shtangeeva I (2010) J Environ Radioact 101:458–463
Vandenhove H, Van Hees M, Wannijn J, Wouters K, Wang L (2007) Environ Pollut 145:577–586
Sheppard SC, Evenden WG (1992) Arch Environ Con Tox 23:117–124
Ramaswami A, Carr P, Burkhardt M (2001) Int J Phytoremediat 3:189–201
WHO, World Health Organization (1996) Guidelines for drinking water quality. World Health Organization, Geneva
Ham GJ, Ewers LW, Wilkins BT (1998) Variations in concentrations of naturally occurring radionuclides in foodstuffs. NRPBM 892, Chilton
Acknowledgments
The authors would like to thank National Science Centre Poland for the financial support under Grant UMO-2012/05/N/NZ7/00978 and Ministry of Science and Higher Education under Grant DS 530-8630-D505-14. The publication is financed from the European Social Fund as part of the project ‘Educators for the elite—integrated training program for Ph.D. students, post-docs and professors as academic teachers at University of Gdansk’ within the framework of Human Capital Operational Programme, Action 4.1.1, improving the quality of educational offer of tertiary education institutions. This publication reflects the views only of the author, and the funder cannot be held responsible for any use which may be made of the information contained therein. Presented map in this article come from the collection of Documentation Centre of Geodesy and Cartography in Warsaw, Poland. The authors also thank M.Sc. Grzegorz Romańczyk for help in plants sampling.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Olszewski, G., Boryło, A. & Skwarzec, B. A study on possible use of Urtica dioica (common nettle) plants as uranium (234U, 238U) contamination bioindicator near phosphogypsum stockpile. J Radioanal Nucl Chem 308, 37–46 (2016). https://doi.org/10.1007/s10967-015-4302-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-015-4302-3