Abstract
Wetlands are amongst the richest, yet most threatened types of habitats on Earth. One major threat is the modification of water regime for human activities, which disrupts normal ecosystem equilibrium. In lacustrine wetlands, reduced flooding allows shrubs to take over, ultimately leading to a shift towards woody communities. To counter this, wetland managers have initiated a variety of measures, including mowing, burning, and pasturing. Because of the short time frames of previous studies on the subject, little is known on their potential negative side effects on the ecosystem. Here, we evaluate the long-term effect of mowing on breeding populations of the five most abundant species in our central European study area (the reed warbler Acrocephalus scirpaceus, the common reed bunting Emberiza schoeniclus, Savi’s warbler Locustella luscinioides, the water rail Rallus aquaticus, and the bearded reedling Panurus biarmicus). This study, of an unprecedented time scale (30 years), shows that rotational mowing has no long-term detrimental effects on birds. However, optimal mowing regime for the birds might often be less frequent than what is usually applied. We recommend that mowing be spaced every 3 years at least, and ideally every 6 years or more. We discuss additional measures that could be implemented to complement mowing. Because of the widespread distribution of the target habitat and species, our study provides readily applicable information for wetland managers in Europe and worldwide.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Wetlands provide necessary ecosystem services (reviewed in Okruszko et al. 2011) and are home to a wide range of animal and vegetal species worldwide (Dudgeon et al. 2006). Despite their importance, most wetlands are in danger. About 87% of the initial natural wetland surface has already been lost over the last 300 years, and this destruction has occurred at an alarming pace during the last century (Davidson 2014). The causes underlying this destruction are varied, ranging from replacement by crops or buildings to drainage for irrigation and transformation for aquaculture (reviewed in Brinson and Malvárez 2002).
Another major threat is increased control over water level in nearby lakes, preventing natural fluctuations of water table. Wetlands are dynamic habitats where early successional states are normally reset by regular floods, and many wetland species profit from regular flooding of their terrestrial habitats. Thus, reduced flooding magnitude allows shrubs to establish, ultimately leading to a transition towards forest and the exclusion of most threatened wetland species (Quintana-Ascencio et al. 2013).
To protect wetlands from forest progression, managers have set up a variety of strategies, including burning, pasturing, and mowing (Middleton et al. 2006). Because mowing strongly modifies vegetation structure, it can also have a temporary detrimental effect on the resident fauna. Several studies have tried to estimate the effect of mowing on forest progression and biodiversity, with contrasted results (reviewed in Valkama et al. 2008). These discrepancies can be explained both by a strong effect of site-specific environmental factors, and the short time scale of the studies (most of them ranging from a single season to a few years). As noted by Vadász et al. (2008), reedbed structure typically takes several years to reach equilibrium after a cut or other types of disturbance, thus short-term studies likely fail to reflect the long-term effects of management. In addition, between-year variance due to factors other than management can further blur the picture. Furthermore, as noted by Báldi (2005), none of these studies have tried to estimate the optimal mowing regime for biodiversity.
Here, we evaluate the long-term effects of rotational wetland mowing on wetland communities over a period of 30 years, and try to estimate what mowing frequency is optimal. We use birds as indicators of overall habitat suitability. We focus on the five most common wetland bird species in the area, which are widely distributed in Europe. We measure bird abundance in plots with rotating mowing treatments (every 2–6 years) by recording the number of males defending territories. Our study is the first to be conducted over a period of 30 years, which allows us to overcome the major shortcomings of short-term studies and to estimate the optimal mowing frequency for the studied species. Because of the wide distribution of the focal species and habitats, our results will provide readily applicable information for wetland managers in Europe and worldwide.
Materials and methods
Study area
The Grande Cariçaie consists of 2300 ha of natural wetlands divided into seven reserves spread across 40 km along the South-Eastern shore of lake Neuchâtel, Switzerland. It is the largest natural lake shore in Switzerland, consisting mostly of reedbeds (dominated by Phragmites australis) and sedge meadows (dominated by Carex elata and Cladium mariscus). Natural open reedbeds were maintained by frequent floods until the 1970s, when water level was artificially stabilized to limit the risk of inundations to nearby cities and cultures (Fig. S1). The resulting decrease in magnitude of water level fluctuations caused shrubs (Alnus incana, A. glutinosa, Frangula alnus, Salix spp.) to take over wetlands, ultimately progressing towards forest. To protect marshes from forest advance, management was initiated in 1982 by mechanically mowing reedbeds to remove organic matter and restrict the growth of shrubs. Mowing was conducted in a rotational manner following a split plot design.
To assess the effect of mowing on breeding birds, we conducted bird censuses at three different locations: Chevroux, Gletterens and Cheyres. All three locations were characterized by a transition from uplands, moist sedge meadows to lacustrine reedbeds. The locations are presented in Fig. 1. Gletterens and Cheyres were separated into different plots in between 0.5 and 2.5 ha (Figs. S2, S3). Every plot was mown every second to sixth year. Control plots were included in Cheyres since the beginning of the experiment (1982) and in Gletterens since 2005, and they were left untouched. Chevroux is notably different from the other locations in that water level is naturally very high, thus preventing shrubs from invading wetlands there. This situation is extremely rare in the area. This location was therefore never mown. It was excluded from most analyses, but was used as a control to estimate natural population fluctuations without human intervention. Table S1 summarizes the different plots and the mowing regimes.
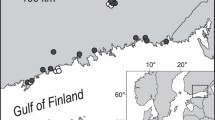
Map of the study area. Green polygons delimit the Grande Cariçaie reserves. The three locations are represented in blue. Top left corner: map of Switzerland, with study area delineated by the black square. Background pictures obtained from the Swiss Federal Office of Topography: www.swisstopo.admin.ch. (Color figure online)
Mowing was performed during the winter by a dedicated caterpillar track machine in order to minimize the impact on soil and fauna. A plot was considered of age 1 the summer following the cut, of age 2 the next year, etc. An open water body was created in 2014 in the control plot in Gletterens, decreasing its surface from 1.1 to 0.7 ha. Because plots of age 5 and 6 look similar in vegetation structure and because of their small number, they were combined for the analyses. A plot was considered as control when untouched for the whole experiment, or 7 years after being last cut and thereafter.
Species of interest
We chose to focus on birds because, being at the top of most trophic chains, they represent good indicators of overall habitat suitability and have been widely used elsewhere (Everard 2008; Butler et al. 2012; but see Sattler et al. 2014). We concentrated on the five most abundant species in the study area, which together utilize the whole range of non-forested, wetland habitats found in the area. Both these habitats and the focus species are widespread throughout most of Europe (e.g. Hungary: Báldi 2005; Netherlands: Graveland 1998, 1999; Spain: Martínez-Vilalta et al. 2002; France: Poulin and Lefebvre 2002).
The reed warbler (Acrocephalus scirpaceus) is the most abundant species in the area. It can nest in any wetland habitat that contains reeds (where the nest is built), but is more abundant in upstream rather than in lacustrine reedbeds. It defends very small territories and can therefore reach high densities (up to 9 couples/ha, Antoniazza 1979).
Savi’s warbler (Locustella luscinioides) lives mostly in between dryer reedbeds and wetter sedge fens, avoiding lacustrine reedbeds. The nest is fixed to the vegetation at approximately 20 cm above water, which makes it very vulnerable to floods (Aebischer, personnal communication).
The common reed bunting (Emberiza schoeniclus) inhabits the whole range of wetland habitats with the exception of lacustrine reedbeds. It occupies rather large territories (on average 0.5 ha) and often feeds far from the nest (Keiser 2007). The nest is built close to the ground in the vegetation.
The water rail (Rallus aquaticus) lives in the most humid part of the marshes (Antoniazza 1979; Brambilla and Rubolini 2004). It places its nests on the vegetation, close to water level. Both the male and the female can sing (Géroudet 1994), which renders the delineation of territories extremely hard.
The bearded reedling (Panurus biarmicus) mostly occupies the transition between wet sedge meadows and reedbeds. This social species nests in small colonies without properly defending territories. When population densities become too high or when habitat suitability decreases, the species forms swarms that disperse to another area, often dozens of kilometres away (Axell 1966; Maumary et al. 2007). The population of the Grande Cariçaie is most likely a mix between the P. b. biarmicus and P. b. russicus subspecies, colonized by such a swarm coming from the Dutch polders at the end of the 1970s. It is naturally absent from Cheyres, most likely because of lack of enough suitable habitats.
Vegetation structure
Since mowing was primarily aimed to control the spread of woody species, we first estimated how successful mowing was at containing shrubs. We estimated shrub layer cover in 3 × 5 m permanent plots placed both in mown (10 plots) and unmown (6 plots) areas in Cheyres during 4 years: 2008, 2009, 2010 and 2012. The exact location of the plots is presented in Fig. S2. Repeating these measurements over 4 years allowed us to account for variance in vegetation cover caused by mowing and random fluctuations. The cover of the shrub layer was always evaluated by the same observer and was attributed to one of the following classes: 0 = very scarce; 1 = 0–1%; 2 = 2–3%; 3 = 4–5%; 4 = 6–10%; 5 = 11–15%; 6 = 16–25%; 7 = 26–50%; 8 = 51–75%; 9 = 76–100%. This method is adapted from Braun-Blanquet (described in Poore 1955) but has more classes to reflect differences at low cover better. We took the mean coverage value of each class for subsequent analyses.
Bird censuses
The censuses were conducted using the spot-mapping method, as described in Antoniazza (1979). We chose this method over mist-netting because it directly records singing males, which is a better proxy for the number of territories than caught individuals. It also gives a more precise estimate of the nest’s location, which is more relevant for instance with reed buntings, which can be seen foraging several hundred meters away from the nest. In addition, mist netting would give biased results due to the nets being more easily detected in recently mown plots, especially at the beginning of the season when vegetation has not fully regrown yet.
Surveys were performed by the same observer all along the experiment, soon after dawn or before dusk when singing activity is maximal. The observer was walking along parallel transects spaced 50 meters apart within the survey location. Birds were identified based on their songs or by eye and their position was reported on a map. Every location was visited seven to eight times within the season. Simultaneous contacts were taken into account and nests and fledgelings were reported when observed.
At the end of the field season, territories were drawn for each species and attributed to plots based on the number and type of contacts in each plot. When a territory was shared between two or more plots, or when it was located partly outside the study area, territories were distributed between the plots. If contacts were scattered equally between two plots, each were attributed 0.5 territories. If one plot contained most of the contacts or the nest, this plot was attributed 0.75 territories, and the other 0.25. The only exception is the reed warbler, which is present in high densities and does not defend proper territories. The number of territories per plot for this species was therefore counted as the mean between the three highest numbers of singing males recorded over all passages.
Censuses started in 1985 in Cheyres, in 1987 in Gletterens and in 1988 in Chevroux and were conducted between April 10th and July 10th. In 1994 and 1995, a flood occurred during the breeding season, destroying many nests of reed bunting and Savi’s warbler. In these 2 years, we used the number of territories before the flood, which was always higher than after the flood.
Statistical analyses
All analyses were conducted in R 3.2.2 (R Core Team 2015). We evaluated whether shrub cover was significantly different in mown vs unmown plots using a linear mixed model with the plot and the year of observation as random factors. The models were built in the R package lme4 (Bates et al. 2013). Because of the non-normal distribution of the data, we confirmed the significance of our models using a permutation ANOVA approach. We compared the observed F value to a distribution of simulated F values obtained by permuting the treatments for 100,000 iterations. The p value was computed as the proportion of simulated F values equal or higher than the observed value.
The population trends were estimated independently for each species and location using linear models with the total density of territories per hectare as response and the year as explanatory variable. A population was considered decreasing if the slope was significantly negative, increasing if the slope was significantly positive, and stable otherwise. Swiss population trends (excluding the Grande Cariçaie) were added as additional controls for evolution unrelated to management. They were obtained from the Swiss ornithological institute and are based on yearly surveys (3–8 visits per year) by experienced volunteers of approximately 80 wetlands all across the country since 1990. Management at these locations is heterogeneous. In many but not all cases the sedge meadows, sometimes also parts of the reed beds are cut once in August or September.
Trends were computed in TRIM, which accounts for missing counts and calculates standard errors using Poisson regression (Pannekoek and van Strien 2001; Van Strien et al. 2001). This was not possible for the bearded reedling as this species is almost absent from Switzerland outside the Grande Cariçaie reserves.
For each species, we tested the effect of years since last cut on the number of territories. For this purpose, we built Generalized Linear Mixed Models (GLMM) with the counts of territories as the response variable with Poisson distribution. We used the age since last cut as the explanatory variable, and included the plot nested within the location as well as the year of observation as random independent factors. Different plot surfaces were taken into account as an offset. Chevroux, being an unmown control for population fluctuations, was not included in this analysis. We performed multiple comparisons between mowing ages using the R package multcomp (described in Hothorn et al. 2008).
We used our GLMMs to predict territory density under different mowing scenarios. For each species, we predicted territory density and averaged them per year. We then simulated different mowing scenarios (mowing every year, every 2 years, etc.) by averaging predicted densities for all concerned years. For instance, for the “every three years” scenario, we averaged predicted densities for years 1, 2 and 3. Since years 5 and 6 were aggregated in previous analyses, we used the same value for these 2 years.
Results
Vegetation structure
We found that shrub cover was lower in mown that in unmown plots (LMM, p = 2.2 × 10−4; permutation LMM, p < 10−4; Figs. 2, S4), confirming its efficiency in reducing shrubs development.
Long-term population trends
Most populations (nine out of 14) were stable over the 30-year period (Table 1; Fig. 3). Only two were decreasing (reed warbler and water rail, both in Cheyres) and three increasing (reed warbler in Gletterens and Chevroux and reed bunting in Chevroux). In all cases, the slopes were close to 0 (range −0.03 to 0.06). No species was consistently increasing or decreasing in either mown areas. In the rest of Switzerland, two species (reed bunting and water rail) were decreasing, one was stable (reed warbler) and one was strongly increasing (Savi’s warbler).
Population trends for each species per location. Left axis: territory density in the three Grande Cariçaie locations. Chevroux is a control and was never mown. Right axis: Swiss index, expressed as percentage of population size in 1990. Error bars represent standard error, given by a Poisson regression. Formal tests of the trends is provided in Table 1. (Color figure online)
Optimal mowing regime
We tested whether the year since last cut was influencing the density of bird territories. Our GLMM analyses revealed a significant effect for all tested species (reed warbler: χ2 = 428.77, df = 5, p < 2 × 10−16; Savi’s warbler: χ2 = 137.46, df = 5, p < 2 × 10−16; reed bunting: χ2 = 168.39, df = 5, p < 2 × 10−16; water rail: χ2 = 66.992, df = 5, p = 4.3 × 10−13; bearded reedling: χ2 = 55.085, df = 5, p = 1.25 × 10−10), confirming the conclusions of previous, shorter-termed studies (e.g. Graveland 1999). Multiple comparisons showed that this was mostly due to plots of age 1 and 2 being of much lower quality (Fig. 4). Territory density in age 1 was consistently lower than in plots of any other ages. Plots of age 2 had significantly higher density than age 1, and significantly less than in all other ages except for the water rail (where all plots older than age 1 showed similar densities), and the reed warbler and the bearded reedling (where plots of age 2 were not significantly different from the controls). Territory density was maximal in plots of ages 3 to 5–6 depending on the species. Control plots had systematically lower densities than those of ages 3 to 5–6, but not significantly so.
Effect of mowing regime on breeding birds. Boxplots represent the residuals of a Generalized Linear Mixed Model modelling the number of territories with the year, location and plot as random factors, and plot surface as an offset. Significantly different groups are denoted by different letters (all p < 0.01). (Color figure online)
Our prediction of average bird densities for different mowing scenarios showed an increase in overall densities until year 6 for all five species (Fig. 5). In the bearded reedling, predicted density under “mowing every 6 years” scenario was even lower than in control, unmown, plots.
Predited territory density under different mowing scenarios. For each species, average density at a given year is computed as the mean between predicted average densities for all years from 1 to this particular year. The reed warbler follows a separate axis (right side) because overall density is much higher in this species. Average density in control, unmown, plots is also represented for comparison. (Color figure online)
Discussion
All previous studies addressing the effect of wetland mowing on the fauna were restricted to one or a few years (e.g. Graveland 1999; Schmidt et al. 2005). Although such studies are helpful in assessing the negative effects of mowing mediated by the reduction of favourable surfaces within that short time-frame, they are insufficient for revealing the long-term effects on vegetation structure and communities, and their consequences on the fauna. In addition, our long-term population trends showed large within-site oscillations over the years (up to threefold in some species) despite little long-term evolution. Therefore, results from studies conducted over short time periods could be confounded by only optimal habitats being occupied during years of low density, while less optimal habitats could also be chosen when density is higher for reasons unrelated to management (e.g. climate, mortality during migration, food availability in wintering grounds). To fill this gap, we measured the effect of mowing on habitat suitability for the five most abundant breeding birds in wetland reserves in Switzerland over a period of 30 years.
Vegetation structure
We found that mowing efficiently reduced shrub development by cutting growing saplings. It is important to note, however, that cutting stems does not totally suppress invasion by shrubs. Alnus, Frangula and Salix, which are the most common woody species that settle in wet meadows, are able to regrow when cut. Furthermore, mowing could even favour the establishment of dispersing seeds by decreasing competition for light with herbaceous species, especially at the borders of forest where propagules are numerous. Mowing therefore restrains shrubs growth, but does not decrease the number of individual woody plants. Thus, shrub cover could quickly increase if mowing is abandoned.
Another possible drawback of mowing is the homogenization of plant communities at the landscape level. Mowing can sometimes increase local diversity, for instance, through the opening of vegetation that leaves some space for pioneer species, or through the creation of ruts by the mowing machine. However, applying the same constraint to a large area can also buffer other, more localised, perturbations, thus decreasing large-scale species and landscape diversity (Deák et al. 2015).
In the Recommendations for management section, we discuss additional management measures that could be implemented to overcome these limitations.
Long-term population trends
Our long-term trends analysis showed no consistent change among species or location. This result confirms that rotational mowing can be used without long-term harm to breeding birds. This result is likely dependant on mowing plots being kept small enough (not more than a few hectares) so as to leave enough favourable habitat each year. Mowing larger surfaces at once would likely cause more casualties in birds and even more so in taxa with more limited dispersal abilities (Schmidt et al. 2005). In addition, freshly cut plots adjacent to ones more suitable for breeding can serve as foraging grounds for birds breeding in the surroundings, thus potentially increasing density there. The absence of overall detectable demographic change when mowing started suggests that birds redistributed in suitable nesting grounds.
Despite no consistent population decrease, two out of the four species present in Cheyres decreased slightly at this location. The two species with decreasing populations (water rail and reed warbler) mostly occupy the wettest portion of the landscape. These results could therefore be explained by a succession towards drier communities despite management, as shown by Güsewell and Le Nédic (2004).
In order to control for factors independent of management, we assessed population trends in the rest of Switzerland. The reed bunting and the water rail were decreasing at national level, while neither of them showed a consistent decrease in the Grande Cariçaie. This suggests that management in the Grande Cariçaie reserves helped to maintain the populations stable despite national decline. The reed warbler was stable in Switzerland and did not consistently increase or decrease in the Grande Cariçaie, indicating that management there is sufficient to sustain stable populations of that species. The only species that was strongly increasing in Switzerland and stable in the Grande Cariçaie was Savi’s warbler. This species started breeding in Switzerland in 1956 and quickly expanded thereafter. It first colonized the Grande Cariçaie, and started expanding to other parts of Switzerland later on (Aebischer and Antoniazza 1995). For this reason, the observed strong increase likely results from ongoing range expansion in Switzerland, while the Grande Cariçaie population has likely reached its carrying capacity already.
Overall, these results show that rotational mowing has no long-term detrimental effects on the demography of the most abundant species at our location.
Optimal mowing regime
In addition to assessing the long-term effects of mowing on breeding birds, our study was aimed at determining the optimal mowing regime for birds. We showed that plots of age 1 are almost systematically avoided, which is consistent with previous results (Graveland 1999; Poulin and Lefebvre 2002). This is most likely explained by the short growing vegetation offering very few breeding opportunities, as most of the nests in age 1 plots were located in restricted patches that could not be mown due to their proximity with water or an obstacle.
Plots of age 2 were also significantly less favourable than older ones for all species except the water rail. This is consistent with the common expectation that more complex structures are more beneficial because they offer more opportunities for breeding, and it shows that vegetation structure takes more than one full growing season to reach an optimal level for birds. The water rail lives in the wettest part of the marshes (Brambilla and Rubolini 2004), where reed is most abundant. Vegetation structure there is different from upstream meadows in that vegetation is more dense, so that reed size and structure could become favourable sooner than in the rest of the marshes. In addition, this species feeds mostly on aquatic invertebrates and thus depends less on high reed stem density than the other species.
Plots of age 3 and older held the highest bird density. Vegetation structure evolution seems to slow after 3 years, with older, dead, decaying stems being replaced by new ones. Interestingly, bird density was systematically lower in unmown controls than year 3 and older plots, although this trend was significant in none of the species. The most likely explanation is that habitat quality quickly improves after mowing, and then plateaus. Only after several years or decades will the habitat shift towards drier and woody communities, thus preventing human life-scaled studies to detect a significant effect.
The fact that bird density is highest in plots of age 3 and 4 does not mean that mowing every 3–4 years is optimal. Indeed, delaying a cut to prevent going through age 1, which harbours very low densities, can be profitable on the long term. Consistently, we observed that predicted average densities were highest in our “every 6 years” scenario, and average density would likely keep on increasing thereafter. Bird density might, however, not rise indefinitely and birds might distribute more evenly if more favourable habitats become available, as was the case before mowing started.
Mowing regimes reported from other studies are often much more frequent than what our results suggest would be optimal (e.g. Graveland 1999; Poulin and Lefebvre 2002; Vadász et al. 2008). One factor that could explain these differences could be higher ecosystem productivity (and therefore vegetation regrowth rate) at other locations, causing the habitat to reach an optimal state more quickly after a cut. However, productivity is often thought to follow a latitudinal gradient (Yu et al. 2013; Gillman et al. 2015), and the Grande Cariçaie reserves are located in central Europe, at an intermediate latitude. Therefore, differences in productivity alone are unlikely to explain why optimal mowing regime at the Grande Cariçaie would be much less frequent than at other locations. For this reason, we encourage managers and researchers to assess the effect of delayed mowing at locations where mowing frequency is higher to (i) verify whether decreasing mowing frequency could favour birds and other taxa and (ii) investigate what factors are responsible for site-specific differences in optimal mowing frequency.
Recommendations for management
Based on our results, we suggest that mowing should take place no more frequently than every 3 years so that optimal habitats be available every year. Ideally, mowing should even not occur more often than every 6 years to delay periods of poor quality that follow a cut. We stress, however, that the aim of managers must not be to “produce birds”, but to preserve natural environments from alteration by conflicting human activities.
Adapting management to local habitat type allows to specifically optimise management to different taxa. While humid, reed-dominated, reedbeds provide ideal nesting grounds for many bird species, drier, sedge-dominated meadows are characterized by a much higher richness in plants and invertebrates. They are also the most susceptible to being invaded by shrubs. Mowing could therefore be spaced in the most humid parts of the marshes, but maintained more frequent in drier areas.
In addition, complementary interventions could be initiated to mitigate the potential drawbacks of mowing. Grazing by cattle is one such alternative. Unlike mowing, which tends to create homogeneous habitats, grazing has a random effect on vegetation, with some stems being eaten and others remaining. Another major difference is that organic matter is not removed when grazed, while it is often exported when mown. Depending on the breed, cattle can be more or less choosy and therefore efficient at removing woody species. The impact of grazing on the soil is also more heterogeneous, which can, under some circumstances, favour the establishment of shrubs. To prevent this, grazing is often used in combination with burning, with promising results (Middleton et al. 2006; Little et al. 2015; Mérő et al. 2015; Mester et al. 2015). Preliminary tests of grazing are ongoing in the Grande Cariçaie reserves and will give insights onto how widely applicable this technique is.
A more specific intervention is the targeted tearing of shrubs. Tests were conducted in the Grande Cariçaie reserves using an excavator terminated by forceps that pulls out both above- and below-ground parts of the plant, thus durably removing shrubs. This method presents the advantage of selectively removing shrubs to decrease competition for light and prevent deviation of the habitat towards wooded communities, while keeping sedge and reed structure intact. Its major drawbacks lie in its costs. It is time-consuming and, unlike grazing and mowing, produces no resource that can be valorised to compensate for its costs. Formal testing is still lacking, but shrub tearing is surely a promising technique in situations where economic productivity is not the central concern.
Another intervention that specifically targets the most humid part of the marshes is the recreation of open water bodies by excavating soil over a few dozen centimetres. These water bodies will be colonised by vegetation over the course of a few decades, thus recreating the natural succession of vegetation stages. This technique presents the advantage of necessitating only a single intervention, while lasting for several decades. However, as it is quite destructive, it must be executed with parsimony, avoiding when possible places of specific biological value.
Conclusion
Our study showed that rotational mowing has no long-term negative effect on bird populations. Instead, birds likely redistribute each year to occupy optimal areas. Recently mown plots were almost systematically avoided, but attractiveness quickly rose thereafter. Plots were most attractive 3 years after being last mown and thereafter. Unmown plots were slightly less attractive than those mown several years previously, likely because the habitat starts to shift towards wooden communities. The wide distribution of the focus species and habitats makes our study applicable to most European wetlands. Based on our results, we recommend that areas important for birds be mown every 3 years at least, but ideally even less frequently, and that additional interventions be used as a complement for combating the establishment of shrubs. Some of these other strategies have yet to be tested formally, and this is an exciting task for further studies. Additional studies should also focus on the interplay between birds and other taxa, to verify to what extent birds are representative of overall habitat quality, and to ensure birds are not favoured over other taxa.
References
Aebischer A, Antoniazza M (1995) Verbreitung und Bestandsentwicklung des Rohrschwirls Locustella luscinioides in der Schweiz. Ornithol Beob 92:435–453
Antoniazza M (1979) Les oiseaux nicheurs des marais non boisés de la rive sud du lac de Neuchâtel. University of Neuchâtel, Neuchâtel
Axell HE (1966) Eruptions of bearded tits during 1959–65. Br Birds 59:513–543
Báldi A (2005) The importance of temporal dynamics of edge effect in reedbed design: a 12-year study on five bird species. Wetl Ecol Manag 13:183–189
Bates D, Maechler M, Bolker B (2013) lme4: linear mixed-effects models using S4 classes. R Packag. version 0.999999-2. 999999
Brambilla M, Rubolini D (2004) Water Rail Rallus aquaticus breeding density and habitat preferences in Northern Italy. Ardea 92:11–17
Brinson MM, Malvárez AI (2002) Temperate freshwater wetlands: types, status, and threats. Environ Conserv 29:115–133
Butler SJ, Freckleton RP, Renwick AR, Norris K (2012) An objective, niche-based approach to indicator species selection. Methods Ecol Evol 3:317–326
Davidson NC (2014) How much wetland has the world lost? Long-term and recent trends in global wetland area. Mar Freshw Res 65:934–941
Deák B, Valkó O, Török P, Kelemen A, Tóth K, Miglécz T, Tóthmérész B (2015) Reed cut, habitat diversity and productivity in wetlands. Ecol Complex 22:121–125
Dudgeon D, Arthington AH, Gessner MO, Kawabata Z-I, Knowler DJ, Lévêque C, Naiman RJ, Prieur-Richard A-H, Soto D, Stiassny MLJ, Sullivan CA (2006) Freshwater biodiversity: importance, threats, status and conservation challenges. Biol Rev Camb Philos Soc 81:163–182
Everard M (2008) Selection of taxa as indicators of river and freshwater wetland quality in the UK. Aquat Conserv Mar Freshw Ecosyst 18:1052–1061
Géroudet P (1994) Grands Echassiers, Gallinacés, Râles d’Europe. Delachaux et Niestlé, Paris
Gillman LN, Wright SD, Cusens J, Mcbride PD, Malhi Y, Whittaker RJ (2015) Latitude, productivity and species richness. Glob Ecol Biogeogr 24:107–117
Graveland J (1998) Reed die-back, water level management and the decline of the Great Reed Warbler Acrocephalus arundinaceus in The Netherlands. Ardea 86:187–201
Graveland J (1999) Effects of reed cutting on density and breeding success of Reed Warbler Acrocephalus scirpacaeus and Sedge Warbler A. schoenobaenus. J Avian Biol 30:469–482
Güsewell S, Le Nédic C (2004) Effects of winter mowing on vegetation succession in a lakeshore fen. Appl Veg Sci 7:41–48
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363
Keiser M (2007) Habitat occupation strategies and breeding behaviour in reed buntings (Emberiza schoeniclus). University of Freiburg, Freiburg
Little IT, Hockey PAR, Jansen R (2015) Assessing biodiversity integrity for the conservation of grazed and burnt grassland systems: avian field metabolic rates as a rapid assessment tool. Biodivers Conserv 24:1443–1471
Martínez-Vilalta J, Bertolero A, Bigas D, Paquet J-Y, Martínez-Vilalta A (2002) Habitat selection of passerine birds nesting in the Ebro Delta reedbeds (NE Spain): management implications. Wetlands 22:318–325
Maumary L, Vallotton L, Knaus P (2007) Les oiseaux de Suisse. Station ornithologique suisse, Sempach
Mérő TO, Lontay L, Lengyel S (2015) Habitat management varying in space and time: the effects of grazing and fire management on marshland birds. J Ornithol 156:579–590
Mester B, Szalai M, Mérő TO, Puky M, Lengyel S (2015) Spatiotemporally variable management by grazing and burning increases marsh diversity and benefits amphibians: a field experiment. Biol Conserv 192:237–246
Middleton BA, Holstein B, van Diggelen R (2006) Biodiversity management of fens and fen meadows by grazing, cutting and burning. Appl Veg Sci 9:307–316
Okruszko T, Duel H, Acreman M, Grygoruk M, Flörke M, Schneider C (2011) Broad-scale ecosystem services of European wetlands—overview of the current situation and future perspectives under different climate and water management scenarios. Hydrol Sci J 56:1501–1517
Pannekoek J, van Strien AJ (2001) TRIM3 manual (TRends & Indices for Monitoring data)
Poore MED (1955) The use of phytosociological methods in ecological investigations: I. The Braun-Blanquet system. J Ecol 43:226–244
Poulin B, Lefebvre G (2002) Effect of winter cutting on the passerine breeding assemblage in French Mediterranean reedbeds. Biodivers Conserv 11:1567–1581
Quintana-Ascencio PF, Fauth JE, Castro Morales LM, Ponzio KJ, Hall D, Snyder K (2013) Taming the beast: managing hydrology to control carolina willow (Salix caroliniana) seedlings and cuttings. Restor Ecol 21:639–647
R Core Team (2015) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Sattler T, Pezzatti GB, Nobis MP, Obrist MK, Roth T, Moretti M (2014) Selection of multiple umbrella species for functional and taxonomic diversity to represent urban biodiversity. Conserv Biol 28:414–426
Schmidt MH, Lefebvre G, Poulin B, Tscharntke T (2005) Reed cutting affects arthropod communities, potentially reducing food for passerine birds. Biol Conserv 121:157–166
Vadász C, Némét Á, Biró C, Csörgó T (2008) The effect of reed cutting on the abundance and diversity of breeding passerines. Acta Zool Acad Sci Hungaricae 54:177–188
Valkama E, Lyytinen S, Koricheva J (2008) The impact of reed management on wildlife: a meta-analytical review of European studies. Biol Conserv 141:364–374
Van Strien AJ, Pannekoek J, Gibbons DW (2001) Indexing European bird population trends using results of national monitoring schemes: a trial of a new method. Bird Study 48:200–213
Yu GR, Zhu XJ, Fu YL, He HL, Wang QF, Wen XF, Li XR, Zhang LM, Zhang L, Su W, Li SG, Sun XM, Zhang YP, Zhang JH, Yan JH, Wang HM, Zhou GS, Jia BR, Xiang WH, Li YN, Zhao L, Wang YF, Shi PL, Chen SP, Xin XP, Zhao FH, Wang YY, Tong CL (2013) Spatial patterns and climate drivers of carbon fluxes in terrestrial ecosystems of China. Glob Change Biol 19:798–810
Acknowledgements
Christa Glauser and Werner Müller (Ala, Schweizerische Gesellschaft für Vogelkunde und Vogelschutz) and Martin Weggler (Orniplan AG, Zürich) and volunteer ornithologists have collected the national trends data. The national data is managed by Claudia Müller and Hans Schmid (Swiss Ornithological Institute). We wish to thank all members of the Association de la Grande Cariçaie for insightful discussions and comments on the manuscript; Jean-Yves Humbert for his advices on the vegetation analyses; Sylvain Antoniazza, Camille-Sophie Cozzarolo, Sarah Schmid, Nicolas Perrin, Ailsa McKenzie and two anonymous reviewers for comments on the manuscript, and Daniel Lee Jeffries for comments and proofreading the manuscript. Open-access publishing fees were paid for by a 242 Grant from the Hubert Tuor Foundation to GL.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by David Hawksworth.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Antoniazza, M., Clerc, C., Le Nédic, C. et al. Long-term effects of rotational wetland mowing on breeding birds: evidence from a 30-year experiment. Biodivers Conserv 27, 749–763 (2018). https://doi.org/10.1007/s10531-017-1462-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-017-1462-1