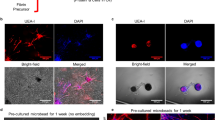
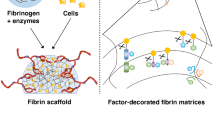
Abstract
Survival of tissue engineered constructs after implantation depends heavily on induction of a vascular response in host tissue, promoting a quick anastomosis of the cellular graft. Additionally, implanted constructs typically induce fibrous capsule formation, effectively preventing graft integration with host tissue. Previously we described the development of a high density microtemplated fibrin scaffold for cardiac tissue engineering applications with tunable degradation and mechanical properties which promoted seeded cell survival and organization in vitro (Thomson et al., Tissue Eng Part A, 2013). Scaffold degradation in vitro was controllable by addition of the serine protease inhibitor aprotinin and/or the fibrin cross-linker Factor XIII (FXIII). The goal of this study was to assess host tissue responses to these fibrin scaffold formulations by determining effects on scaffold degradation, angiogenic responses, and fibrous capsule formation in a subcutaneous implant model. Aprotinin significantly decreased scaffold degradation over 2 weeks of implantation. A significant increase in capillary infiltration of aprotinin implants was found after 1 and 2 weeks, with a significantly greater amount of capillaries reaching the interior of aprotinin scaffolds. Interestingly, after 2 weeks the aprotinin scaffolds had a significantly thinner, yet apparently more cellular fibrous capsule than unmodified scaffolds. These results indicate aprotinin not only inhibits fibrin scaffold degradation, but also induces significant responses in the host tissue. These included an angiogenic response resulting in increased vascularization of the scaffold material over a relatively short period of time. In addition, aprotinin release from scaffolds may reduce fibrous capsule formation, which could help promote improved integration of cell-seeded scaffolds with host tissue.






Similar content being viewed by others
References
Thomson KS, Korte FS, Giachelli CM, Ratner BD, Regnier M, Scatena M (2013) Prevascularized microtemplated fibrin scaffolds for cardiac tissue engineering applications. Tissue Eng Part A 19(7–8):967–977
Linnes MP, Ratner BD, Giachelli CM (2007) A fibrinogen-based precision microporous scaffold for tissue engineering. Biomaterials 28:5298
Naderi H, Matin MM, Bahrami AR (2011) Review paper: critical issues in tissue engineering: biomaterials, cell sources, angiogenesis, and drug delivery systems. J Biomater Appl 26:383
Anderson JM (2001) Biological responses to materials. Annu Rev Mater Res 31:81
Cosson M, Debodinance P, Boukerrou M, Chauvet MP, Lobry P, Crépin G, Ego A (2003) Mechanical properties of synthetic implants used in the repair of prolapse and urinary incontinence in women: which is the ideal material? Int Urogynecol J Pelvic Floor Dysfunct 14:169
Kidd KR, Nagle RB, Williams SK (2002) Angiogenesis and neovascularization associated with extracellular matrix-modified porous implants. J Biomed Mater Res 59:366
Bootle-Wilbraham C, Tazzyman S, Thompson W, Stirk C, Lewis C (2001) Fibrin fragment E stimulates the proliferation, migration and differentiation of human microvascular endothelial cells in vitro. Angiogenesis 4:269
Ge M, Ryan TJ, Lum H, Malik AB (1991) Fibrinogen degradation product fragment D increases endothelial monolayer permeability. Am J Physiol 261:L283
Monroe DM, Hoffman M (2006) What does it take to make the perfect clot? Arterioscler Thromb Vasc Biol 26:41
Ahmed TA, Dare EV, Hincke M (2008) Fibrin: a versatile scaffold for tissue engineering applications. Tissue Eng Part B Rev 14:199
Barsotti MC, Felice F, Balbarini A, Di Stefano R (2011) Fibrin as a scaffold for cardiac tissue engineering. Biotechnol Appl Biochem 58:301
Chen X, Aledia AS, Ghajar CM, Griffith CK, Putnam AJ, Hughes CC, George SC (2009) Prevascularization of a fibrin-based tissue construct accelerates the formation of functional anastomosis with host vasculature. Tissue Eng Part A 15:1363
Giraud MN, Ayuni E, Cook S, Siepe M, Carrel TP, Tevaearai HT (2008) Hydrogel-based engineered skeletal muscle grafts normalize heart function early after myocardial infarction. Artif Organs 32:692
Ryu JH, Kim IK, Cho SW, Cho MC, Hwang KK, Piao H, Piao S, Lim SH, Hong YS, Choi CY, Yoo KJ, Kim BS (2005) Implantation of bone marrow mononuclear cells using injectable fibrin matrix enhances neovascularization in infarcted myocardium. Biomaterials 26:319
Christman KL, Fok HH, Sievers RE, Fang Q, Lee RJ (2004) Fibrin glue alone and skeletal myoblasts in a fibrin scaffold preserve cardiac function after myocardial infarction. Tissue Eng 10:403
Christman KL, Vardanian AJ, Fang Q, Sievers RE, Fok HH, Lee RJ (2004) Injectable fibrin scaffold improves cell transplant survival, reduces infarct expansion, and induces neovasculature formation in ischemic myocardium. J Am Coll Cardiol 44:654
Chekanov V, Akhtar M, Tchekanov G, Dangas G, Shehzad MZ, Tio F, Adamian M, Colombo A, Roubin G, Leon MB, Moses JW, Kipshidze NN (2003) Transplantation of autologous endothelial cells induces angiogenesis. Pacing Clin Electrophysiol 26:496
Huang YC, Khait L, Birla RK (2007) Contractile three-dimensional bioengineered heart muscle for myocardial regeneration. J Biomed Mater Res A 80:719
Jockenhoevel S, Zund G, Hoerstrup SP, Chalabi K, Sachweh JS, Demircan L, Messmer BJ, Turina M (2001) Fibrin gel—advantages of a new scaffold in cardiovascular tissue engineering. Eur J Cardiothorac Surg 19:424
Stevens K, Kreutziger K, Dupras S, Korte F, Regnier M, Muskheli V, Nourse M, Bendixen K, Reinecke H, Murry C (2009) Physiological function and transplantation of scaffold-free and vascularized human cardiac muscle tissue. Proc Natl Acad Sci USA 106:16568
Tulloch NL, Muskheli V, Razumova MV, Korte FS, Regnier M, Hauch KD, Pabon L, Reinecke H, Murry CE (2011) Growth of engineered human myocardium with mechanical loading and vascular coculture. Circ Res 109:47
Koutsioumpa M, Hatziapostolou M, Mikelis C, Koolwijk P, Papadimitriou E (2009) Aprotinin stimulates angiogenesis and human endothelial cell migration through the growth factor pleiotrophin and its receptor protein tyrosine phosphatase beta/zeta. Eur J Pharmacol 602:245
Cholewinski E, Dietrich M, Flanagan TC, Schmitz-Rode T, Jockenhoevel S (2009) Tranexamic acid–an alternative to aprotinin in fibrin-based cardiovascular tissue engineering. Tissue Eng Part A 15:3645
Ahmed TA, Griffith M, Hincke M (2007) Characterization and inhibition of fibrin hydrogel-degrading enzymes during development of tissue engineering scaffolds. Tissue Eng 13:1469
McEvoy MD, Reeves ST, Reves JG, Spinale FG (2007) Aprotinin in cardiac surgery: a review of conventional and novel mechanisms of action. Anesth Analg 105:949
Bull DA, Maurer J (2003) Aprotinin and preservation of myocardial function after ischemia-reperfusion injury. Ann Thorac Surg 75:S735
Hickerson WL, Nur I, Meidler R (2011) A comparison of the mechanical, kinetic, and biochemical properties of fibrin clots formed with two different fibrin sealants. Blood Coagul Fibrinolysis 22:19
Buser Z, Kuelling F, Liu J, Liebenberg E, Thorne KJ, Coughlin D, Lotz JC (1976) Biological and biomechanical effects of fibrin injection into porcine intervertebral discs. Spine 36:E1201
Lorentz KM, Kontos S, Frey P, Hubbell JA (2011) Engineered aprotinin for improved stability of fibrin biomaterials. Biomaterials 32:430
Ye Q, Zünd G, Benedikt P, Jockenhoevel S, Hoerstrup SP, Sakyama S, Hubbell JA, Turina M (2000) Fibrin gel as a three dimensional matrix in cardiovascular tissue engineering. Eur J Cardiothorac Surg 17:587
Smith JD, Chen A, Ernst LA, Waggoner AS, Campbell PG (2007) Immobilization of aprotinin to fibrinogen as a novel method for controlling degradation of fibrin gels. Bioconjug Chem 18:695
Fritz H, Wunderer G (1983) Biochemistry and applications of aprotinin, the kallikrein inhibitor from bovine organs. Arzneimittelforschung 33:479
Carmona P, Molina M, Rodríguez-Casado A (2003) Raman study of the thermal behaviour and conformational stability of basic pancreatic trypsin inhibitor. Eur Biophys J 32:137
Bryers JD, Giachelli CM, Ratner BD (2012) Engineering biomaterials to integrate and heal: the biocompatibility paradigm shifts. Biotechnol Bioeng 109:1898
Thompson WD, Smith EB, Stirk CM, Marshall FI, Stout AJ, Kocchar A (1992) Angiogenic activity of fibrin degradation products is located in fibrin fragment E. J Pathol 168:47
Dvorak HF, Harvey VS, Estrella P, Brown LF, McDonagh J, Dvorak AM (1987) Fibrin containing gels induce angiogenesis: implications for tumor stroma generation and wound healing. Lab Invest 57:673
van Hinsbergh VW, Collen A, Koolwijk P (2001) Role of fibrin matrix in angiogenesis. Ann NY Acad Sci 936:426
Jurasz P, Santos-Martinez MJ, Radomska A, Radomski MW (2006) Generation of platelet angiostatin mediated by urokinase plasminogen activator: effects on angiogenesis. J Thromb Haemost 4:1095
Radziwon-Balicka A, de la Moncada Rosa C, Zielnik B, Doroszko A, Jurasz P (2013) Temporal and pharmacological characterization of angiostatin release and generation by human platelets: implications for endothelial cell migration. PLoS One 8:e59281
Madden LR, Mortisen DJ, Sussman EM, Dupras SK, Fugate JA, Cuy JL, Hauch KD, Laflamme MA, Murry CE, Ratner BD (2010) Proangiogenic scaffolds as functional templates for cardiac tissue engineering. Proc Natl Acad Sci USA 107:15211
Hill GE, Diego RP, Stammers AH, Huffman SM, Pohorecki R (1998) Aprotinin enhances the endogenous release of interleukin-10 after cardiac operations. Ann Thorac Surg 65:66
McEvoy MD, Taylor AG, Zavadzkas JA, Mains IM, Ford RL, Stroud RE, Jeffords LB, Beck CU, Reeves ST, Spinale FG (2008) Aprotinin exerts differential and dose-dependent effects on myocardial contractility, oxidative stress, and cytokine release after ischemia-reperfusion. Ann Thorac Surg 86:568
Asimakopoulos G, Lidington EA, Mason J, Haskard DO, Taylor KM, Landis RC (2001) Effect of aprotinin on endothelial cell activation. J Thorac Cardiovasc Surg 122:123
Hill GE, Pohorecki R, Alonso A, Rennard SI, Robbins RA (1996) Aprotinin reduces interleukin-8 production and lung neutrophil accumulation after cardiopulmonary bypass. Anesth Analg 83:696
Brown BN, Valentin JE, Stewart-Akers AM, McCabe GP, Badylak SF (2009) Macrophage phenotype and remodeling outcomes in response to biologic scaffolds with and without a cellular component. Biomaterials 30:1482
Lee CG, Homer RJ, Zhu Z, Lanone S, Wang X, Koteliansky V, Shipley JM, Gotwals P, Noble P, Chen Q, Senior RM, Elias JA (2001) Interleukin-13 induces tissue fibrosis by selectively stimulating and activating transforming growth factor beta(1). J Exp Med 194:809
Rolfe B, Mooney J, Zhang B, Jahnke S, Le S-J, Chau Y-Q, Huang Q, Wang H, Campbell G, Campbell J (2011) The fibrotic response to implanted biomaterials: implications for tissue engineering. In: Eberli D (ed) Regenerative medicine and tissue engineering - cells and biomaterials. InTech. doi:10.5772/21790. Available from: http://www.intechopen.com/books/regenerative-medicine-and-tissue-engineering-cells-andbiomaterials/the-fibrotic-response-to-implanted-biomaterials-implications-for-tissue-engineering
Obokata H, Yamato M, Tsuneda S, Okano T (2011) Reproducible subcutaneous transplantation of cell sheets into recipient mice. Nat Protoc 6:1053
Lindbom L, Arfors KE (1984) Non-homogeneous blood flow distribution in the rabbit tenuissimus muscle: differential control of total blood flow and capillary perfusion. Acta Physiol Scand 122:225
Acknowledgments
The authors sincerely thank Mandy Lund, Matt Coons, Veronica Muskheli and Elizabeth Gay for their assistance with histological sample preparations and analysis, and Marc Takeno and Alex Chen for their assistance with polarized light imaging. This work was supported by National Institutes of Health (NIH) Grant R01HL064387 to MR, MS and CEM, and P01 HL094374, P01 GM081719, U01 HL100405, and R01 HL084642 to CEM.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thomson, K.S., Dupras, S.K., Murry, C.E. et al. Proangiogenic microtemplated fibrin scaffolds containing aprotinin promote improved wound healing responses. Angiogenesis 17, 195–205 (2014). https://doi.org/10.1007/s10456-013-9388-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10456-013-9388-z