Abstract
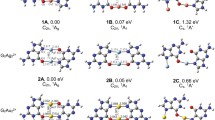
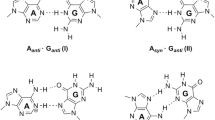
Dinuclear metal-mediated hetero base pairs with the d10–d10 closed-shell interactions have significant stability. It is interesting to identify whether coinage metal-mediated Wobble base pairs are also stable. Geometric and electronic structures of the lowest-lying isomers of GUM22+ (G = guanine, U = uracil, M = Cu, Ag, and Au) cluster ions were investigated with density functional theory. In the lowest-lying isomers of these dinuclear metal-mediated base pairs, the 2-oxo-4-hydroxy-trans-N1H isomer of uracil is derived from the canonical tautomer of uracil by the hydrogen atom transfer. M22+ cations remain as an unbroken unit and interact with the G···U ligand through two sets of closely linear N···M···O units, while the reciprocal hydrogen bonds between the Wobble base pair (G–U) are entirely substituted by the M–N or M–O interactions in these complexes. The atoms in molecules and EDA–NOCV calculations really reveal that the σ interactions in GUM22+ cations are the paramount term of ΔEOrb. The obtained instantaneous interaction energies ΔEint and bond dissociation energies of the metal–ligand interactions give the trend of the bond strength as Cu > Au > Ag.







Similar content being viewed by others
References
G. H. Clever, C. Kaul, and T. Carell (2007). Angew. Chem. Int. Ed. 46, 6226–6236.
K. Tanaka and M. Shionoya (2007). Coord. Chem. Rev. 251, 2732–2742.
W. He, R. M. Franzini, and C. Achim (2008). Inorg. Chem. 55, 545–612.
J. Müller (2008). Eur. J. Inorg. Chem. 2008, 3749–3763.
R. Mas-Ballesté, O. Castillo, P. J. Sanz Miguel, D. Olea, J. Gómez-Herrero, and F. Zamora (2009). Eur. J. Inorg. Chem. 2009, 2885–2896.
J. Liu, Z. Cao, and Y. Lu (2009). Chem. Rev. 109, 1948–1998.
G. H. Clever and M. Shionoya (2010). Coord. Chem. Rev. 254, 2391–2402.
A. Ono, H. Torigoe, Y. Tanaka, and I. Okamoto (2011). Chem. Soc. Rev. 40, 5855–5866.
Y. Takezawa and M. Shionoya (2012). Acc. Chem. Res. 45, 2066–2076.
S. Mandal, M. Hebenbrock, and J. Müller (2016). Angew. Chem. Int. Ed. 55, 15520–15523.
S. Mandal and J. Müller (2017). Curr. Opin. Chem. Biol. 37, 71–79.
S. Taherpour, O. Golubev, and T. Lönnberg (2016). Inorg. Chim. Acta. 452, 43–49.
A. Ono and H. Togashi (2004). Angew. Chem. Int. Ed. 43, 4300–4302.
T. Ehrenschwender, W. Schmucker, C. Wellner, T. Augenstein, P. Carl, J. Harmer, F. Breher, and H. A. Wagenknecht (2013). Chem. Eur. J. 19, 12547–12552.
E. Toomey, J. Xu, S. Vecchioni, L. Rothschild, S. Wind, and G. E. Fernandes (2016). J. Phys. Chem. C 120, 7804–7809.
K. S. Park, C. Jung, and H. G. Park (2010). Angew. Chem. Int. Ed. 49, 9757–9760.
T. Carell (2011). Nature 469, 45–46.
J. Liu and Y. Lu (2007). Angew. Chem. 119, 7731–7734.
X. Liu, C.-H. Lu, and I. Willner (2014). Acc. Chem. Res. 47, 1673–1680.
A. Rioz-Martínez and G. Roelfes (2015). Curr. Opin. Chem. Biol. 25, 80–87.
D. A. Megger, C. F. Guerra, J. Hoffmann, B. Brutschy, F. M. Bickelhaupt, and J. Müller (2011). Chem. Eur. J. 17, 6533–6544.
M. Su, M. Tomás-Gamasa, and T. Carell (2015). Chem. Sci. 6, 632–638.
H. Mei, S. A. Ingale, and F. Seela (2014). Chem. Eur. J. 20, 16248–16257.
H. Mei, H. Yang, I. Röhl, and F. Seela (2014). ChemPlusChem 79, 914–918.
H. Yang, H. Mei, and F. Seela (2015). Chem. Eur. J. 21, 10207–10219.
I. Sinha, C. F. Guerra, and J. Müller (2015). Angew. Chem. Int. Ed. 54, 3603–3606.
S. Mandal, A. Hepp, and J. Müller (2015). Dalton Trans. 44, 3540–3543.
S. Mandal, M. Hebenbrock, and J. Müller (2018). Inorg. Chim. Acta. 472, 229–233.
J. Kondo, T. Sugawara, H. Saneyoshi, and A. Ono (2017). Chem. Commun. 53, 11747–11750.
S. Mandal, M. Hebenbrock, and J. Müller (2017). Chem. Eur. J. 23, 5962–5965.
J. M. Méndez-Arriaga, C. R. Maldonado, J. A. Dobado, and M. A. Galindo (2018). Chem. Eur. J. 24, 1–8.
J. V. Burda, J. Špřoner, J. Leszczynski, and P. Hobza (1997). J. Phys. Chem. B 101, 9670–9677.
E. S. Kryachko and F. Remacle (2005). J. Phys. Chem. B 109, 22746–22757.
A. Kumar, P. C. Mishra, and S. Suhai (2006). J. Phys. Chem. A 110, 7719–7727.
P. J. Mohan, A. Datta, S. S. Mallajosyula, and S. K. Pati (2006). J. Phys. Chem. B 110, 18661–18664.
N. Vyas and A. K. Ojha (2012). Comput. Theor. Chem. 984, 93–101.
E. S. Kryachko and F. Remacle (2005). Nano Lett. 5, 735–739.
G.-J. Cao, H.-G. Xu, R.-Z. Li, and W. Zheng (2012). J. Chem. Phys. 136, 014305.
G.-J. Cao, H.-G. Xu, W.-J. Zheng, and J. Li (2014). Phys. Chem. Chem. Phys. 16, 2835–2928.
P. Wang, H.-G. Xu, G.-J. Cao, W.-J. Zhang, X.-L. Xu, and W.-J. Zheng (2017). J. Phys. Chem. A 121, 8973–8981.
J. Valdespino-Saenz and A. Martínez (2008). J. Phys. Chem. A 112, 2408–2414.
G.-J. Cao, H.-G. Xu, X.-L. Xu, P. Wang, and W.-J. Zheng (2016). Int. J. Mass Spectrom. 407, 118–125.
M. V. Vázquez and A. Martínez (2008). J. Phys. Chem. A 112, 1033–1039.
N. Russo, M. Toscano, and A. Grand (2003). J. Mass Spectrom. 38, 265–270.
L. A. Espinosa Leal and O. Lopez-Acevedo (2015). Nanotechnol. Rev. 4, 173–191.
S. J. Stohs and D. Bagchi (1995). Free Radic. Biol. Med. 18, 321–336.
K. Yamamoto and S. Kawanishi (1989). J. Biol. Chem. 264, 15435–15440.
J. L. Sagripanti and K. H. Kraeme (1989). J. Biol. Chem. 264, 1729–1734.
V. I. Danilov, V. M. Anisimov, N. Kurita, and D. Hovorun (2005). Chem. Phys. Lett. 412, 285–293.
J. Šponer, M. Sabat, J. V. Burda, J. Leszczynski, and P. Hobza (1999). J. Phys. Chem. B 103, 2528–2534.
J. M. Weber, J. A. Kelley, W. H. Robertson, and M. A. Johnson (2001). J. Chem. Phys. 114, 2698–2706.
M. D. Topal and J. R. Fresco (1976). Nature 263, 285–289.
J. Florián and J. Leszczyński (1996). J. Am. Chem. Soc. 118, 3010–3017.
G.-J. Cao (2017). Sci. Rep. 7, 14896.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, Ö. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox Gaussian 09, Revision C. 01 (Gaussian, Inc., Wallingford, 2010).
A. D. Becke (1993). J. Chem. Phys. 98, 5648–5652.
C. Lee, W. Yang, and R. G. Parr (1988). Phys. Rev. B 37, 785–789.
D. Figgen, G. Rauhut, M. Dolg, and H. Stoll (2005). Chem. Phys. 311, 227–244.
K. A. Peterson and C. Puzzarini (2005). Theor. Chem. Acc. 114, 283–296.
J. P. Foster and F. Weinhold (1980). J. Am. Chem. Soc. 102, 7211–7218.
R. F. W. Bader (1991). Chem. Rev. 91, 893–928.
R. F. W. Bader (1998). J. Phys. Chem. A 102, 7314–7323.
R. F. W. Bader, et al. Atoms in Molecules (Wiley, New York, 1990).
T. Lu and F. Chen (2012). J. Comput. Chem. 33, 580–592.
A. D. Becke (1988). Phys. Rev. A 38, 3098–3100.
J. P. Perdew and W. Yue (1986). Phys. Rev. B 33, 8800–8802.
C. F. Guerra, J. G. Snijders, G. T. Velde, and E. J. Baerends (1998). Theor. Chem. Acc. 99, 391–403.
G. T. Velde, F. M. Bickelhaupt, E. J. Baerends, C. F. Guerra, S. J. A. V. Gisbergen, J. G. Snijders, and T. Ziegler (2001). J. Comput. Chem. 22, 931–967.
See http://www.scm.com for ADF2013.01, SCM, Theoretical Chemistry, Vrije Universiteit, Amsterdam, The Netherlands. Accessed 01 July 2017.
E. V. Lenthe, A. Ehlers, and E. J. Baerends (1999). J. Chem. Phys. 110, 8943–8953.
E. V. Lenthe and E. Jan Baerends (2000). J. Chem. Phys. 112, 8279–8292.
G.-J. Cao (2018). J. Chem. Phys. 149, 144308.
G.-J. Cao and W.-J. Zheng (2013). Acta Phys. Chim Sin. 29, 2135–2147.
P. Pyykkö (2004). Angew. Chem. Int. Ed. 43, 4412–4456.
P. Pyykkö (2005). Inorg. Chim. Acta. 358, 4113–4130.
P. Pyykko (2008). Chem. Soc. Rev. 37, 1967–1997.
P. Pyykkö (1988). Chem. Rev. 88, 563–594.
P. Pyykkö (1979). Acc. Chem. Res. 12, 276–281.
P. Pyykkö (2002). Angew. Chem. Int. Ed. 41, 3573–3578.
I. Mayer (1984). Int. J. Quantum Chem. 26, 151–154.
I. Mayer (1985). Theor. Chim. Acta 67, 315–322.
I. Mayer (1983). Chem. Phys. Lett. 97, 270–274.
A. J. Bridgeman, G. Cavigliasso, L. R. Ireland, and J. Rothery (2001). J. Chem. Soc. Dalton Trans. (14), 2095–2108. https://doi.org/10.1039/B102094N.
P. L. Ayers, R. J. Boyd, P. Bultinck, M. Caffarel, R. Carbó-Dorca, M. Causá, J. Cioslowski, J. Contreras-Garcia, D. L. Cooper, P. Coppens, C. Gatti, S. Grabowsky, P. Lazzeretti, P. Macchi, Á. Martín Pendás, P. L. A. Popelier, K. Ruedenberg, H. Rzepa, A. Savin, A. Sax, W. H. E. Schwarz, S. Shahbazian, B. Silvi, M. Solà, and V. Tsirelson (2015). Comput. Theor. Chem. 1053, 2–16.
G.-J. Cao, W. H. Eugen Schwar, and J. Li (2015). Inorg. Chem. 54, 3695–3701.
P. Jerabek, H. W. Roesky, G. Bertrand, and G. Frenking (2014). J. Am. Chem. Soc. 136, 17123–17135.
Acknowledgements
This work was supported by the Natural Science Foundation of China (Grant No. 21501114), the Natural Science Foundation of Shanxi Province (Grant No. 2015021048), and the Open Fund of Beijing National Laboratory for Molecular Sciences (Grant No. BNLMS20150051).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cao, GJ., Hou, HL. Dinuclear Metal-Mediated Guanine–Uracil Base Pairs: Theoretical Studies of GUM22+ (M = Cu, Ag, and Au) Ions. J Clust Sci 30, 439–448 (2019). https://doi.org/10.1007/s10876-019-01503-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-019-01503-0