Abstract
We present a novel methodology and strategy to predict pressures and flow rates in the global cardiovascular network in different postures varying from supine to upright. A closed-loop, multiscale mathematical model of the entire cardiovascular system (CVS) is developed through an integration of one-dimensional (1D) modeling of the large systemic arteries and veins, and zero-dimensional (0D) lumped-parameter modeling of the heart, the cardiac-pulmonary circulation, the cardiac and venous valves, as well as the microcirculation. A versatile junction model is proposed and incorporated into the 1D model to cope with splitting and/or merging flows across a multibranched junction, which is validated to be capable of estimating both subcritical and supercritical flows while ensuring the mass conservation and total pressure continuity. To model gravitational effects on global hemodynamics during postural change, a robust venous valve model is further established for the 1D venous flows and distributed throughout the entire venous network with consideration of its anatomically realistic numbers and locations. The present integrated model is proven to enable reasonable prediction of pressure and flow rate waveforms associated with cardiopulmonary circulation, systemic circulation in arteries and veins, as well as microcirculation within normal physiological ranges, particularly in mean venous pressures, which well match the in vivo measurements. Applications of the cardiovascular model at different postures demonstrate that gravity exerts remarkable influence on arterial and venous pressures, venous returns and cardiac outputs whereas venous pressures below the heart level show a specific correlation between central venous and hydrostatic pressures in right atrium and veins.
















Similar content being viewed by others
References
MacWilliam, J.A.: Postural effects on heart-rate and blood-pressure. Q. J. Exp. Physiol. 23, 1–33 (1933)
Martin-Du Pan, R.C., Benoit, R., Girardier, L.: The role of body position and gravity in the symptoms and treatment of various medical diseases. Swiss Med. Wkly. 134, 543–551 (2004)
Gisolf, J.: Postural changes in humans: effects of gravity on the circulation. [Ph.D. Thesis], University of Amsterdam, the Netherlands (2005)
Wright, H.P., Osborn, S.B.: Effect of posture on venous velocity, measured with 24NaCl. Br. Heart J. 14, 325–330 (1952)
Nicolaides, A.N.: Investigation of chronic venous insufficiency a consensus statement. Circulation 102, e126–e163 (2000)
Atta, H.M.: Varicose veins: role of mechanotransduction of venous hypertension. Int. J. Vasc. Med. 2012, 538627 (2012). doi:10.1155/2012/538627
Blomqvist, C.G., Stone, H.L.: Cardiovascular adjustments to gravitational stress. Compr. Physiol. 1025–1063 (2011). doi:10.1002/cphy.cp020328
Gisolf, J., Gisolf, A., Van Lieshout, J.J., et al.: The siphon controversy: an integration of concepts and the brain as baffle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 289, R627–R629 (2005)
Dawson, E.A., Secher, N.H., Dalsgaard, M.K., et al.: Standing up to the challenge of standing: a siphon does not support cerebral blood flow in humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 287, R911–R914 (2004)
Olesen, N.D., van Lieshout, J.J., Fisher, J.P., et al.: Case report: (Pre)syncopal symptoms associated with a negative internal jugular venous pressure. Front. Physiol. 5, 317 (2014)
Liu, H., Liang, F.Y., Wong, J., et al.: Multi-scale modeling of hemodynamics in the cardiovascular system. Acta Mech. Sin. 31, 446–464 (2015). doi:10.1007/s10409-015-0460-3
van de Vosse, F.N., Stergiopulos, N.: Pulse wave propagation in the arterial tree. Annu. Rev. Fluid Mech. 43, 467–499 (2011)
Taylor, C.A., Figueroa, C.A.: Patient-specific modeling of cardiovascular mechanics. Annu. Rev. Biomed. Eng. 11, 109–134 (2009)
Mynard, J.P., Smolich, J.J.: One-dimensional haemodynamic modeling and wave dynamics in the entire adult circulation. Ann. Biomed. Eng. 43, 1443–1460 (2015)
Liang, F.Y., Takagi, S., Himeno, R., et al.: Biomechanical characterization of ventricular-arterial coupling during aging: a multi-scale model study. J. Biomech. 42, 692–704 (2009)
Avolio, A.P.: Multi-branched model of the human arterial system. Med. Biol. Eng. Comput. 18, 709–718 (1980)
Zhang, X., Noda, S., Himeno, R., et al.: Cardiovascular disease-induced thermal responses during passive heat stress: an integrated computational study. Int. J. Numer. Method Biomed. Eng. 32, e02768 (2016). doi:10.1002/cnm.2768
Westerhof, N., Lankhaar, J.W., Westerhof, B.E.: The arterial windkessel. Med. Biol. Eng. Comput. 47, 131–141 (2009)
De Pater, L., Van den Berg, J.W.: An electrical analogue of the entire human circulatory system. Med. Electron. Biol. Eng. 2, 161–166 (1964)
Snyder, M.F., Rideout, V.C.: Computer simulation studies of the venous circulation. IEEE Trans. Biomed. Eng. 4, 325–334 (1969)
Sun, Y., Beshara, M., Lucariello, R.J., et al.: A comprehensive model for right-left heart interaction under the influence of pericardium and baroreflex. Am. J. Physiol. Heart Circ. Physiol. 272, H1499–H1515 (1997)
Heldt, T., Shim, E.B., Kamm, R.D., et al.: Computational modeling of cardiovascular response to orthostatic stress. J. Appl. Physiol. 92, 1239–1254 (2002)
Olufsen, M.S., Peskin, C.S., Kim, W.Y., et al.: Numerical simulation and experimental validation of blood flow in arteries with structured-tree outflow conditions. Ann. Biomed. Eng. 28, 1281–1299 (2000)
Sherwin, S.J., Franke, V., Peiró, J., et al.: One-dimensional modelling of a vascular network in space-time variables. J. Eng. Math. 47, 217–250 (2003)
Formaggia, L., Lamponi, D., Tuveri, M., et al.: Numerical modeling of 1D arterial networks coupled with a lumped parameters description of the heart. Comput. Methods Biomech. Biomed. Eng. 9, 273–288 (2006)
Montecinos, G.I., Müller, L.O., Toro, E.F.: Hyperbolic reformulation of a 1D viscoelastic blood flow model and ADER finite volume schemes. J. Comput. Phys. 266, 101–123 (2014)
Ho, H., Mithraratne, K., Hunter, P.: Numerical simulation of blood flow in an anatomically-accurate cerebral venous tree. IEEE Trans. Med. Imaging 32, 85–91 (2013)
Shapiro, A.H.: Steady flow in collapsible tubes. J. Biomech. Eng. 99, 126–147 (1977)
Morgan, P., Parker, K.H.: A mathematical model of flow through a collapsible tube–I. Model and steady flow results. J. Biomech. 22, 1263–1270 (1989)
Pedley, T.J., Luo, X.Y.: Modelling flow and oscillations in collapsible tubes. Theor. Comput. Fluid Dyn. 10, 277–294 (1998)
Bassez, S., Flaud, P., Chauveau, M.: Modeling of the deformation of flexible tubes using a single law: application to veins of the lower limb in man. J. Biomech. Eng. 123, 58–65 (2011)
Marchandise, E., Flaud, P.: Accurate modelling of unsteady flows in collapsible tubes. Comput. Methods Biomech. Biomed. Eng. 13, 279–290 (2010)
Fullana, J., Zaleski, S.: A branched one-dimensional model of vessel networks. J. Fluid. Mech. 621, 183–204 (2009)
Müller, L.O., Toro, E.F.: A global multiscale mathematical model for the human circulation with emphasis on the venous system. Int. J. Numer. Method Biomed. Eng. 30, 681–725 (2014)
Olufsen, M.S., Ottesen, J.T., Tran, H.T., et al.: Blood pressure and blood flow variation during postural change from sitting to standing: model development and validation. J. Appl. Physiol. 99, 1523–1537 (2005)
van Heusden, K., Gisolf, J., Stok, W.J., et al.: Mathematical modeling of gravitational effects on the circulation: importance of the time course of venous pooling and blood volume changes in the lungs. Am. J. Physiol. Heart Circ. Physiol. 291, H2152–H2165 (2006)
Lim, E., Chan, G.S., Dokos, S., et al.: A cardiovascular mathematical model of graded head-up tilt. PLoS ONE 8, e77357 (2013)
Stergiopulos, N., Young, D.F., Rogge, T.R.: Computer simulation of arterial flow with applications to arterial and aortic stenoses. J. Biomech. 25, 1477–1488 (1992)
Wild, R., Pedley, T.J., Riley, D.S.: Viscous flow in collapsible tubes of slowly varying elliptical cross-section. J. Fluid Mech. 81, 273–294 (1977)
Mynard, J.P.: Computer modelling and wave intensity analysis of perinatal cardiovascular function and dysfunction. [Ph.D. Thesis], Murdoch Childrens Research Institute, Australia (2011)
Siviglia, A., Toffolon, M.: Steady analysis of transcritical flows in collapsible tubes with discontinuous mechanical properties: implications for arteries and veins. J. Fluid Mech. 736, 195–215 (2013)
Wang, J.J., Parker, K.H.: Wave propagation in a model of the arterial circulation. J. Biomech. 37, 457–470 (2004)
Mynard, J.P., Davidson, M.R., Penny, D.J., et al.: A simple, versatile valve model for use in lumped parameter and one-dimensional cardiovascular models. Int. J. Numer. Method Biomed. Eng. 28, 626–641 (2012)
Sun, Y., Sjoberg, B.J., Ask, P., et al.: Mathematical model that characterizes transmitral and pulmonary venous flow velocity patterns. Am. J. Physiol. Heart Circ. Physiol. 268, H476–H489 (1995)
He, Y., Liu, H., Himeno, R.: A one-dimensional thermo-fluid model of blood circulation in the human upper limb. Int. J. Heat Mass Transf. 47, 2735–2745 (2004)
Smith, N.P., Pullan, A.J., Hunter, P.J.: An anatomically based model of transient coronary blood flow in the heart. SIAM J. Appl. Math. 62, 990–1018 (2002)
Harmon Jr., J.V., Edwards, W.D.: Venous valves in subclavian and internal jugular veins. Frequency, position, and structure in 100 autopsy cases. Am. J. Cardiovasc. Pathol. 1, 51–54 (1987)
Nishihara, J., Takeuchi, Y., Miyake, M., et al.: Distribution and morphology of valves in the human external jugular vein: indications for utilization in microvascular anastomosis. Int. J. Oral. Maxillofac. Surg. 54, 879–882 (1996)
Chou, C.H., Chao, A.C., Hu, H.H.: Ultrasonographic evaluation of vertebral venous valves. AJNR Am. J. Neuroradiol. 23, 1418–1420 (2002)
Celepci, H., Brenner, E.: Position of valves within the subclavian and axillary veins. J. Vasc. Surg. 54, 70S–76S (2011)
Iimura, A., Nakamura, Y., Itoh, M.: Anatomical study of distribution of valves of the cutaneous veins of adult’s limbs. Ann. Anat. 185, 91–95 (2003)
Baptista-Silva, J.C.C., Dias, A.L., Cricenti, S.V., et al.: Anatomy of the basilic vein in the arm and its importance for surgery. Braz. J. Morphol. Sci. 20, 171–175 (2003)
Santos, C.A.S.D., Figueiredo, L.F.P.D., Gusmão, L.C.B.D., et al.: Valves of the common brachial vein: anatomical study. J. Vasc. Br. 6, 35–41 (2007)
Kiray, A., Ergur, I., Tayefi, H., at al.: Anatomical evaluation of the superficial veins of the upper extremity as graft donor source in microvascular reconstructions: a cadaveric study. Acta Orthop. Traumatol. Turc. 47, 405–410 (2013)
Thiranagama, R., Chamberlain, A.T., Wood, B.A.: Valves in superficial limb veins of humans and nonhuman primates. Clin. Anat. 2, 135–145 (1989)
Mühlberger, D., Morandini, L., Brenner, E.: An anatomical study of femoral vein valves near the saphenofemoral junction. J. Vasc. Surg. 48, 994–999 (2008)
Schweighofer, G., Mühlberger, D., Brenner, E.: The anatomy of the small saphenous vein: fascial and neural relations, saphenofemoral junction, and valves. J. Vasc. Surg. 51, 982–989 (2010)
Moore, H.M., Gohel, M., Davies, A.H.: Number and location of venous valves within the popliteal and femoral veins-a review of the literature. J. Anat. 219, 439–443 (2011)
Gottlob, R., May, R.: Venous Valves: Morphology, Function, Radiology. Surgery. Springer, New York (2012)
Franklin, K.J.: Valves in veins: an historical survey. J. R. Soc. Med. 21, 1–33 (1927)
Reymond, P., Merenda, F., Perren, F., et al.: Validation of a one-dimensional model of the systemic arterial tree. Am. J. Physiol. Heart Circ. Physiol. 297, H208–H222 (2009)
Wexler, L., Bergel, D.H., Gabe, I.T., et al.: Velocity of blood flow in normal human venae cavae. Circ. Res. 23, 349–359 (1968)
Ochsner, A., Colp, R., Burch, G.E.: Normal blood pressure in the superficial venous system of man at rest in the supine position. Circulation 3, 674–680 (1951)
Meissner, M.H.: Lower extremity venous anatomy. Semin. Interv. Radiol. 22, 147–156 (2005). doi:10.1055/s-2005-921948
Groothuis, J.T., Poelkens, F., Wouters, C.W., et al.: Leg intravenous pressure during head-up tilt. J. Appl. Physiol. 105, 811–815 (2008)
Gisolf, J., Van Lieshout, J.J., Van Heusden, K., et al.: Human cerebral venous outflow pathway depends on posture and central venous pressure. J. Physiol. 560, 317–327 (2004)
Keijsers, J.M.T., Leguy, C.A.D., Huberts, W., et al.: A 1D pulse wave propagation model of the hemodynamics of calf muscle pump function. Int. J. Numer. Method Biomed. Eng. 31, e02716 (2015)
Acknowledgements
Hao Liu was partly supported by a Grant-in-Aid for Scientific Research (Grant 17300141), Japan Society for the Promotion of Science and Research and Development of the Next Generation Integrated Simulation of Living Matter, JST, a part of the Development and Use of the Next Generation Supercomputer Project of the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan. XZ acknowledges the RIKEN Junior Research Associate Program.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A: Cardiopulmonary model
Modeling of cardiac chambers
The heart is modeled as four chambers with cardiac contraction represented by a time-varying elastance E(t) [15],
where \(E_{\mathrm {A}}\) denotes the elastance amplitude, \(E_{\mathrm {B}}\) the elastance baseline, and e(t) the normalized time-varying elastance. The term e(t) can be further expressed for ventricles as
and for atria as
where \(t_{\mathrm {0}}\) represents the time period of a cardiac cycle (s), \(t_{\mathrm {vcp}}, t_{\mathrm {acp}}, t_{\mathrm {vrp}}\), and \(t_{\mathrm {arp}}\) the durations of ventricular/atrial contraction/relaxation (s), respectively, and \(t_{\mathrm {ac}}\) and \(t_{\mathrm {ar}}\) the time points at which the atria begin to contract and relax (s). The cardiac chamber pressure can be further obtained as
where V is the cardiac chamber volume, \(V_{\mathrm {0}}\) the dead chamber volume, and S the viscoelasticity coefficient of the cardiac wall.
Modeling of pulmonary circulation
Pulmonary circulation is modeled with three compartments: pulmonary artery, the pulmonary capillary, and the pulmonary vein. The elastance (E) of pulmonary circulation is defined by [21]
where \(E_{\mathrm {0}}\) denotes the zero volume elastance, V the blood volume, and \(\phi \) the volume constant. Pulmonary pressure is then obtained as
Appendix B: 0D–1D coupling algorithm
Here we present two numerical methods associated with coupling of a 1D vascular network and 0D lumped-parameter models. As depicted in Fig. 2, the 1D vessels that supply blood to the 0D lumped-parameter models are defined as feeding vessels and include terminal arteries, superior vena cava, and inferior vena cava, whereas the 1D vessels that collect blood from the 0D lumped-parameter models are called draining vessels and consist of ascending aorta and terminal veins.
Characteristic method
With respect to this method, the characteristic variants are used to set boundary conditions. The 1D governing equations may be transformed into a set of characteristic equations and, when the viscous resistance is ignored, can be reformulated as
with
where \(W_{f,b} =u\pm \int \limits _{A_{0} }^A {\frac{c(\tau )}{\tau }} \mathrm{d}\tau \) expresses the characteristic invariants of the system, and \(\lambda _{f,b} =u\pm c\) denotes the propagation speeds of the characteristic invariants. The characteristic invariants \(W_{f,b} (t^{n+1})\) at the time \(n+1\) can be updated through an extrapolation from \(W_{f,b} (t^{n})\) at the previous time step n.
For the first node of the draining vessel, the characteristic invariant at the next time step \((n+1)\) can be updated as
and for the last node of the feeding vessel, we have
where \({x}_{\mathrm {1}}\) denotes the first node, \({x}_{\mathrm {m}}\) the last node, and \(\Delta {t}\) the time step. A flowchart of the characteristic variable method for 0D–1D coupling is described in detail in Fig. B1a. The limitation of this method is that it is only valid under the subcritical condition (\(S<1\)).
Ghost-point method
For this method, a so-called ghost point is used to set boundary conditions. At the interface connecting the 0D and 1D models, we assume that the index of the last gridpoint of the vessel is m, and the index of the first gridpoint of the vessel is 1. Then the continuity equation at the ghost point \({m}-1/2\) of the feeding vessel can be discretized such that
Similarly, the continuity equation at the ghost point 1/2 of the draining vessel can be obtained as
Thus the flow rate \({Q}^{n+1}\) can be expressed in terms of \({A}^{n+1}\):
A schematic description of the flowchart associated with the ghost-point method for the 0D–1D coupling is also given in Fig. B1b.
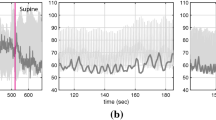
Figure B2 shows a comparison of the simulated pressures using the two methods. Obviously, there is almost no visible difference between the characteristic and ghost-point. It should be pointed out that the characteristic method is merely valid in the subcritical state, whereas the ghost-point method can be applied equally to the supercritical state as well, which makes it more robust and, hence, effective for 0D–1D coupling. Therefore, we used the ghost-point method to couple the 1D vascular network model with the 0D lumped-parameter model to close up the multiscale hemodynamic model for the entire CVS.
Rights and permissions
About this article
Cite this article
Zhang, X., Noda, S., Himeno, R. et al. Gravitational effects on global hemodynamics in different postures: A closed-loop multiscale mathematical analysis. Acta Mech. Sin. 33, 595–618 (2017). https://doi.org/10.1007/s10409-016-0621-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10409-016-0621-z