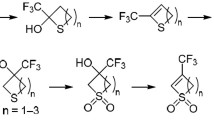
The treatment of 5-aryl-4′,6′-dioxo-2,3,3a,4,5,6-hexahydrospiro-[benzothiophene-4,5′-1,3-dioxane] 1,1-dioxides with boron trifluoride etherate in dichloroethane leads to the formation of products of intramolecular acylation, namely, the corresponding 10-oxo-1H-5,5a,10a,10b-tetrahydrofluoreno-[2,1-b]thiophene dioxides. In all cases, 5-aryl-4-carboxyhexahydrobenzo[b]thiophene 1,1-dioxides were also isolated. The product ratio depends on the structure of the aromatic substituent at C(5) of the spiro- adducts. The structure of 4-carboxy-5-(2-methoxyphenyl)-7-methyl -2,3,3a,4,5,6-hexahydro-benzo[b]thiophene 1,1-dioxide was confirmed by X-ray diffraction structural analysis.
Similar content being viewed by others
References
E. E. Shults, G. N. Andreev, M. M. Shakirov, N. I. Komarova, I. Yu. Bagryanskaya, and Yu. V. Gatilov, Zh. Org. Khim., 44, 1165 (2008).
J. R. Hanson, Nat. Prod. Rep., 21, 312 (2004).
C.-I. Chang, J.-Y. Chang, C.-C. Kuo, W.-Y. Pan, and Y.-H. Kuo, Planta Med., 71, 72 (2005).
T. Minami, M. Iwamoto, H. Ohtsu, H. Ohishi, R. Tanaka, and A. Yoshitake, Planta Med., 68, 742 (2002).
M. Iwamoto, H. Ohtsu, H. Tokuda, H. Nishino, S. Matsunaga, and R. Tanaka, Bioorg. Med. Chem., 9, 1911 (2001).
E. Fillion and D. Fishlock, J. Am. Chem. Soc., 127, 13144 (2005).
L. Planas, M. Mogi, H. Takita, T. Kajimoto, and M. Node, J. Org. Chem., 71, 2896 (2006).
M. Banerjee, R. Mukhopadhyay, B. Achari, and A. K. Banerjee, J. Org. Chem., 71, 2787 (2006).
G. Liang, Yue Xu, I. B. Seiple, and D. Trauner, J. Am. Chem. Soc., 128, 11022 (2006).
G. N. Andreev, E. E. Shults, A. A. Volkov, M. M. Shakirov, I. Yu. Bagryanskaya, Yu. V. Gatilov, and G. A. Tolstikov, Zh. Org. Khim., 40, 892 (2004).
E. Fillion and D. Fishlock, Org. Lett., 5, 4653 (2003).
E. Fillion, A. M. Dumas, and S. A. Hogg, J. Org. Chem., 71, 9899 (2006).
A. L. Spek, Platon, A Multipurpose Crystallographic Tool (Version 10M), Utrecht Univesity, Utrecht, The Netherlands (2003); A. L. Spek, J. Appl. Crystallogr., 36, 7 (2003).
F. H. Allen, O. Kennard, D. G. Watson, L. Bramer, A. G. Orpen, and R. Taylor, J. Chem. Soc., Perkin Trans. 2, S1 (1987).
V. M. Lynch, D. Daniel, S. F. Martin, and B. E. Davis, Acta Crystallogr., C47, 1340 (1991).
G. R. Desiraju, Chem. Commun., 1475 (1997).
C. S. Argyle, K. G. Mason, M. A. Smith, and E. S. Stern, J. Chem. Soc. (C), 2176 (1967).
G. M. Sheldrick, SHELX-97 release 97-2, University of Göttingen, Germany (1998).
Author information
Authors and Affiliations
Corresponding author
Additional information
*2Dedicated to Academician of the Russian Academy of Sciences B. A. Trofimov on his 70th jubilee.
*For Communication 9 see [1].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1503–1512, October, 2008.
Rights and permissions
About this article
Cite this article
Shults, E.E., Andreev, G.N., Shakirov, M.M. et al. Diels-alder reaction with cyclic sulfones. 9.* Synthesis of 10-oxo-1h-tetrahydrofluo-reno[2,1-b]thiophene dioxides*2 . Chem Heterocycl Comp 44, 1220–1228 (2008). https://doi.org/10.1007/s10593-009-0174-1
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-009-0174-1