Abstract
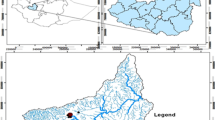
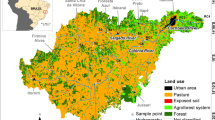
The Cerrado is the second largest Brazilian biome and contains the headwaters of three major hydrological basins in Brazil. In spite of the biological and ecological relevance of this biome, there is little information about how land use changes affect the chemistry of low-order streams in the Cerrado. To evaluate these effects streams that drain areas under natural, rural, and urban land cover were sampled near Brasília, Brazil. Water samples were collected between September 2004 and December 2006. Chemical concentrations generally followed the pattern of Urban > Rural > Natural. Median conductivity of stream water of 21.6 (interquartile: 22.7) μS/cm in urban streams was three and five-fold greater relative to rural and natural areas, respectively. In the wet season, despite of increasing discharge, concentration of many solutes were higher, particularly in rural and natural streams. Streams also presented higher total dissolved N (TDN) loads from natural to rural and urban although DIN:DON ratios did not differ significantly. In natural and urban streams TDN was 80 and 77% dissolved organic N, respectively. These results indicate that alterations in land cover from natural to rural and urban are changing stream water chemistry in the Cerrado with increasing solute concentrations, in addition to increased TDN output in areas under urban cover, with potential effects on ecosystem function.









Similar content being viewed by others
References
Biggs TW et al (2002) Relative influence of natural watershed properties and human disturbance on stream solute concentrations in the southwestern Brazilian Amazon basin. Water Resour Res 38. doi:10.1029/2001WR000271
Alexander RB, Smith RA, Schwarz GE (2000) Effect of stream channel size on the delivery of nitrogen to the Gulf of Mexico. Nature 403:758–761
Ballester MRV et al (2003) A remote sensing/GIS-based physical template to understand the biogeochemistry of the Ji-Paraná river basin (Western Amazonia). Remote Sens Environ 87:429–445
Biggs TW et al (2004) Natural controls and human impacts on stream nutrient concentrations in a deforested region of the Brazilian Amazon basin. Biogeochemistry 68:227–257
Biggs TW, Dunne T, Muraoka T (2006) Transport of water, solutes, and nutrients from a pasture hillslope, southwestern Brazilian Amazon. Hydrol Process 20:2527–2547
Bustamante MMC, Medina E, Asner GP, Nardoto GB, Garcia-Montiel DC (2004) Nitrogen cycling in tropical and temperate savannas. Biogeochemistry 79:209–237
Campbell JL et al (2004) Input-Output budget of inorganic for 24 forest watershed in the northeastern United States: a review. Water Air Soil Pollut 151:373–396
Clesceri LS, Greenberg AE and Eaton AD (1998) Standard methods for the examination of water and wastewater. United Book Press, Baltimore, pp 3-63–3-65; 3-82–3-83; 3-87–3-89
Daniel MHB et al (2002) Effects of urban sewage on dissolved oxygen, dissolved inorganic and organic carbon, and electrical conductivity of small streams along a gradient of urbanization in the Piracicaba river basin. Water Air Soil Pollut 136:189–206
Downing JA et al (1999) The impact of accelerating land-use change on the N-cycle of tropical aquatic ecosystems: current conditions and projected changes. Biogeochemistry 46:109–148
Duff JH, Triska FJ (1990) Denitrification in sediments from the hyporheic zone adjacent to a small forested stream. Can J Fish Aqua Sci 47:1140–1147
Fernandes ACM (2007) Macroinvertebrados bentônicos como indicadores biológicos de qualidade da água: proposta para elaboração de um índice de integridade biológica. PhD thesis, University of Brasilia
Figueiredo RO, Markewitz D, Davidson EA, Schuler AE, Watrin OS, Silva PS (2010) Land-use affects on the chemical attributes of low-order streams in the eastern Amazon. J Geophys Res 115:14
Forti MC, Neal C, Jenkins A (1995) Modeling perspective of the deforestation impact in stream water quality of small preserved forested areas in the amazonian rainforest. Water Air Soil Pollut 79:325–337
Forti MC et al (2000) Hydrogeochemistry of small catchment in northeastern Amazonia: a comparison between natural with deforestation parts of the catchment (Serra do Navio, Amapá State, Brazil). Water Air Soil Pollut 118:263–279
Fox RL, Kamprath EJ (1970) Phosphate sorption isotherms for evaluating the phosphate requirements of soils. Soil Sci Soc Am Proc 34:902–907
Germer S, Neill C, Krusche AV, Elsenbeer H (2010) Influence of land-use change on near-surface hydrological processes: undisturbed forest to pasture. J Hydrol 380(3–4):473–480
Grimm NB, Fisher SG (1984) Exchange between interstitial and surface water: implications for stream metabolism and nutrient cycling. Hydrobiologia 111:219–228
Herpin U et al (2002) Biogeochemical dynamics following land use change from forest to pasture in a humid tropical area (Rondonia, Brazil): a multi-element approach by means of XRF-spectroscopy. Sci Total Environ 286:97–109
Houghton RA (1990) The future role of tropical forests in affecting the carbon dioxide concentration of the atmosphere. Ambio 19:204–209
IBGE (2004) Mapa de biomas do Brasil. Escala 1:5.000.000. Rio de Janeiro. http://mapas.ibge.gov.br/biomas2/viewer.htm. Cited 13 Feb 2007
Jordan TE, Correll DL, Weller DE (1997) Effects of agriculture on discharge of nutrients from coastal plain watershed of Chesapeake Bay. J Environ Qual 26:836–848
Kemp MJ, Dodds WK (2002) The influence of ammonium, nitrate and dissolved oxygen on uptake, nitrification and denitrification rates associated with prairie stream substrata. Limnol Oceanogr 47:1380–1393
Klink CA, Machado RB (2005) Conservation of the Brazilian Cerrado. Conserv Biol 19:707–713
Klink CA, Macedo RH, Mueller CC (1995) De grão em grão, o Cerrado perde espaço. In: Martins ES, Alho CJR (eds) Cerrado: Impactos do processo de ocupação. Documento para Discussão. WWF and PRO-CER, Brasília, p 66
Koroleff F (1983) Total and organic nitrogen. In: Grasshoff K et al (eds) Methods of seawater analysis. Verlag Chemie, Weinheim, pp 162–173
Krusche AV et al (2003) Acid rain and nitrogen deposition in a sub-tropical watershed (Piracicaba): ecosystem consequences. Environ Pollut 121:389–399
Markewitz D et al (2001) Control of cation concentrations in stream water by surface soil process in an Amazonian watershed. Nature 410:802–805
Markewitz D et al (2004) Nutrient loss and redistribution after forest clearing on a highly weathered soil in Amazonia. Ecol Appl 14:177–199
Markewitz D et al (2006) Dissolved rainfall inputs and streamwater outputs in an undisturbed watershed on highly weathered soils in the Brazilian cerrado. Hydrol Process 20:2615–2639
McClain ME, Richey JE, Pimentel TP (1994) Groundwater nitrogen dynamics at the terrestrial-lotic interface of a small catchment in the Central Amazon Basin. Biogeochemistry 27:113–127
Mcgrath DA et al (2001) Effects of land-use change on soil nutrient dynamics in Amazonia. Ecosystems 4:625–645
Melillo JM et al (1996) Tropical deforestation and the global carbon budget. Annu Rev Energy Environ 21:293–310
Miranda LM et al (2000) Produtividade do feijoeiro em resposta à adubação fosfatada e a regimes de irrigação em solo de cerrado. Pesq Agropec Bras 35:703–710
Moraes JM et al (2006) Water storage and runoff processes in plinthic soils under forest and pasture in Eastern Amazonia. Hydrol Process 20:2509–2526
Mulholland PJ et al (2008) Stream denitrification across biomes and its response to anthropogenic nitrate loading. Nature 452:202–206
Neill C et al (1995) Nitrogen dynamics in soil of forests and pasture in the western Brazilian Amazon basin. Soil Biol Biogeochem 27:1167–1175
Neill C et al (2001) Deforestation for pasture alters nitrogen and phosphorus in small Amazonian streams. Ecol Appl 11:1817–1828
Neill C et al (2006) Deforestation alters the hydraulic and biogeochemical characteristics of small lowland Amazonian streams. Hydrol Process 20:2563–2580
Ometto JPHB et al (2000) Effects of land use on water chemistry and macroinvertebrates in two streams of the Piracicaba river basin, south-east Brazil. Freshw Biol 44:327–337
Parron LM (2004) Aspectos da ciclagem de nutrientes em função do gradiente topográfico, em uma mata de galeria no Distrito Federal. PhD thesis, University of Brasilia
PE IN (2009) Deforestation in Brazilian Amazonia. Instituto Nacional de Pesquisas Espaciais, São José dos Campos, Brazil
Pedron FA et al (2004) Solos urbanos. Ciência Rural 34:1647–1653
Reatto A, Martins ES, Farias MFR, Silva AV, Carvalho Jr OA. (2004) Mapa pedológico digital—SIG atualizado do Distrito Federal Escala 1:100.000 e uma síntese do texto explicativo. Ministério da Agricultura Pecuária e Abastecimento. Documentos 120
Richey JE et al (1997) Organic matter and nutrient dynamics in river corridors of the Amazon basin and their response to anthropogenic change. Ciência e Cultura J Braz Assoc Adv Sci 49:98–110
Salati E, Vose PB (1984) Amazon basin: a system in equilibrium. Science 225:129–138
Sano EE et al (2008) Mapeamento semidetalhado do uso da terra do Bioma Cerrado. Pesquisa Agropecuária Brasileira 43:153–156
Silva JSO (2008) Características químicas da água de córregos do Distrito Federal sob diferentes usos e cobertura do solo. PhD thesis, University of Brasília
Sousa DMG, Lobato E, Rein TA (2002) Adubação com fósforo. In: Sousa DMG, Lobato E (eds) Cerrado: correção do solo e adubação. Embrapa Cerrados, Planaltina-DF, p 416
Spera ST, Correia JR, Reatto A (2006) Solos do bioma Cerrado: propriedades químicas e físico-hídricas sob uso e manejo de adubos verdes. In: Carvalho AM, Amabile RF (eds) Cerrado: adubação verde. Embrapa Cerrados, Planaltina-DF, pp 41–65
Thomas MS et al (2004) Influences of land use and stream size on particulate and dissolved materials in a small Amazonian stream network. Biogeochemistry 68:135–151
Triska FJ, Kennedy VC, Avanzino RJ, Zellweger GW, Bencala KE (1989) Retention and transport of nutrients in a third-order stream in northwestern California: hyporheic processes. Ecology 70:1893–1905
Vitousek PM (1980) Nitrogen losses from disturbed ecosystems–ecological considerations. In: Rosswall TH, Soderlund R (eds) Nitrogen cycling in West African ecosystems. SCOPE/UNEP International Nitrogen Unit, Stockholm, pp 39–53
Vitousek PM, Gosz JR, Grier CG, Melillo JM, Reiners WA, Todd RL (1979) Nitrate losses from disturbed ecosystems. Science 204:469–474
Williams MR, Melack JM (1997) Solute export from forest and partially deforest catchments in the central Amazon. Biogeochemistry 38:67–102
Williams MR, Fisher TR, Melack JM (1997) Solute dynamics in soil water and groundwater in a central Amazon catchment under going deforestation. Biogeochemistry 38:303–335
Xuluc-Tolosa FJ, Vester HFM, Ramírez-Marcial N, Castellanos-Albores J, Lawrance D (2003) Leaf litter decomposition of tree species in three successional phases of tropical dry secondary forest in Campeche, Mexico. For Ecol Manag 174:401–412
Acknowledgments
The authors thanks to the administration and staff of the Ecological Reserve of IBGE and Águas Emendadas Ecological Station for the support to this study. We are also very grateful to Mr. Dimas and Sigfrido for allowing us to conduct this research on their properties. We would like also to thank the team of Centro de Energia Nuclear na Agricultura/USP, IESA/UFG and Ester Albuquerque, Viviane Teixeira de Miranda, Gilberto do Banho Cossac and Adriano Porto from the Department of Ecology of the University of Brasília (UnB) for assistance during field and laboratory measurements. Dr Cristina Brandão (FT) and Dr. Eloi Campos (UnB) contributed with valuable advice throughout this study. This research was funded by NASA under Large Scale Biosphere–Atmosphere in Amazonia (LBA) project ND-07 and by EPA. JSO Silva received a scholarship from CNPq (Brazil).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Silva, J.S.O., da Cunha Bustamante, M.M., Markewitz, D. et al. Effects of land cover on chemical characteristics of streams in the Cerrado region of Brazil. Biogeochemistry 105, 75–88 (2011). https://doi.org/10.1007/s10533-010-9557-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-010-9557-8