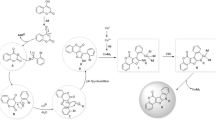
The Vilsmeier formylation of bifunctional Fischer base analogs in the dipyrrolobenzoquinoxaline and indoloindole series was studied with various substrate–Vilsmeier complex ratios. The isomeric Fischer bases of the indoloindole series were formylated smoothly with the formation of the corresponding diformyl derivatives: 2,7-di(formylmethylidene)-1,3,3,6,8,8-hexamethylindolino[7,6-g]indoline and 2,9-di(formylmethylidene)-1,1,3,8,10,10-hexamethylindolino[4,5-e]indoline. In the case of 1,4,5,8-tetra-hydro-1,1,8,8-tetramethyl-2,7-dimethylidenebenzo[g]dipyrrolo[1,2,3-de:3,2,1-ij]quinoxaline reaction at 35°C with a large excess of the Vilsmeier complex (1:45), the diformyl derivative 2,7-di-(formylmethylidene)-1,4,5,8-tetrahydro-1,1,8,8-tetramethylbenzo[g]dipyrrolo[1,2,3-de:3,2,1-ij]quin-oxaline was obtained with a 67% yield. At 60°C and reagent molar ratio of 1:5, a cyanine dye of unknown structure was evidently formed.
Similar content being viewed by others
References
Sh. A. Samsoniya, M. V. Trapaidze, N. N. Nikoleishvili, K. G. Japaridze, J. P. Maisuradze, and U. Kazmaier, Khim. Geterotsikl. Soedin., 1255 (2010). [Chem. Heterocycl. Compd., 46, 1016 (2010).]
Sh. A. Samsoniya, M. V. Trapaidze, N. N. Nikoleishvili, K. G. Japaridze, J. P. Maisuradze, and U. Kazmaier, Khim. Geterotsikl. Soedin., 1259 (2010). [Chem. Heterocycl. Compd., 46, 1020 (2010).]
Yu. M. Chunaev and N. M. Przhiyalgovskaya, in: Advances of Science and Technology, Organic Chemistry Series [in Russian], Vol. 14, VINITI, Moscow (1990), 127 pp.
Sh. A. Samsoniya, M. V. Trapaidze, N. N. Nikoleishvili, K. G. Japaridze, J. P. Maisuradze, U. Kazmaier, Khim. Geterotsikl. Soedin., 1336 (2011). [Chem. Heterocycl. Compd., 47, 1098 (2011).]
M. N. Khan, J.-P. Fleury, P. Baumlin, and C. Hubschwerlen, Tetrahedron, 41, 5341 (1985).
H. Fritz, Chem. Ber., 92, 1809 (1959).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Samsoniya, S.A., Trapaidze, M.V. & Nikoleishvili, N.N. Bisindoles. 43*. Dipyrroloquinoxalines. 2*. Synthesis of the bis-analogs of the fischer aldehyde under vilsmeier conditions. Chem Heterocycl Comp 49, 540–544 (2013). https://doi.org/10.1007/s10593-013-1279-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-013-1279-0