Abstract
In 2017, a clinical disease outbreak resulted in substantial mortality of adults and larvae of cultured Chinese rice-field eels (Monopterus albus) on a farm in Hubei, Central China. A rhabdovirus was isolated from moribund specimens, and typical clinical symptoms associated with an outbreak included an enlarged and swollen head. This differed from previous observations. Histological changes included necrosis and cavities of various sizes within the brain and kidney. Homogenized tissues of diseased Chinese rice-field eels were screened for viral isolation using six different fish cell lines. A rhabdovirus was isolated following observation of cytopathic effect (CPE) in a gibel carp brain (GiCB) cell line and confirmed by RT-PCR. Electron microscopy showed large numbers of rhabdovirus-shaped particles in the cytoplasm of the brain cells of the diseased Chinese rice-field eels and in the infected GiCB cell line. This virus has been named “Chinese rice-field eel rhabdovirus” (CrERV), and the complete nucleotide sequence of CrERV was cloned. This rhabdovirus is composed of 11,545 nucleotides with the following genomic organization: 3′-N-P-M-G-L-5′. The genes are separated by conserved gene junctions, and phylogenetic analysis of the L sequence revealed that CrERV forms a separate branch with Siniperca chuatsi rhabdovirus (SCRV) and hybrid snakehead rhabdovirus C1207 (HSHRV-C1207). This is the first report of the complete sequence of CrERV from the Chinese rice-field eel in China.
Similar content being viewed by others
Introduction
The Chinese rice-field eel, Monopterus albus (family, Synbranchidae; order, Synbranchiformes), is a major economic freshwater species in China and other Asian countries [1]. It can breathe air and crawl over land [2]. The Chinese rice-field eel has recently become a target species for aquaculture in China due to its rapid growth and adaptability to cage-culture conditions [3].
Previous studies on Chinese rice-field eels have focused on sex reversal characteristics [4], artificial rearing techniques for larvae [5], and dietary nutritional requirements [6]. However, production of larvae and adults remains un-industrialized, and artificial rearing of Chinese rice-field eels has been impacted by mortality events due to unknown causes [7]. Nutrition, breeding density, and water quality contribute to the low survival rates of the Chinese rice-field eel larvae [7, 8]. Moreover, previous studies have shown that parasitic, bacterial, and viral infections can cause serious losses in adult eels under intensive culture conditions [9,10,11]. In China, specific diseases that affect both Chinese rice-field eel larvae and adults are yet to be reported.
In the summer of 2017, a farm that produces Chinese rice-field eels in Hubei, Central China, reported a massive mortality (90–100%) event that resulted in significant economic losses. Despite dietary changes and administration of various treatments, the mortality rate remained high. Examination of Chinese rice-field eels showed limited evidence of parasitic or bacterial disease, and therefore samples of moribund eels were screened for viral isolation. It was determined that a rhabdovirus was the causative agent of the clinical disease outbreaks following isolation in cell culture, electron microscopy, PCR, and genomic characterization. To analyze the molecular characteristics of the Chinese rice-field eel rhabdovirus (CrERV), whole-genome analysis was completed, and sequences were compared to those of other known rhabdoviruses. This study identifies a new rhabdovirus and provides a foundation for further studies on the prevention and treatment of this new viral disease.
Materials and methods
Specimen collection
Diseased Chinese rice-field eels (body length: 4 ± 0.5 cm [larvae], 35 ± 1.0 cm [adults]; weight: 0.3 ± 0.05 g [larvae], 67.5 ± 2.0 g [adults]) with a swollen head syndrome were collected from a Chinese rice-field eel cage-culture farm located in Qianjiang, Hubei Province, China. The diseased specimens (approximately 40) were transferred to the laboratory for pathogen diagnosis. For the infection experiment, 60 healthy Chinese rice-field eels (length, 13–15 cm), which were confirmed to be free of CrERV by RT-PCR, were randomly divided into experimental and control groups (30 eels in each group). The eels were acclimated in tanks (4.5 × 103 l) with flow-through aerated water (municipal water) at 25 °C, and water quality was monitored daily. Eels were fed regularly with a commercial diet (extruded feed, Tongle, China) for 2 weeks prior to the infection experiment.
Parasitology and bacteriology
The exterior mucus, skin, and whole body of the diseased eels (approximately 10) were sampled for parasites and screened using an optical microscope (DM2500; Leica, Germany). For bacterial identification, the brain, liver, spleen, and kidney tissues of the eels with the typical swollen head syndrome were aseptically collected and plated on appropriate bacterial agar medium (Brain Heart Infusion Broth,BHI), and PCR was performed using the primers for 16S rRNA as described previously [12].
Histopathological analysis
The brain, liver, spleen, spinal cord, and kidney tissues of the diseased eels were collected and fixed in 4% paraformaldehyde, dehydrated, and embedded in paraffin. Sections from paraffin blocks were cut at 5-µm, and slides were stained with hematoxylin and eosin using standard procedures. Slides were examined for histopathological changes using a light microscope (DM2500; Leica, Germany).
Cell culture and virus isolation
For virus isolation, six fish cell lines were used, namely, epithelioma papilloma cyprinid (EPC), grass carp ovary (GCO), channel catfish kidney (CCK), rainbow trout gonadal (RTG-2), grass carp kidney (CIK) and gibel carp brain (GiCB) cell lines. The cells were grown in Dulbecco’s modified Eagle’s medium (DMEM) with 10% fetal bovine serum at 20 °C (RTG-2), 25 °C (EPC, GCO, GiCB, and CCK), or 28 °C (CIK). The tissue samples were homogenized in Dulbecco’s phosphate-buffered saline (DPBS), frozen and thawed three times at -80 °C, and then clarified by centrifugation at 4000 × g for 20 min (Sigma-3K15, USA) at 4 °C. The supernatant was filtered (0.22-µm filter; Nalgene, USA) and inoculated onto confluent monolayers of the six cell lines at 1:10, 1:100, and 1:1000 dilutions in 24-well tissue culture plates. The same volume of DMEM was used for the negative mock-infected control, and DMEM was added to each of the six cell lines. The inoculated cells were incubated at 20 °C (RTG-2), 25 °C (EPC, GCO, GiCB, and CCK), or 28 °C (CIK) and checked daily using a microscope. For cells that showed a cytopathic effect (CPE), supernatant was collected and RNA was extracted for detection of CrERV by RT-PCR. If CPE was not observed, the cultures were used for further passages; when three blind passages showed no CPE, the samples were considered negative for the virus.
Electron microscopy
Brain, liver, spleen, and kidney tissues from the diseased Chinese rice-field eels and infected cells were fixed overnight in 2.5% glutaraldehyde as described previously [12]. Samples were then post-fixed in 1% osmium tetroxide for 1.5 h, washed with PBS, dehydrated in graded ethanol, and embedded in epoxy resin (Epon 812, USA). Ultra-thin sections were prepared using a microtome (Leica UC, Germany), stained with 2% uranyl acetate and lead citrate, and examined using a transmission electron microscope (Hitachi-7650, Japan).
Full-length viral genome sequencing
RNA was extracted from purified virus in TRIzol Reagent (Invitrogen) according to the manufacturer’s protocol. Reverse transcription was performed using an ImProm-IITM Reverse Transcription System (TaKaRa, Japan). A PCR primer pair (CrERV-F/R) was designed based on previously published nucleoprotein gene sequences specific to viruses in the genus Perhabdovirus (Table 1), and RT-PCR was performed using a PCR amplification kit (TaKaRa, Japan). The cDNA samples were used in the subsequent PCR (35 cycles of 95 °C for 5 min, 94 °C for 1 min, 58 °C for 1 min, and 72 °C for 1 min, followed by extension at 72 °C for 10 min). A 833-bp PCR product was amplified.
For the diseased eels, PCR was performed using nine groups of primers (CrERV-F1/R1–CrERV-F9/R9; Table 1) designed to obtain the sequences of CrERV using an overlap method [13]. The 5′ and 3′ regions of the rhabdovirus in the diseased Chinese rice-field eels were identified by performing rapid amplification of cDNA ends (RACE) PCR using a Clontech SMART cDNA synthesis kit (TaKaRa, Japan). To extend the 5′ region of the cDNA sequence, 5′ RACE was performed using a gene-specific primer (CrERV-rR1–rR2; Table 1). 3′ RACE was performed using an oligo (dT) adaptor primer with a TaKaRa RNA PCR Kit (TaKaRa, Japan) (CrERV-rF1–rF2; Table 1). The PCR product was checked using agarose gel electrophoresis, and the target DNA fragment was purified using a Wizard® SV Gel and PCR Clean-up System (Promega, USA). The purified PCR products were ligated into the pMD 19-T vector (TaKaRa, Japan) at 4 °C overnight for cloning and sequencing. Virus was detected using RT-PCR and RACE with the primers listed in Table 1.
Phylogenetic analysis
Multiple rhabdovirus sequences were obtained from the GenBank database and compared to the sequences obtained in this study. The CrERV sequence was analyzed using the DNASTAR software package (Lasergene). Multiple alignments of the amino acid sequences were performed using Clustal W, and sequence identities were calculated using GeneDoc. A phylogenetic tree was constructed by the neighbor-joining method and evaluated using the Poisson correction model with 1000 bootstrap replicates in MEGA 5.0.
Infection experiment
To link viral isolation to disease causation, eels were challenged with live CrERV grown in GiCB cell cultures; 100 μl of live CrERV in supernatant was injected intraperitoneally into the eels (1.0 × 106 TCID50 ml−1 per eel). The control group was mock infected using DPBS (Sigma, USA). Clinical signs of infection and mortality were monitored over a period of 14 days post-challenge. Moribund or dead fish in the experimental group and healthy fish in the control group were randomly sampled (20% of daily mortality was sampled and tested) and tested for the presence of the rhabdovirus by RT-PCR, and re-isolation of the virus via cell culture was attempted to confirm the cause of death.
Results
Clinical signs and laboratory examination
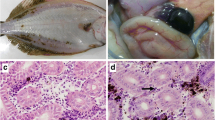
Behavioral signs of disease in larval and adult eels included gasping at the water surface, abnormal movement and loss of equilibrium, aberrant swimming, and anorexia (Fig. 1A). Clinical signs included an enlarged/swollen head that became more obvious as the disease progressed; hemorrhaging was also observed on the skin of the infected adults (Fig. 1B, C, and D). No pathogenic bacteria were isolated from clinically diseased eel samples, and microscopic examination of the skin revealed no parasites.
Gross pathological signs and histology of Chinese rice-field eels naturally infected with the rhabdovirus. (A) External appearance of an infected larva showing loss of equilibrium, disorganized swimming, and anorexia. (B, C) Swollen heads of the infected larvae. (D) Swollen head and hemorrhaging of the skin of an infected adult. (E, F) Widespread multifocal necrosis and various cavities in the infected brain (arrows). (G) Kidney section showing focal hemorrhage and cellular cavities in the infected eel (arrows). (H) Spinal cord section showing widespread multifocal necrosis (arrows)
Histopathological analysis
Various degrees of damage and necrosis consistent with viral infection were observed in the brain, kidney, and spinal cord of the affected eels. No histological abnormalities were observed in the corresponding organs from healthy individuals. The brain appeared most severely affected, showing several different levels of multifocal necrosis and various-sized cavities (Fig. 1E and F). The kidney was affected and showed severe focal hemorrhaging, and the kidney and spinal cord cells exhibited necrosis along with numerous cells with pyknotic and karyolytic nuclei (Fig. 1G and H).
Virus isolation
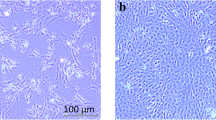
The tissue homogenate filtrate of the diseased Chinese rice-field eels was used for virus isolation from the EPC, GCO, CCK, RTG-2, CIK, and GiCB cell lines. The GiCB cell line was observed to be the most susceptible after three consecutive blind passages. After successive passages on the GiCB cell line, CPE became more consistent, and typical rhabdovirus CPE occurred as early as 72 h post-inoculation (Fig. 2A and B). The original virus showed moderate infectivity in the EPC cell line when compared with the GiCB cell line. The samples inoculated onto the GCO, CCK, RTG-2, and CIK cell lines did not show CPE.
Cytopathic effect caused by the rhabdovirus in the GiCB cell line at 3 days post-inoculation (dpi) and transmission electron microscopy of the brain of a naturally infected Chinese rice-field eel and GiCB cells after infection. (A) Control cell line. (B) Infected cell line. (C, D) Virus in the brain and kidney tissues of the infected eels. (E, F) Virus located in the GiCB cell line after showing typical CPE
Electron microscopy
Using electron microscopy, a large number of virus particles were located in the cytoplasm of brain and kidney cells from diseased eels. The characteristic bullet shape was characteristic of rhabdoviruses, and the size was approximately 60 × 120 nm (Fig. 2C and D). The thin sections confirmed the presence of similar viral particles in infected GiCB cells (Fig. 2E and F). Other virus-like particles were not observed in fish or in any cell lines.
Complete genome sequence
The genome sequence was determined, and a novel rhabdovirus was identified in Chinese rice-field eel larvae and adults. This virus has been named “Chinese rice-field eel rhabdovirus” (CrERV) (GenBank accession no: MH319839), and its genome is 11,545 bp in length. The basic genome of CrERV is composed of (from 3′-5′) a nucleoprotein (N), phosphoprotein (P), matrix protein (M), glycoprotein (G), and large major protein (L), similar to other members of the genera Perhabdovirus and Sprivivirus [14, 15] (Fig. 3A). The structural protein genes of CrERV are flanked by conserved regulatory gene junction sequences, and a 71-bp 3′ leader RNA region and a 61-bp 5′ trailer region are located at the 3′ and 5′ ends of the genome, respectively (untranslated regions at the 3′ and 5′ ends called “leader” and “trailer,” respectively). Almost all rhabdoviruses have five genes that encode the structural proteins (N, P, M, G, and L) (Fig. 3A).
Comparison of the genomic structure of the rhabdovirus in the Chinese rice-field eel and other animals and plants and analysis of gene complementarities in the CrERV genome. (A) A typical member of each genus is represented. Each arrow represents the position of a long open reading frame (ORF). Lines represent untranslated regions (UTRs). Other alternate ORFs occur in some genes; only ORFs (≥120 nucleotides) that appear likely to be expressed are shown. (B) Complementarities of the 3′ and 5′ ends of the CrERV genome. The first 10 nucleotides of the 3′ end are complementary to the 5′-end nucleotides of the genomic RNA
Genomic structure analysis
The N gene of CrERV extends from nucleotides 72 to 1527, with a single ORF of 1290 nucleotides that encodes a protein of 429 amino acids. The predicted molecular mass of the N protein is 47.7 kDa, and the calculated isoelectric point (pI) is 5.37. The N gene contains the highly conserved amino acid motif SPYSA, which is present at amino acid position 289–293. The start codon ATG is located 5 nucleotides upstream in CrERV.
The P gene is 971 nucleotides in length. Unlike other rhabdoviruses that contain alternate ORFs, such as Siniperca chuatsi rhabdovirus (SCRV) and vesicular stomatitis Indiana virus (VSIV), the P gene of CrERV has one large ORF (864 nucleotides) that encodes a protein of 287 amino acids with a calculated molecular mass of 31.6 kDa and a pI of 4.43. The P protein has a predicted potential tyrosine phosphorylation site and five potential serine/threonine phosphorylation sites. It also contains a 44-amino-acid proline-rich region extending from amino acid 158 to 202.
The putative M gene contains 946 nucleotides that encode a protein of 208 amino acids with a calculated molecular mass of 23.4 kDa and a pI of 7.81. A PPPY motif is present between amino acid 19 and 22, and the M protein has six potential serine/threonine phosphorylation sites.
The G gene consists of 1659 nucleotides and has a single ORF of 1527 nucleotides that encodes a protein of 508 amino acids with a calculated molecular mass of 56.3 kDa and a pI of 7.15. The G protein is predicted to contain a signal peptide sequence at amino acids 1–15 and a 22-amino-acid transmembrane region between amino acids 454 and 476. Fourteen cysteine residues are present, and the characteristics of the first 12 are comparable with those in perhabdovirus G proteins. The G protein of CrERV also contains three putative N-glycosylation sites at amino acids 203–206, 387–390, and 426–429.
The L gene is the last and largest transcriptional unit, extending from nucleotides 5118 to 11,484. It contains a single ORF of 6327 nucleotides flanked by 119 and 13 nucleotides of 5′ and 3′ untranslated regions, respectively, and it encodes a putative protein of 2078 amino acids. The calculated molecular mass is 237.7 kDa, and its pI is 7.92. A previous study has shown six conserved blocks (blocks I–VI) in the L proteins of members of the order Mononegavirales by multiple sequence alignment [16]. In our study, these conserved blocks were found to be located between amino acids 223 and 1701 of the CrERV L protein. Blocks II and III are the most conserved and contain five conserved motifs (pre-A–D) [16]. The features of each gene of CrERV are listed in Table 2.
All of the ORFs of CrERV are separated by gene junctions. The CrERV gene junction contains a transcription stop/polyadenylation signal, 3–4 untranscribed spacer nucleotides, and a transcription initiation signal (Table 3). The transcription initiation signal, AACAG, and termination/polyadenylation signal, TATG (A)7, of CrERV are relatively conserved. Except for the spacer sequence of the M–G junction (CTT), all of the untranscribed spacers are CTTT. The distances of the N, P, M, G, and L genes from the translation start codons and transcription initiation signals are 23, 5, 23, 7, and 5 nucleotides, respectively, and the distances from the transcription stop/polyadenylation and translation stop codons are 127, 86, 101, 108, and 108 nucleotides, respectively. The 3′- and 5′- terminal sequences of CrERV exhibit complementarities located at the first 10 nucleotides (Fig. 3B).
Phylogenetic analysis and classification
To determine the phylogenetic position of CrERV in the family Rhabdoviridae, a phylogenetic tree based on the L gene sequences of multiple rhabdoviruses from 18 genera was constructed (Fig. 4). Viruses belonging to all known species of fish rhabdoviruses clustered in three branches corresponding to the genera Novirhabdovirus, Perhabdovirus, and Sprivivirus. CrERV was most closely related to the newly isolated strains SCRV and hybrid snakehead rhabdovirus C1207 (HSHRV-C1207), which tended to cluster with members of the genus Perhabdovirus and differ greatly from the other rhabdovirus lineages. This suggests that CrERV may belong to the genus Perhabdovirus.
RT-PCR detection of rhabdovirus
RT-PCR amplification was performed using RNA extracted from three naturally infected Chinese rice-field eels, three artificially infected Chinese rice-field eels, and three passages of the virus isolated from GiCB cell lines. The tissues of the diseased eels and GiCB cell lines showing typical CPE post-inoculation yielded rhabdovirus-specific PCR products of approximately 833 bp (Fig. 5A).
RT-PCR results for CrERV in the Chinese rice-field eels and the GiCB cell line as well as the pathogenicity of CrERV to healthy Chinese rice-field eels. (A) Lanes 1–3, naturally infected group; Lanes 4–6, artificially infected group; Lanes 7–9, GiCB cell infected group; Lane 10, positive control, Lane 11, negative control; M, DL1000 DNA marker; (B) The rate of mortality of the Chinese rice-field eels post-challenge
Infection experiment
In the experimental group, mortality was observed on day 5 post-challenge, and it was characterized by a decrease in appetite and typical swelling of the head. Internally, petechial hemorrhaging was detected in the kidney. No mortality or clinical signs were observed in the control group. In contrast, the cumulative mortality reached 80% in the experimental group (Fig. 5B). Electron microscopy results demonstrated the presence of the virus, and the size and shape of the viral particles were similar to those observed in the naturally infected fish. The PCR results confirmed that the dead eels in the experimental group were positive for the rhabdovirus (data not shown).
Discussion
The family Rhabdoviridae is composed of 18 genera and 135 species of viruses with negative-sense RNA [17, 18]. Rhabdovirus virions are 100–430 nm in length and 45–100 nm in diameter, and the genomes are usually single-stranded RNA with partially complementary termini [19]. Almost all rhabdovirus genomes have five genes that encode the N, P, M, G, and L structural proteins [20]; however, numerous rhabdovirus genomes have additional genes in alternate ORFs or genes within the structural protein genes that encode other proteins (http://www.ictvonline.org/virusTaxonomy.asp). The family is ecologically diverse, with members that infect plants or animals (including mammals, birds, reptiles, and fish) [21]. Members of the genera Novirhabdovirus, Perhabdovirus, and Sprivivirus infect a wide range of fish species [22], and rhabdoviruses have been isolated from marine and freshwater fish species, such as rainbow trout [23], mandarin fish [13], eel [24], cyprinids [25], and snakehead [26, 27]. The distribution and movement of cultured fish can result in the spread of such viruses to other fish species in China and elsewhere. In this study, we isolated and identified a novel rhabdovirus from a Chinese rice-field eel collected in Hubei Province, China. Epidemiological investigations showed that this rhabdovirus causes a disease that generally occurs between March and May at a water temperature of 15–22 °C. When the water temperature is higher than 25 °C, mortality in eels is negligible (data not shown). Temperature-related mortality has also been associated with other rhabdoviruses. For example, the severity of disease due to rhabdovirus infection in the rainbow trout has been found to be temperature dependent, which suggests a critical role of water temperature for rhabdovirus infection in fish [28].
In our study, clinical signs, gross pathology, and histopathology of the diseased Chinese rice-field eels were comparable to those observed in a range of other fish species [29]. Externally, the main feature of the diseased fish was a swollen head. In moribund Chinese rice-field eels, this clinical sign was obvious, and electron microscopy revealed large rhabdovirus particles dispersed primarily in the cytoplasm of brain cells. Further examination showed that bacterial pathogens or parasites were not the cause of disease or head swelling. Based on clinical signs, lesions, and laboratory tests, we confirmed that CrERV was the causative agent of the disease at the Chinese rice-field eel farm in Qianjiang, Hubei Province, China.
In previous studies, many cell lines have been used to isolate rhabdoviruses; however, only a few are ideal for isolation and propagation of rhabdoviruses, such as fathead minnow, EPC, Chinese perch brain, and grass carp fin cell lines [29, 30]. In this study, GiCB was the most susceptible cell line and produced typical CPE as early as 3 days post-inoculation. Electron microscopy revealed the typical bullet-shaped morphology of rhabdoviruses, with virions ranging from 100 to 150 nm in length and 45 to 70 nm in diameter. The virus morphology and size were consistent with those of rhabdoviruses from other fish species [29, 30]. Moreover, the N gene of the rhabdovirus was detected beyond the sixth passage in the infected GiCB cell line.
To the best of our knowledge, this is the first report of a new rhabdovirus isolated from infected Chinese rice-field eels in China based on whole-genome sequencing. Previously, only the N, P, and G gene sequences of rhabdoviruses from Chinese rice-field eels were reported, and analysis of the N, P, and G gene sequences revealed a close relationship to members of the genus Vesiculovirus [11], which differs from the results presented here. We determined the complete genome sequence (11,545 nucleotides) of CrERV, and the gene organization of CrERV was found to be similar to that of rhabdoviruses in the genus Perhabdovirus. The transcription initiation signal, AACAG, and termination/polyadenylation signal, TATG (A)7, of CrERV are relatively conserved and similar to those found in SVCV and SCRV [13, 14]. The 3′ leader region of CrERV is 71 nucleotides, which is similar to that of SCRV but slightly shorter than that of HSHRV-C1207 (72 nucleotides) and longer than that of SHRV (55 nucleotides). The leader sequence of CrERV begins with ACG and is conserved in all known leader RNA sequences of the members of the genera Perhabdovirus, Lyssavirus, Ephemerovirus, and Vesiculovirus [27]. The 5′ trailer region of CrERV is 61 nucleotides long, which is similar to those of SCRV and HSHRV-C1207 but shorter than those of some novirhabdoviruses, such as IHNV (102 nucleotides), HIRRV (73 nucleotides), and longer than some members of the genus Sprivivirus, such as SVCV (19 nucleotides) [31].
The sequences of the N, G, and L proteins of CrERV were compared to those of other rhabdoviruses, and they shared the greatest similarity with members of the genus Perhabdovirus. The highest sequence similarity (94.5% identity) was found with proteins of SCRV, HSHRV, and HSHRV-C1207 [13, 26, 27]. CrERV is distinct from the members of the genus Novirhabdovirus, and it showed the least similarity to SHRV (10.1% sequence identity). Many rhabdoviruses have additional proteins. For example, novirhabdoviruses express a non-structural protein that is necessary for efficient replication and helps to evade the innate antiviral response of the finfish host [32]. Vesiculoviruses express two small proteins (C and C′) from an alternate ORF in the P gene [33]. There are also reports that, in several rhabdoviruses, P has a fundamental role in evading the innate antiviral response of the host [13, 18]. In our study, the P gene of CrERV was found to be 971 nucleotides long and the ORF was 864 nucleotides long, which is longer than that of SCRV and slightly shorter than those of HSHRV-C1207 and EVEX [13, 24, 27]. However, an alternate ORF was not found in the P gene of CrERV, which is inconsistent with what has been observed in SCRV, HSHRV-C1207, and EVEX, but similar to what has been reported for PRV [15]. These results differ from our findings, which may be due to the different host species and mechanisms for evading the host innate antiviral response.
Previously, rhabdoviruses were distinguished serologically and according to virion morphology; however, current characterization associated with species distinction relies on phylogenetic relatedness [18]. In this study, the L gene was used to elucidate the evolutionary relationship of CrERV within the family Rhabdoviridae due to the presence of broadly conserved domains and the rarity of genetic recombination, as well as the fact that the L gene is widely used to classify members of many virus species [13]. It was found that isolates obtained from the Chinese rice-field eel larvae formed a monophyletic lineage with SCRV and HSHRV-C1207 (bootstrap values, 100%). This lineage was genetically related (65%) to members of the genus Perhabdovirus, suggesting that CrERV, together with SCRV and HSHRV-C1207, may belong to the genus Perhabdovirus.
In conclusion, we have described the isolation and identification of CrERV from larval and adult Chinese rice-field eels for the first time. This virus is linked to outbreaks on farms in China, and it appears that water temperature plays a role in the disease severity of disease caused by CrERV. The complete genome sequence of CrERV shows that it is closely related to known SCRV and HSHRV-C1207 isolates. CrERV readily infects GiCB cells and induce obvious CPE. Infection trials showed that CrERV is virulent to Chinese rice-field eels, and based on the findings presented here we recommend that farmers work to improve husbandry by treating water sources and disinfecting eggs whenever possible. Future studies should focus on developing effective diagnostic methods and exploring control and prevention strategies, such as vaccine development, water treatment options, and egg disinfection protocols, to reduce the spread of this virus and limit its impact on farmers in China and worldwide.
References
Zhou R, Cheng H, Zhang Q, Guo Y, Richard RC, Terrence RT (2002) SRY-related genes in the genome of the rice field eel (Monopterus albus). Genet Sel Evo l 34:129–137
Jang S, Zhou F, Xia L, Zhao W, Cheng H, Zhou R (2006) Construction of a BAC library and identification of Dmrt1gene of the rice field eel, Monopterus albus. Biochem Biophys Res Commun 348:775–780
Xu QQ, Wan J, Pan TS, Zhou JW, Ai KT, Yang DQ (2015) Antiviral genes in the Asian swamp eel Monopterus albus: comparison of tissue expression and inducible expression of six interferon regulatory factors. Genes Genom 37:429–440
Yin SW, Liu Y (2010) Compositions and functions of the hatching froth from rice field eel (Monopterus albus, Zuiew). Fish Physiol Biochem 36:195–200
Zou JX (1996) Artificial propagation and embryonic development of Monopterus albus. Technol Inf 23:27–30 (in Chinese)
Zhou QB, Wu HD, Zhu CS, Yan XH (2011) Effects of dietary lipids on tissue fatty acids profile, growth and reproductive performance of female rice field eel (Monopterus albus). Fish Physiol Biochem 37:433–445
Qu GC (2003) Reasons for the large number of deaths in the early stage of stocking of Monopterus albus and the measures to be taken. Fishery Guide to be Rich 14:48–49
Hu BL (2003) Techniques of artificial propagation and seedling cultivation in Asian swamp eel. Fishery Guide Rich 17:31–32
Chai JY, Sohn WM, Na BK, Park JB, Jeoung HG, Hoang EH, Htoon TT, Tin HH (2015) Larval Gnathostoma spinigerum detected in Asian Swamp eels, Monopterus albus, purchased from a local market in Yangon, Myanmar. Korean J Parasitol 5:619–625
Shao JC, Yuan JF, Shen YL, Hu RX, Gu ZM (2016) First isolation and characterization of Edwardsiella tarda from diseased Asian swamp eel, Monopterus albus (Zuiew). Aquac Res 47:3684–3688
Ou T, Zhu RL, Chen ZY, Zhang QY (2013) Isolation and identification of a lethal rhabdovirus from farmed rice field eels Monopterus albus. Dis Aquat Organ 106:197–206
Xu J, Zeng LB, Zhang H, Zhou Y, Ma J, Fan YD (2013) Cyprinid herpesvirus 2 infection emerged in cultured gibel carp, Carassius auratus gibelio in China. Vet Microbiol 166:138–144
Tao JJ, Zhou GZ, Gui JF (2008) Genomic sequence of mandarin fish rhabdovirus with an unusual small non-transcriptional ORF. Virus Res 132:86–96
Bjorklund HV, Higman KH, Kurath G (1996) The glycoprotein genes and gene junctions of the fish rhabdoviruses spring viremia of carp virus and hirame rhabdovirus: analysis of relationships with other rhabdoviruses. Virus Res 42:65–80
Gadd T, Viljamaa DS, Holopainen R, Koski P, Jakava VM (2013) Characterization of perch rhabdovirus (PRV) in farmed grayling Thymallus thymallus. Dis Aquat Org 106:117–127
Poch O, Blumberg BM, Bougueleret L, Tordo N (1990) Sequence comparison of five polymerases (L proteins) of unsegmented negative-strand RNA viruses: theoretical assignment of functional domains. J Gen Virol 71:1153–1162
Wagner RR, Rose JK (1996) Rhabdoviridae: the viruses and their replication. Fields Virol 3:1221–1244
Walker PJ, Blasdell K, Calisher CH, Dietzgen RG, Kondo H, Kurath G, Longdon B, Stone DM, Tesh RB (2018) ICTV Virus Taxonomy Profile: Rhabdoviridae. J Gen Virol. https://doi.org/10.1099/jgv.0.001020
Assenberg R, Delmas O, Morin B, Graham SC, De Lamballerie X, Laubert C, Coutard B, Grimes JM, Neyts J (2010) Genomics and structure /function studies of Rhabdoviridae proteins involved in replication and transcription. Antiviral Res 87:149–161
Schütze H, Mundt E, Mettenleiter TC (1999) Complete genomic sequence of viral haemorrhagic septicemia virus, a fish rhabdovirus. Virus Gen 19:59–65
Ivanov I, Yabukarski F, Ruigrok RW, Jamin M (2011) Structural insights into the rhabdovirus transcription/replication complex. Virus Res 162:126–137
King AMQ, Adams MJ, Carstens EB, Lefkowitz EJ (2012) Virus Taxonomy: Classification and Nomenclature of Viruses. Refer. Res. Book News 4:1730–1756
Morzunov SP, Winton JR, Nichol ST (1995) The complete genome structure and phylogenetic relationship of infectious hematopoietic necrosis virus. Virus Res 38:175–192
Richard G, Steven B, Elsa A (2012) Complete genomic sequence and taxonomic position of eel virus European X (EVEX), a rhabdovirus of European eel. Virus Res 166:1–12
Padhi A, Verghese B (2012) Molecular evolutionary and epidemiological dynamics of a highly pathogenic fish rhabdovirus, the spring viremia of carp virus (SVCV). Vet Microbiol 156:54–63
Liu XD, Wen Y, Hu XQ, Wang WW, Liang XF, Li J, Vikram V, Lin L (2015) Breaking the host range: mandarin fish is susceptible to a vesiculovirus derived from snakehead fish. J Gen Virol 96:775–781
Zeng WW, Wang Q, Wang YY, Liu C, Liang HR, Fang X, Wu SQ (2013) Genomic characterization and taxonomic position of a rhabdovirus from a hybrid snakehead. Arch Virol 159:2469–2473
Sohrab A, Mehdi S, Karim M, Sara S, Hooman RH (2016) Isolation and identification of viral hemorrhagic septicemia virus (VHSV) from farmed rainbow trout (Oncorhynchus mykiss) in Iran. Acta Tropica 156:30–36
Fu XZ, Lin Q, Liang H, Liu L, Huang Z, Li N, Su J (2017) The biological features and genetic diversity of novel fish rhabdovirus isolates in China. Arch Virol 162:2829–2834
Zhang Q, Li Z (1999) Three viruses was observed in diseased Chinese perch Siniperca chuatsi. Chin Sci Bull 44:192–195
Ammayappan A, Vakharia VN (2009) Molecular characterization of the Great Lakes viral hemorrhagic septicemia virus (VHSV) isolate from USA. Virol J 6:171–187
Biacchesi S (2010) The reverse genetics applied to fish RNA viruses. Vet Res 42:12
Spiropoulou CF, Nichol ST (1993) A small highly basic protein is encoded in overlapping frame within the P gene of vesicular stomatitis virus. J Virol 67:3103–3110
Acknowledgments
This work was supported by the Central Public-Interest Scientific Institution Basal Research Fund, CAFS (grant numbers: No. 2018JBF01; NO. 2017HY-ZD1005) and the Science and Technology Support Program of Hubei Province (grant number: 2015BBA234).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
The study was performed in strict accordance with the Guide for the Care and Use of Laboratory Animals Monitoring Committee of Hubei Province, China, and the protocol was approved by the Committee on the Ethics of Animal Experiments at the Yangtze River Fisheries Research Institute, Chinese Academy of Fishery Sciences. The Chinese rice-field eels were euthanized for 20–30 min in 1 mg of MS-222 (Sigma) per ml to limit their suffering before tissue collection.
Additional information
Handling Editor: William G Dundon.
Rights and permissions
About this article
Cite this article
Liu, W., Fan, Y., Li, Z. et al. Isolation, identification, and classification of a novel rhabdovirus from diseased Chinese rice-field eels (Monopterus albus). Arch Virol 164, 105–116 (2019). https://doi.org/10.1007/s00705-018-4054-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-018-4054-9