Abstract
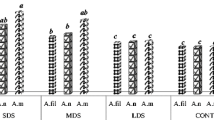
The effect of drought stress on total phenols, hydroxycinnamic acids, and antioxidant capacity in Cebú, DK2027, and DK2034 maize hybrids was evaluated. Compared to irrigation treatment, total phenols decreased in drought stress treatment by 35.34%, 5.59%, and 31.57% in Cebú, DK2027, and DK2034, respectively. In addition, the levels of t-ferulic, c-ferulic and p-coumaric acids decreased by 17.74%, 23.93%, and 29.83% in Cebú, by 8.92%, 8.62%, and 24.03% in DK2027, and by 13.66%, 11.03%, and 10.38% in DK2034, respectively. The predominant hydroxycinnamic acid from bran extracts was t-ferulic (67% to 72%), followed by c-ferulic (20% to 21%), and p-coumaric (7% to 11%) for both irrigation and drought stress treatments, while caffeic and sinapic acids were found in traces or were not detected. The antioxidant capacity decreased by 8%, 3%, and 9% in trolox equivalents and 7%, 17%, and 14% in percentage of radical scavenging activity for Cebú, DK2027, and DK2034 genotypes, respectively. The reduction of total phenols, hydroxycinnamic acids, and antioxidant capacity is due to the radical scavenging and photoprotective actions of hydroxycinnamic acids against highly reactive oxygen species generated under drought stress treatment.
Zusammenfassung
Die Wirkung von Trockenstress auf Gesamtphenole, Hydroxyzimtsäuren und die antioxidative Kapazität in Cebú‑, DK2027- und DK2034-Maishybriden wurde untersucht. Verglichen mit dem Bewässerungsregime sanken während der Trockenstressbehandlung die Gesamtphenole um 35,34 %, 5,59 % bzw. 31,57 % in Cebú, DK2027 und DK2034. Darüber hinaus sank der Gehalt an t-Ferulasäure, c-Ferulasäure und p-Cumarsäure um 17,74 %, 23,93 % bzw. 29,83 % in Cebú, um 8,92 %, 8,62 % und 24,03 % in DK2027 und um 13,66 %, 11,03 % bzw. 10,38 % in DK2034. Die vorherrschende Hydroxyzimtsäure aus Kleieextrakten war im Bewässerungs- und Trockenstressregime t-Ferulasäure (67 %–72 %), gefolgt von c-Ferulasäure (20 %–21 %) und p-Cumarsäure (7 %–11 %). Kaffee- und Sinapinsäure wurden nur in Spuren oder überhaupt nicht gefunden. Die antioxidative Kapazität verringerte sich um 8 %, 3 % und 9 % gemessen in Trolox-Äquivalenten und um 7 %, 17 % und 14 % gemessen als prozentualer Anteil der radikalfangenden Aktivität für Cebú‑, DK2027- und DK2034-Genotypen. Die Verringerung der Gesamtphenole, der Hydroxyzimtsäuren und der antioxidativen Kapazität ist zurückzuführen auf die radikalfangenden und photoprotektiven Wirkungen der Hydroxyzimtsäuren gegen reaktive Sauerstoffspezies, die unter Trockenstress entstehen.



Similar content being viewed by others
References
Ali Q, Ashraf M (2011) Induction of drought tolerance in maize (Zea mays L.) due to exogenous application of trehalose: growth, photosynthesis, water relations and oxidative defense mechanism. J Agron Crop Sci 197(4):258–271
Chen JH, Ho CT (1997) Antioxidant activities of caffeic acid and its related hydroxycinnamic acid compounds. J Agric Food Chem 45(7):2374–2378
Chun OK, Kim DO (2004) Consideration on equivalent chemicals in total phenolic assay of chlorogenic acid-rich plums. Food Res Int 37(4):337–342
Farooq M, Wahid A, Kobayashi N, Fujita D, Basra SMA (2009) Plant drought stress: effects, mechanisms and management. Agron Sustain Dev 29(1):185–212
García-Plazaola JI, Becerril JM (2000) Effects of drought on photoprotective mechanisms in European beech (Fagus sylvatica L.) seedlings from different provenances. Trees (Berl West) 14(8):485–490
Guo J, Zhang J, Wang W, Liu T, Xin Z (2015) Isolation and identification of bound compounds from corn bran and their antioxidant and angiotensin I‑converting enzyme inhibitory activities. Eur Food Res Technol 241(1):37–47
Huang D, Ou B, Prior RL (2005) The chemistry behind antioxidant capacity assays. J Agric Food Chem 53(6):1841–1856
Hura T, Hura K, Grzesiak S (2008) Contents of total phenolics and ferulic acid, and PAL activity during water potential changes in leaves of maize single-cross hybrids of different drought tolerance. J Agron Crop Sci 194(2):104–112
Inglett GE, Chen D (2011) Antioxidant activity and phenolic content of air-classified corn bran. Cereal Chem J 88(1):36–40
Kanski J, Aksenova M, Stoyanova A, Butterfield DA (2002) Ferulic acid antioxidant protection against hydroxyl and peroxyl radical oxidation in synaptosomal and neuronal cell culture systems in vitro: structure–activity studies. J Nutr Biochem 13(5):273–281
Kim KH, Tsao R, Yang R, Cui SW (2006) Phenolic acid profiles and antioxidant activities of wheat bran extracts and the effect of hydrolysis conditions. Food Chem 95(3):466–473
Manach C, Scalbert A, Morand C, Rémésy C, Jiménez L (2004) Polyphenols: food sources and bioavailability. Am J Clin Nutr 79(5):727–747
Minitab 14 statistical software (2004) Computer software. Minitab, State College (www.minitab.com)
Naczk M, Shahidi F (2004) Extraction and analysis of phenolics in food. J Chromatogr A 1054(1–2):95–111
Prior RL, Wu X, Schaich K (2005) Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J Agric Food Chem 53(10):4290–4302
Servicio de Información Agroalimentaria y Pesquera [Food and Fisheries Statistics Service] (SIAP) (2017) Anuario Estadístico de la Producción Agrícola. 2017. http://www.siap.gob.mx/cierre-de-la-produccion-agricola-por-cultivo/. Accessed 11.12.2017
Sharma P, Jha AB, Dubey RS, Pessarakli M (2012) Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J Bot 2012:1–26 (Article ID 217037)
Srinivasan M, Sudheer AR, Menon VP (2007) Ferulic acid: therapeutic potential through its antioxidant property. J Clin Biochem Nutr 40(2):92–100
Teixeira J, Gaspar A, Garrido EM, Garrido J, Borges F (2013) Hydroxycinnamic acid antioxidants: an electrochemical overview. Biomed Res Int 2013:1–11 (Article ID 251754)
Thaipong K, Boonprakob U, Crosby K, Cisneros-Zevallos L, Byrne DH (2006) Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J Food Compost Anal 19(6–7):669–675
Vitaglione P, Napolitano A, Fogliano V (2008) Cereal dietary fibre: a natural functional ingredient to deliver phenolic compounds into the gut. Trends Food Sci Technol 19(9):451–463
Vuletić M, Hadži-Tašković Šukalović V, Marković K, Kravić N, Vučinić Ž, Maksimović V (2014) Differential response of antioxidative systems of maize (Zea mays L.) roots cell walls to osmotic and heavy metal stress. Plant Biol (Stuttg) 16(1):88–96
Zhao YX, Ding MY, Liu DL (2005a) Phenolic acids analysis in Ligusticum chuanxiong using HPLC. J Chromatogr Sci 43(8):389–393
Zhao Z, Egashira Y, Sanada H (2005b) Phenolic antioxidants richly contained in corn bran are slightly bioavailable in rats. J Agric Food Chem 53(12):5030–5035
Acknowledgements
Authors would like to acknowledge the financial support for this research provided by Consejo Nacional de Ciencia y Tecnología (CONACYT) through Investigación Científica Básica SEP-CONACYT 169635 granted to Guillermo Niño-Medina and for the scholarship granted to Eleazar Lugo-Cruz. Thanks to Monsanto Company for the maize hybrids seeds provided.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
E. Lugo-Cruz, F. Zavala-García, H. Rodríguez-Fuentes, V. Urías-Orona, J.A. Vidales-Contreras, R. Carranza-De La Rosa, and G. Niño-Medina declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Lugo-Cruz, E., Zavala-García, F., Rodríguez-Fuentes, H. et al. The Effect of Drought Stress on Nutraceutical Properties of Zea mays Bran. Gesunde Pflanzen 70, 179–184 (2018). https://doi.org/10.1007/s10343-018-0429-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10343-018-0429-9