Summary
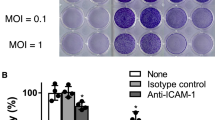
Virotherapy is an emerging strategy for the treatment of cancer that utilizes both replication-competent and genetically modified viruses to selectively kill tumor cells. We have previously shown that Coxsackievirus A21 (CVA21), a common-cold producing enterovirus, is an effective oncolytic agent against human melanoma, prostate, and breast cancer xenografts in vivo. CVA21 specifically targets and lytically infects susceptible cells expressing the CVA21 cellular receptors, intercellular adhesion molecule-1 (ICAM-1) and decay-accelerating factor (DAF). Herein, the efficacy of CVA21 administered in combination with doxorubicin hydrochloride as a new therapeutic regimen for cancer was investigated. Flow cytometric analysis demonstrated that the human breast, colorectal, and pancreatic cancer cell lines examined expressed moderate levels of surface ICAM-1 and DAF, whilst a normal breast cell line expressed only minimal levels. When CVA21 was combined with doxorubicin hydrochloride, synergistically enhanced cell death was observed when CVA21 was administered both simultaneously or 24 h prior to doxorubicin hydrochloride exposure. Doxorubicin hydrochloride had no effect on CVA21 replication. Through the use of an orthotopic (MDA-MB-231-luc) xenograft SCID mouse model of human breast cancer we showed that a single intravenous injection of CVA21 in combination with an intraperitoneal injection of doxorubicin hydrochloride resulted in significantly greater tumor reduction compared to either agent alone. Overall, these findings highlight the exciting potential of CVA21, administered in combination with doxorubicin hydrochloride, as a new therapeutic regimen for cancer.








Similar content being viewed by others
Abbreviations
- ANOVA:
-
One-way analysis of variance
- CI:
-
Combination index
- CVA21:
-
Coxsackievirus A21
- DAF:
-
Decay-accelerating factor
- DMSO:
-
Dimethyl sulfoxide
- FBS:
-
Fetal bovine serum
- 5-FU:
-
5-fluorouracil
- HSV-1:
-
Herpes Simplex Virus-1
- ICAM-1:
-
Intercellular adhesion molecule-1
- MOI:
-
Multiplicity of infection
- PBS:
-
Phosphate buffered saline
- SCID:
-
Severe combined immunodeficient
- SEM:
-
Standard error of the mean
- SPF:
-
Specific-pathogen-free
- TCID50 :
-
50% tissue culture infective dose
- UV:
-
Ultraviolet
References
Parkin DM (2001) Global cancer statistics in the year 2000. Lancet Oncol 2(9):533–543
Greenberg PA, Hortobagyi GN, Smith TL et al (1996) Long-term follow-up of patients with complete remission following combination chemotherapy for metastatic breast cancer. J Clin Oncol 14(8):2197–2205
Fazal S, Saif MW (2007) Supportive and palliative care of pancreatic cancer. J Pancreas 8:240–253
Pecora AL, Rizvi N, Cohen GI et al (2002) Phase I trial of intravenous administration of PV701, an oncolytic virus, in patients with advanced solid cancers. J Clin Oncol 20(9):2251–2266
Hirasawa K, Nishikawa SG, Norman KL et al (2002) Oncolytic reovirus against ovarian and colon cancer. Cancer Res 62(6):1696–1701
Stojdl DF, Lichty B, Knowles S et al (2000) Exploiting tumor-specific defects in the interferon pathway with a previously unknown oncolytic virus. Nat Med 6(7):821–825
Buckland FE, Bynoe ML, Tyrrell DA (1965) Experiments on the spread of colds. II. Studies in volunteers with Coxsackievirus A21. J Hyg (Lond) 63:327–343
Couch RB, Cate TR, Gerone PJ et al (1965) Production of illness with a small-particle aerosol of Coxsackie A21. J Clin Invest 44:535–542
Spickard A, Evans H, Knight V et al (1963) Acute respiratory disease in normal volunteers associated with Coxsackie A-21 viral infection. III. Response to nasopharyngeal and enteric inoculation. J Clin Invest 42:840–852
Shafren DR, Au GG, Nguyen T et al (2004) Systemic therapy of malignant human melanoma tumors by a common cold-producing enterovirus, Coxsackievirus A21. Clin Cancer Res 10(1 Pt 1):53–60
Au GG, Lindberg AM, Barry RD et al (2005) Oncolysis of vascular malignant human melanoma tumors by Coxsackievirus A21. Int J Oncol 26(6):1471–1476
Berry LJ, Au GG, Barry RD et al (2008) Potent oncolytic activity of human enteroviruses against human prostate cancer. Prostate 68(6):577–587
Skelding KA, Barry RD, Shafren DR (2009) Systemic targeting of metastatic human breast tumor xenografts by Coxsackievirus A21. Breast Cancer Res Treat 113:21–30
Au GG, Lincz LF, Enno A et al (2007) Oncolytic Coxsackievirus A21 as a novel therapy for multiple myeloma. Br J Haematol 137:133–141
Garber K (2006) China approves world’s first oncolytic virus therapy for cancer treatment. J Natl Cancer Inst 98:298–300
Sung C-K, Choi B, Wanna G et al (2008) Combined VSV oncolytic virus and chemotherapy for squamous cell carcinoma. Laryngoscope 118:237–242
Watanabe I, Kasuya H, Nomura N et al (2008) Effects of tumor selective replication-competent herpes viruses in combination with gemcitabine on pancreatic cancer. Cancer Chemother Pharmacol 61:875–882
Heise C, Lemmon M, Kirn D (2000) Efficacy with a replication-selective adenovirus plus cisplatin-based chemotherapy: dependence on sequencing but not p53 functional status or route of administration. Clin Cancer Res 6:4908–4914
Li Y, Yu DC, Chen Y et al (2001) A hepatocellular carcinoma-specific adenovirus variants, CV890, eliminates distant human liver tumors in combination with doxorubicin. Cancer Res 61:6428–6436
Dias JD, Guse K, Nokisalmi P et al (2010) Multimodal approach using oncolytic adenovirus, cetuximab, chemotherapy and radiotherapy in HNSCC low passage tumour cell cultures. Eur J Cancer 46:625–635
Chahlavi A, Todo T, Martuza RL et al (1999) Replication-competent herpes simplex virus vector G207 and cisplatin combination therapy for head and neck squamous cell carcinoma. Neoplasia 1:162–169
Hoffmann D, Bayer W, Wildner O (2007) Local and distant immune-mediated control of colon cancer growth with fusogenic membrane glycoproteins in combination with viral oncolysis. Hum Gene Ther 18:435–450
Angelova AL, Aprahamian M, Grekova SP et al (2009) Improvement of gemcitabine-based therapy of pancreatic carcinoma by means of oncolytic parvovirus H-IPV. Clin Cancer Res 15(2):511–519
Sei S, Mussio JK, Yang Q et al (2009) Synergistic antitumor activity of oncolytic reovirus and chemotherapeutic agents in non-small cell lung cancer cells. Mol Cancer 8:47
Shafren DR, Bates RC, Agrez MV et al (1995) Coxsackieviruses B1, B3, and B5 use decay accelerating factor as a receptor for cell attachment. J Virol 69(6):3873–3877
Coyne KE, Hall SE, Thompson S et al (1992) Mapping of epitopes, glycosylation sites, and complement regulatory domains in human decay accelerating factor. J Immunol 149(9):2906–2913
Boyd AW, Wawryk SO, Burns GF et al (1988) Intercellular adhesion molecule 1 (ICAM-1) has a central role in cell-cell contact-mediated immune mechanisms. Proc Natl Acad Sci USA 85(9):3095–3099
Andersson P, Alm S, Edman K et al (2005) A novel and rapid method to quantify cytolytic replication of picornaviruses in cell culture. J Virol Methods 130:117–123
Ling YH, El-Naggar AK, Priebe W et al (1996) Cell cycle-dependent cytotoxicity, G2/M phase arrest, and disruption of p34cdc2/cyclin B1 activity induced by doxorubicin in synchronized P388 cells. Mol Pharmacol 49:832–841
Piso P, Dahlke MH, Ghali N et al (2007) Multimodality treatment of peritoneal carcinomatosis from colorectal cancer: first results of a new German centre for peritoneal surface malignancies. Int J Colorectal Dis 22:1295–1300
Kouvaraki MA, Ajani JA, Hoff P et al (2004) Fluorouracil, doxorubicin, and streptozocin in the treatment of patients with locally advanced and metastatic pancreatic endocrine carcinomas. J Clin Oncol 22:4762–4771
Gurnani M, Lipari P, Dell J et al (1999) Adenovirus-mediated p53 therapy has greater efficacy when combined with chemotherapy against human head and neck, ovarian, prostate, and breast cancer. Cancer Chemother Pharmacol 44:143–151
Kambara H, Saeki Y, Chiocca EA (2005) Cyclophosphamide allows for in vivo dose reduction of a potent oncolytic virus. Cancer Res 65:11255–11258
Ichihara S, Yamada Y, Kawai Y et al (2007) Roles of oxidative stress and Akt signaling in doxorubicin cardiotoxicity. Biochem Biophys Res Commun 359:27–33
Palmieri FM, Perez EA (2003) Recent advances in adjuvant therapy for breast cancer. Semin Oncol Nurs 19:10–16
Aapro MS (2001) Adjuvant therapy of primary breast cancer: a review of key findings from the 7th international conference, St Gallen, February 2001. Oncologist 6:376–385
Acknowledgements
We gratefully acknowledge those investigators mentioned in the text for the provision of monoclonal antibodies, cell lines and virus that enabled this study to be undertaken. We would like to thank Dr Gough Au, Dr Susanne Johansson, Dr Linda Berry, Dr Erin Haley, and Ms Catherine Delahunty for scientific advice and technical assistance throughout this project. Funding for this work was derived from grants received from the Biotechnology Innovation Fund and Hunter Medical Research Institute. Kathryn Skelding is a National Breast Cancer Foundation Scholar.
Declaration of competing interests
Darren Shafren and Richard Barry hold shares in Viralytics Ltd and are currently conducting research sponsored by this company.
Authors’ contributions
KS participated in the design of the study, carried out the in vitro and in vivo work, and drafted the manuscript. RB participated in the design of the study. DS participated in the design of the study, and was involved in the revision of the manuscript. All authors approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
Combination index for MDA-MB-231 cells simultaneously treated with a combination of CVA21 and paclitaxel, tamoxifen, or 5-FU. Combination indices (CI) were calculated using CalcuSyn, as described in the Materials and methods. CI values less than 0.9 were determined to be synergistic, between 0.9 and 1.1 to be additive, and greater than 1.1 to be antagonistic. (GIF 56 kb)
Supplementary Figure 1
Effect of doxorubicin hydrochloride, paclitaxel, 5-FU, and tamoxifen on CVA21 replication. MDA-MB-231 cell monolayers were inoculated with CVA21 (MOI = 10 TCID50/cell). Following the removal of unbound virus, the cell monolayers were incubated with (A) 2.5 or 0.32 μM doxorubicin hydrochloride, (B) 8 or 2 nM paclitaxel, (C) 10 nM or 10 μM 5-FU, or (D) 10 μM or 313 nM tamoxifen for 48 h. At selected time points (0, 2, 4, 6, 8, 10, 24, and 48 h), cell suspensions were freeze-thawed, clarified, and virus yield was determined by viral lytic assay on SK-Mel-28 cells. (GIF 40 kb)
Rights and permissions
About this article
Cite this article
Skelding, K.A., Barry, R.D. & Shafren, D.R. Enhanced oncolysis mediated by Coxsackievirus A21 in combination with doxorubicin hydrochloride. Invest New Drugs 30, 568–581 (2012). https://doi.org/10.1007/s10637-010-9614-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-010-9614-0