Abstract
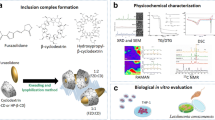
Hydroxymethylnitrofurazone (NFOH) is active against Trypanosoma cruzi; however, its low solubility and high toxicity precludes its current use in treatment of parasitosis. Cyclodextrin can be used as a drug carrier system, as it is able to form inclusion (host–guest) complexes with a wide variety of organic (guest) molecules. Several reports have shown the interesting use of modified β-cyclodextrins in pharmaceutical formulation, to improve the bioavailability of drugs and to decrease their toxicity. The aim of this work was to characterize inclusion complexes formed between NFOH and dimethyl-β-cyclodextrin (DM-β-CD) by complexation/release kinetics and solubility isotherm experiments using ultraviolet (UV)-visible spectrophotometry and by the measurement of the dynamics information obtained from T 1 relaxation times and diffusion (DOSY) experiments using nuclear magnetic resonance (NMR) spectroscopy. The complex was prepared at different NFOH and DM-β-CD molar ratios. The UV-visible measurements were recorded in a spectrophotometer, and NMR experiments were recorded at 20°C on a NMR spectrometer (Varian Inova) operating at 500 MHz. Longitudinal relaxation times were obtained by the conventional inversion-recovery method and the DOSY experiments were carried out using the BPPSTE sequence. The kinetics of complexation revealed that 30 h is enough for stabilization of the NFOH absorbance in presence of cyclodextrin. Solubility isotherm studies show a favorable complexation and increase in solubility when NFOH interacts with cyclodextrin. The analysis of the NMR-derived diffusion coefficients and T 1 relaxation times shows that in the presence of DM-β-CD, NFOH decreases its mobility in solution, indicating that this antichagasic compound interacts with the cyclodextrin cavity. The release kinetics assays showed that NFOH changes its release profile when in the presence of cyclodextrin due to complexation. This study was focused on the physicochemical characterization of drug-delivery formulations that may serve as potentially new therapeutic options for the treatment of Chagas’ disease.
Similar content being viewed by others
References
Chung, M.C., Guido, R.V.C., Martinelli, T.F., Gonçalves, M.F., Polli, M.C., Botelho, K.C.A., Varanda, E.A., Colli, W., Miranda, M.T.M., Ferreira, E.I.: Synthesis and in vitro evaluation of potential antichagasic hydroxymethylnitrofurazone (NFOH-121): a new nitrofurazone prodrug. Bioorganic Med. Chem. 11, 4779–4783 (2003)
Rajewski, R.A., Stella, V.J.: Pharmaceutical applications of cyclodextrins. 2. In vivo drug delivery. J. Pharm. Sci. 85, 1142–1169 (1996)
Connors, K.A.: The stability of cyclodextrin complexes in solution. Chem. Rev. 97, 1325–1357 (1997)
Higuchi, T., Connors, K.A.: Phase-solubility techniques. Adv. Anal. Chem. Inst. 4, 117–212 (1965)
Loftsson, T., Hreindóttir, D., Másson, M.: Evaluation of cyclodextrins solubilization of drugs. Int. J. Pharm. 302, 18–28 (2005)
Loftsson, T., Hreindóttir, D., Másson, M.: The complexation efficiency. J. Incl. Phenom. Macrocycl. Chem. 57, 545–552 (2007)
Fraceto, L.F., Spisni, A., Schreier, S., de Paula, E.: Differential effects of uncharged aminoamide local anesthetics on phospholipid bilayers, as monitored by H-NMR measurements. Biophys. Chemist. 115, 11–18 (2005)
Waldeck, A.R., Kuchel, P.W., Lennon, A.J., Chapman, B.E.: NMR diffusion measurements to characterize membrane transport and solute binding. Progr. Nucl. Magn. Reson. Spectrosc. 30, 39–68 (1997)
Fraceto, L.F., Moraes, C.M., Abrami, P., Gonçalves, M.M., Andreo Filho, N., Fernandes, S.A., de Paula, E. Preparação e caracterização Físico-química de complexos de inclusão entre anestésicos locais e hidróxipropil-beta- ciclodextrina. Quim Nova 30, 777–784 (2007)
Jesus, M.B., Pinto, L.M.A., Fraceto, L.F., Takahata, Y., Lino, A.C.S., Jaime, C., de Paula, E. Theoretical and experimental study of a praziquantel and beta-cyclodextrin inclusion complex using molecular mechanic calculations and 1H-Nuclear Magnetic Resonance. J. Pharm. Biomed. Anal 41, 1428–1432 (2006)
Wimmer, R., Aachamann, F.L., Larsen, K.L., Petersen, S.B.: NMR diffusion as a novel tool for measuring the association constant between cyclodextrin and guest molecules. Carbohydrate Res. 337, 841–849 (2002)
Loftsson, T., Brewster, M.E.: Pharmaceutical applications of cyclodextrins. 1. Drug solubilization and stabilization. J. Pharm. Sci. 85, 1017–1025 (1996)
Loukas, Y.L., Vraka, V., Gregoriadis, G.: Novel non-acidic formulations of haloperidol complexed with beta-cyclodextrin derivatives. J. Pharm. Biom. Anal. 16, 263–268 (1998)
Owens, P.K., Fell, A.F., Coleman, M.W., Kinns, M., Berridge, J.C.: Use of 1H-NMR spectroscopy to determine the enantioselective mechanism of neutral and anionic cyclodextrins in capillary electrophoresis. J. Pharm. Biomed. Anal. 15, 1603–1619 (1997)
Johnson Jr., C.S.: Diffusion ordered nuclear magnetic resonance spectroscopy: principles and applications progress in nuclear magnetic resonance. Spectroscopy 34, 203–256 (1999)
Rymdém, R., Carlfors, J., Stilbs, P.J.: Substrate binding to cyclodextrins in aqueous solution: a multicomponent self diffusion study. J. Incl. Phenom. Macro. 1, 159–168 (1983)
Bakkour, Y., Vermeersch, G., Morcellet, M., Boschin, F., Martel, B., Azaroual, N.: Formation of cyclodextrin inclusion complexes with doxycyclin-hyclate: NMR investigation of their characterization and stability. J. Incl. Phenom. Macrocycl. Chem. 54, 109–114 (2006)
Araújo, D.R., Pinto, L.M.A., Braga, A.F.A., de Paula, E.: Local anesthetic formulation for controlled release: therapeutics applications. Rev. Bras. Anestesiol 53, 663–671 (2003)
Araújo, D.R., Cereda, C.M.S., Brunetto, G.B., Pinto, L.M.A., Santana, M.H.A., de Paula, E.: Encapsulation of mepivacaine prolongs the analgesia provided by sciatic nerve blockade in mice. Can. J. Anesth. 51, 566–572 (2004)
Araújo, D.R., Fraceto, L.F., Braga, A.F.A., de Paula, E.: Drug-delivery systems for racemic bupivacaine (S50-R50) and bupivacaine enantiomeric mixture (S75-R25):cyclodextrins complexation effects on sciatic nerve blockade in mice. Rev. Bras. Anestesiol. 55, 316–328 (2005)
Moraes, C.M., Abrami, P., de Paula, E., Braga, A.F., Fraceto, L.F.: Study of the interaction between S(-) bupivacaine and 2-hydroxypropyl-beta-cyclodextrin. Int. J. Pharm. 331, 99–106 (2007)
Moraes, C.M., Abrami, P., Gonçalves, M.M., Andreo-Filho, N., Fernandes, S.A., de Paula, E., Fraceto, L.F.: Preparação e caracterização físico-química de complexos de inclusão entre anestésicos locais e hidroxipropil-β-ciclodextrina. Quím Nova 30, 777–84 (2007)
Moraes, C.M., Abrami, P., de Araújo, D.R., Braga, A.F.A., Issa, M.G., Ferraz, H.G., de Paula, E., Fraceto, L.F.: Characterization of lidocaine: hydroxypropyl-β-cyclodextrin. J. Incl. Phenom. Macrocycl. Chem. 57, 313–316 (2007)
Araujo, D.R., Tsuneda, S.S., Cereda, M.S., Carvallho, F.D.G.E., Preté, P.S.C., Fernandes, S.A., Yokaichiya, F., Franco, M.K.K.D., Mazzaro, I., Fraceto, L.F., Braga, A.F.A., de Paula, E.: Development and pharmacological evaluation of ropivacaine-2-hydroxypropyl-β-cyclodextrin inclusion complex. Eur. J. Pharm. Sci. 33, 60–71 (2008)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grillo, R., Melo, N.F.S., Moraes, C.M. et al. Hydroxymethylnitrofurazone:Dimethyl-β-cyclodextrin Inclusion Complex: A Physical–Chemistry Characterization. J Biol Phys 33, 445–453 (2007). https://doi.org/10.1007/s10867-008-9054-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10867-008-9054-7