Abstract
Purpose
To compare percutaneous balloon compression (PBC) and radiofrequency thermocoagulation (RFTC) for the treatment of trigeminal neuralgia.
Methods
This was a retrospective single-center analysis of data from 230 patients with trigeminal neuralgia who underwent 202 PBC (46%) and 234 RFTC (54%) from 2002 to 2019. Comparison of demographic data and trigeminal neuralgia characteristics between procedures as well as assessment of 1) initial pain relief by an improved Barrow Neurological Institute (BNI) pain intensity scale of I–III; 2) recurrence-free survival of patients with a follow-up of at least 6 months by Kaplan-Meier analysis; 3) risk factors for failed initial pain relief and recurrence-free survival by regression analysis; and 4) complications and adverse events.
Results
Initial pain relief was achieved in 353 (84.2%) procedures and showed no significant difference between PBC (83.7%) and RFTC (84.9%). Patients who suffered from multiple sclerosis (odds ratio 5.34) or had a higher preoperative BNI (odds ratio 2.01) showed a higher risk of not becoming pain free. Recurrence-free survival in 283 procedures was longer for PBC (44%) with 481 days compared to RFTC (56%) with 421 days (p=0.036) but without statistical significance. The only factors that showed a significant influence on longer recurrence-free survival rates were a postoperative BNI ≤ II (P=<0.0001) and a BNI facial numbness score ≥ 3 (p = 0.009). The complication rate of 22.2% as well as zero mortality showed no difference between the two procedures (p=0.162).
Conclusion
Both percutaneous interventions led to a comparable initial pain relief and recurrence-free survival with a low and comparable probability of complications. An individualized approach, considering the advantages and disadvantages of each intervention, should guide the decision-making process. Prospective comparative trials are urgently needed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Trigeminal neuralgia is a rare disease characterized by recurrent paroxysmal pain that lasts only for seconds to minutes in the distribution of one or multiple branches of the trigeminal nerve [3]. The International Headache Society classified trigeminal neuralgia based on the perceived cause into 1) classical trigeminal neuralgia due to a neurovascular conflict with morphological changes, 2) secondary trigeminal neuralgia due to another disease, e.g., tumor or multiple sclerosis, and 3) idiopathic trigeminal neuralgia with no or no compressive neurovascular conflict [18]. Another classification takes into account the phenotype of the disease, which differs between purely paroxysmal trigeminal neuralgia and trigeminal neuralgia with concomitant continuous pain [14]. First-line treatment consists of sodium channel blockers such as carbamazepine, followed by other anticonvulsive drugs and their combinations [3]. If drug therapy fails to relieve pain or is not tolerated by the patient, a plethora of surgical procedures may be offered. While microvascular decompression of the trigeminal nerve root should be the first choice in patients with classical and idiopathic trigeminal neuralgia, ablative procedures of the trigeminal ganglion have gained acceptance as minimally invasive techniques [4]. They may become useful in cases of idiopathic trigeminal neuralgia and secondary trigeminal neuralgia but also in classical trigeminal neuralgia if patients are not eligible for microvascular decompression or if microvascular decompression fails to relieve pain. Radiofrequency thermocoagulation (RFTC) and percutaneous balloon compression (PBC) are both relatively simple and well-established techniques that offer adequate pain relief combined with a good safety profile [11]. They mainly differ in the surgical method as well as in their specificity of nerve injury. While RFTC, popularized by Sweet and Wepsic in 1974, allows somatotopic nerve mapping and therefore selective lesioning of one or multiple divisions of the trigeminal nerve, it has to be performed while the patient is awake and cooperative [32]. In contrast, PBC, first described by Mullan and Lichtor in 1983, is less selective than RFTC but does not require an awake patient [25]. It is thought to selectively injure larger myelinated fibers and therefore is mainly useful in trigeminal neuralgia with pain in the ophthalmic division, as the small fibers of the corneal reflex may be spared [29]. While both techniques have already been evaluated alone or in comparison with other techniques, to the best of our knowledge, only a few studies have compared both methods with each other [15, 16, 26]. Therefore, we present our single-center experience comparing RFTC and PBC in patients with trigeminal neuralgia with regard to initial pain relief, recurrence-free survival and associated complications.
Methods
Patient cohort and study design
In this retrospective single-center study, we identified all patients who underwent PBC or RFTC of the trigeminal ganglion between January 2002 and December 2019 at the Medical University of Vienna to treat medically refractory trigeminal neuralgia. The diagnosis of trigeminal neuralgia was confirmed by reviewing the medical records and identifying typical neuralgia symptoms as defined by the International Headache Society [18]. Patients had to meet the following criteria for inclusion: 1) diagnosis of classical, idiopathic or secondary trigeminal neuralgia as defined by the European Academy of Neurology [3]; 2) age older than 18 years; and 3) complete medical record. Patients who had previous surgery for trigeminal neuralgia were included in the study. The study protocol was approved by the local ethics committee of the Medical University of Vienna (EK 1105/2020) and is in accordance with the Helsinki Declaration of Human Rights.
Surgical procedures
The decision of whether to use PBC or RFTC to treat trigeminal neuralgia was made individually by one of three treating surgeons (Fig. 1). Factors influencing this decision were: the trigeminal division involved, the possibility of cooperating with the patient and the success of prior treatments. In general, patients with ophthalmic nerve involvement and patients who could not cooperate during an operation were primarily selected for PBC, while RFTC was preferred in patients with mandibular and maxillary involvement. If possible, the type of procedure was changed for follow-up interventions in the event of non-existent or only short-lasting therapeutic success. All operations were performed in the operating room. In PBC and RFTC, the same percutaneous approach to the foramen ovale was used as described by Härtel [17]. Patients were placed supine with their head in a reverse occipitomental position. A C-arm was used to allow fluoroscopic guidance. All patients underwent basic monitoring by electrocardiography, oxygen saturation and blood pressure measurements. After the procedure, patients were discharged the following day.
Radiofrequency thermocoagulation
During RFTC, intravenous sedation and a nasopharyngeal airway were used. A 20-gauge standard radiofrequency lesioning needle with a straight 5 mm active tip (Radimed®) was inserted through the foramen ovale, positioning the needle tip in the trigeminal cistern as described by Tatli et al. [35]. Correct placement was confirmed by muscle contractions after motor stimulation (2 Hz, 0.1 ms). Having the patient responsive allowed fine needle adjustment according to the patients’ sensations during sensory stimulation (50 Hz, 1 ms). If paresthesia occurred in the affected division of the trigeminal nerve, patients received RFTC after the depth of anesthesia was adjusted. RFTC was performed at 70°C for 60 s. With the patient awake again, it was checked whether hypesthesia was present in the coagulated nerve division, which ended the procedure. If no hypesthesia was present, RFTC with the same settings was repeated once again after verifying the correct localization by stimulation. The needle was then removed, and the puncture side was held under pressure over five minutes.
Percutaneous balloon compression
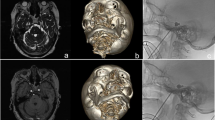
An awake and cooperative patient is not required during PBC and therefore, general anesthesia was used. A 14-gauge cannula was inserted through the cheek and advanced just through the foramen ovale. The inner stylet was then removed. A 4 French Fogarty® embolectomy catheter was passed through the cannula and positioned in the trigeminal cistern with its tip at the trigeminal pore (Fig. 2, A). The balloon was then inflated twice with 0.7 ml of contrast agent (50%) and physiological saline solution (50%) over 60 s. Fluoroscopy confirmed adequate placement and shape of the balloon (Fig. 2, B). After compression, the cannula and the balloon were removed, and the puncture site was held under pressure over 5 min.
Fluoroscopy-guided percutaneous balloon compression in lateral view. A) The tip of the cannula slightly enters the foramen ovale, while the deflated Fogarty® embolectomy catheter is positioned with its tip at the border of the clivus. B) When the balloon is inflated, it ideally adopts the shape of the trigeminal cistern and reaches a pear-like shape with its tip lying in the trigeminal pore
Data collection and outcome measures
Data collection was obtained through patient admission charts, operative notes, preoperative MRT, discharge letters and follow-up reports. The clinical status of the patients was assessed by a neurosurgeon upon admission, discharge and during follow-up examinations. Since follow-up examinations in the outpatient clinic were not carried out in a standardized manner, all patients were contacted and summoned to the outpatient clinic for a personal follow-up visit for the sake of this study. Demographic data, trigeminal neuralgia symptoms including onset of trigeminal neuralgia, duration of symptoms, age at surgery, pain distribution, International Headache Society classification of trigeminal neuralgia (classic, idiopathic, secondary) and category of trigeminal neuralgia pain (purely paroxysmal vs. concomitant continuous), previous treatment, antiepileptic drugs and the Barrow Neurological Institute (BNI) pain intensity scale were recorded [30]. The BNI pain intensity scale classifies facial pain into I) no pain, no medication; II) occasional pain, not requiring medication; III) some pain, adequately controlled with medication; IV) some pain, not adequately controlled with medication; and V) severe pain/no pain relief [30].
Complications during and side effects after the procedures, such as intraoperative bradycardia, hypesthesia, corneal anesthesia, masseter muscle weakness, and anesthesia dolorosa, were noted for all cases, including those interventions that had to be canceled due to an anesthesiologic or a technical problem.
Initial pain relief was defined as a BNI of I, II and III with a one-point improvement in the first 4 weeks after surgery. Initial pain relief was documented for all patients who had undergone a successful procedure.
Recurrence of trigeminal neuralgia was defined as an increase in BNI after initial pain relief at the operated side. Recurrence-free survival was only calculated if patients had a follow-up of at least 6 months.
Statistical analysis
Statistical calculations were carried out using GraphPad Prism version 9.1.2 (GraphPad Software, San Diego, California, USA) and R version 4.1.2 (R Foundation for Statistical Computing, Vienna, Austria). A two-sided p-value of < 0.05 was considered statistically significant. Continuous variables following a normal distribution were assessed by analysis of variance and Student’s t-test. Kruskal-Wallis and Mann-Whitney U tests were used for nonparametric interval-scaled variables. Chi-square or Fisher’s exact tests were used for categorical data. Multivariate logistic regression was used to identify individual variables that might influence initial pain relief after surgery. Kaplan-Meier survival analysis was performed with a time interval from surgery to the date of recurring pain and/or increase in antiepileptic medication. If pain was not recurrent, patients were censored at the last follow-up. Survival analysis was performed by using Mantel-Cox log-rank tests to identify individual variables associated with pain recurrence. Multivariate Cox proportional hazard regressions were used to quantify the effect of variables on recurrence-free survival.
Results
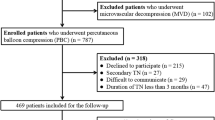
Eligible for the study were 230 patients who underwent 436 procedures with 202 (46%) PBC and 234 (54%) RFTC between 2002 and 2019. Seventeen procedures were terminated prior to ablation and excluded from the initial pain relief and recurrence-free survival analyses. A further 70 procedures had a follow-up shorter than six months and were excluded from the recurrence-free survival calculation (Fig. 3).
Initial pain relief
Table 1 shows a detailed comparison between RFTC and PBC with regard to patient characteristics and initial pain outcome. In comparing the two procedures, a significant difference could only be seen for an older age at surgery (p=0.001), the operating time (p=<0.0001) and the presence of multiple sclerosis (p=0.015). Patients who underwent PBC had a higher median age of 70.5 years, shorter mean operating time of 18.1 ± 9.4 min and suffered less often from multiple sclerosis (30.7%) compared to 66 years, 33.3 ± 14.8 min and 42.7% in the RFTC group.
Initial pain relief after surgery was achieved in 353 (84.2%) procedures and was well balanced between the two groups, with initial pain relief rates of 84.9% for PBC and 83.7% for RFTC.
Multivariate logistic regression analyses showed that patients with multiple sclerosis and patients with a low preoperative BNI had worse initial pain relief with odds ratios of 5.53 and 1.99, respectively (Table 2).
Pain recurrence
Recurrence-free survival analysis was performed in 160 patients who underwent 283 (65%) procedures with a slight predominance for RFTC (56%) compared to PBC (44%) (Fig. 4). The median recurrence-free survival after PBC was not significantly longer (481 days) than that after RFTC (421 days), with a hazard ratio of 0.965 (Table 3). After one year, the recurrence rate was 56.1% for PBC and 56% for RFTC. Multivariate Cox proportional hazard regressions showed that the only factors that contributed to a significantly longer recurrence-free survival were a postoperative BNI pain intensity scale ≤ II (p = <0.001) and a BNI facial numbness score ≥ 3 (p = 0.009; Fig. 4). However, sufficient data on postoperative hypesthesia were only available in 176/283 (62.2%) procedures. Factors that were assumed to have an influence on recurrence-free survival such as: older age, longer duration of symptoms, concomitant continuous pain and the presence of multiple sclerosis showed no influence (Table 3). In Kaplan-Meier survival analysis, no advantage between PBC and RFTC could be seen in the treatment of multiple sclerosis patients (p = 0.103).
Kaplan-Meier survival analysis of patients with trigeminal neuralgia shows a slight advantage for percutaneous balloon compression over radiofrequency thermocoagulation in the probability of pain recurrence. Median pain recurrence was 481 days after percutaneous balloon compression in comparison to 421 days after radiofrequency thermocoagulation. While the classification of trigeminal neuralgia shows no influence, pronounced postoperative hypesthesia is likely to be a positive predictor on the duration of the postoperative pain-free interval. BNI, Barrow Neurologic Institute
Complications
We encountered an overall complication rate of 22.2% with a mortality rate of 0% (Table 4). The complication rate was slightly higher for PBC (25.3%) than for RFTC (19.7%) but did not reach significance (p=0.162). Surgery had to be terminated in 17 (3.9%) procedures because of 1) severe hemorrhage that was not suspended during the operation (1,6%), 2) the inability to correctly place the needle or the balloon (1,4%) and 3) the occurrence of an anesthesiologic problem related to the airway or the circulatory function regardless of the intervention (0.9%). Diplopia was encountered in 4 procedures but never in RFTC. Bradycardia commonly seen in ablative procedures had to be treated with medication in 60% of all cases (n=161; 36,9%). There was no case of corneal keratitis or anesthesia dolorosa after any percutaneous surgery. When analyzing risk factors for complications, there was no significant correlation with age (p=0.956), previous ablative procedures (p=0.102), ASA comorbidity score (0.411), or the presence of multiple sclerosis (0.351).
Discussion
PBC and RFTC are both minimally invasive techniques that offer good initial pain relief with a low-risk profile. The disadvantage comes with a significant recurrence rate that varies greatly between studies, ranging from 15 to 64% [1, 5, 9, 16, 20, 22, 27, 31, 34, 37, 39]. This large variation is partly attributed to differences in patient selection and institutional policies between centers. Comparative evaluations coming from a single center are scarce [15, 16, 26].
In our study, we showed that initial pain relief is comparable between PBC (84.9%) and RFTC (83.7%), while recurrence-free survival of PBC was found to be longer but not statistically significant in multivariate analysis, with a median recurrence-free survival of 481 days compared to 421 days after RFTC. After one year, the recurrence rate was 56.1% for PBC and 56% for RFTC. The recurrence rates given in the literature vary widely between 20% - 64% for PBC [5, 7, 9, 12, 22, 23, 31, 36] and 18–25% for RFTC [6, 16, 20, 36, 37].
This significant difference in the pain recurrence rate between our results and those reported in the literature can possibly be attributed to contrasting definitions of pain relief and recurrence, individual follow-up periods and varying compositions in the patient populations among centers. In our study, the definition of pain recurrence was set very strictly, meaning that every single pain attack and every increase in pain medication was already defined as a recurrence even if the patient improved again afterwards. Others, such as the study by Noorani et al., which showed a recurrence rate of only 25.4% for PBC and 38.2% for RFTC at 1 year, defined recurrence less strict as severe trigeminal pain not fully relieved by medication [26].
The patient population in our study was well balanced between both treatment groups with the exception of age, the presence of multiple sclerosis and the affected division of the trigeminal nerve, reflecting our treatment algorithm over the study period. Patients in the PBC group were significantly older, corroborating the fact that RFTC was performed on awake patients and testing in elderly patients may be complicated by potential cognitive impairment caused by sedation [28]. Likewise, patients with ophthalmic involvement were primarily selected for PBC to prevent corneal reflex impairment. As shown in animal studies, PBC preferentially affects medium and large myelinated fibers and spares small fibers [8]. In only 21/156 (13.5%) patients with multiple sclerosis the ophthalmic division was involved, explaining the high rate of multiple sclerosis patients among the RFTC group in our cohort.
Risk factors for failed initial pain relief and recurrence
Multiple sclerosis and a low preoperative BNI pain intensity scale were found to be the only risk factors for a poor initial pain relief. The influence of the BNI with an odds ratio of 1.99 can easily be explained by the clearer defined difference between the preoperative and postoperative subjectively perceived pain in a very small group of patients (n=7 with a preoperative BNI pain intensity scale ≤ 3).
The dual pathology of multiple sclerosis and trigeminal neuralgia has been shown to have higher recurrence rates in multiple studies, and treatment failure occurs in most patients independently of the type of procedure [10, 24, 40]. While initial pain relief for patients with multiple sclerosis was poor in our study with an odds ratio of 5.53, no influence could be shown for recurrence-free survival. Furthermore, the chosen treatment modality did not show any influence in this subgroup of patients.
Mohammad-Mohammadi et al. showed that PBC (n=82) had the highest initial pain relief compared to RFTC (n=15), glycerol rhizolysis (n=89), microvascular decompression (n=10), stereotactic radiosurgery (n=52), and peripheral neurectomies (n=28). In their study, the median recurrence-free survival (initial procedure/repeat procedure) for PBC was 17/29 months and only 5/9 months for RFTC, which is comparable to the median recurrence-free survival rate of 370 days (≈12 months) in our study [24].
Patients with idiopathic trigeminal neuralgia had a higher but not significant initial failure rate than patients with classical trigeminal neuralgia. Furthermore, a trend towards earlier pain recurrence was also seen in this patient group. Why this patient group harbored poorer results is purely speculative, since the mechanism of damage is just not understood. Furthermore, it could be shown in surgical series that a large part (60%) of idiopathic trigeminal neuralgia defined by preoperative neuroimaging is likely also based on a vascular-nerve conflict [19].
Only a few studies have addressed predictive factors for long-term symptom improvement after PBC or RFTC. The only factors associated with a longer recurrence-free survival in our study were a low postoperative BNI pain intensity scale ≤ II and pronounced postoperative hypesthesia. Many studies anecdotally describe a positive correlation between postoperative hypesthesia and duration of pain relief [2, 26]. Reference is often made to a study by Taha et al. (1995), who performed thermal rhizotomy in 144 patients and who was able to correlate postoperative sensory loss with the rate of pain recurrence. In their cohort analgesia occurred in 46%, dense hypalgesia in 42% and mild hypesthesia in 12% of all patients with 14-year recurrence rates of 20%, 25% and 60%, respectively [33]. Due to the small number of patients in each group (postoperative BNI facial numbness score of 1, n = 20), the question if slight hypesthesia has a better recurrence-free survival than no postoperative hypesthesia cannot be seriously answered by our study. However, our data, as well as those available in the literature, allow the bothersome conclusion that a severely damaged trigeminal ganglion leads to a better recurrence-free survival.
While isolated third division pain has been shown to be a potential prognostic factor for a longer recurrence-free survival after RFTC, this could not be reproduced in our study [21]. Other subjective factors, such as a pear-shaped balloon during PBC, were not collected in our study [2, 22].
Furthermore, our study population shows a high number of patients needing follow-up surgeries (59.6%), secondary trigeminal neuralgia (42%) and concomitant continuous pain (16.9%). In all three groups, poorer initial pain relief and recurrence-free survival rates have been described in the literature, a finding that could, however, not be reproduced in our single-center series [10, 13, 26, 41].
Complications
Both treatment modalities showed zero mortality and an overall complication rate of 25.3% for PBC and 19.7% for RFTC. While this overall complication rate seems high at first glance, one needs to take into account that paraesthesia and a BNI facial numbness score ≥ III were also counted as complications. Anesthesia dolorosa did not occur in any patient, and unwanted ophthalmic hypesthesia occurred in only two patients. The low rates of diplopia, masseter muscle weakness and meningitis are comparable with the current literature [38]. As most patients with ophthalmic division involvement undergo PBC, the rate of diplopia is lower in RFTC. The opposite is the case with masseter muscle weakness, which occurred primarily in patients after RFTC and mandibular division involvement.
Surgery had to be discontinued in 17 (3.9%) cases, of which 13 (2%) cases were related to the surgical procedure. Reasons for abundance were i) pronounced bleeding in the cheek area and ii) inability to enter the foramen ovale. Both complications are more common with PBC given the larger and therefore more traumatic cannula that needs to be used. Rare causes such as Eagle syndrome with an elongated styloid process and calcified stylohyoid ligament also occurred in our cohort. In these cases, technical adjuncts such as intraoperative navigation could help increase safety.
Intraoperative bradycardia occurred more frequently in PBC due to the trigeminal depressor response when entering through the foramen ovale with the larger needle or when inflating the balloon. This well-known phenomenon needs to be communicated with the anesthesiologist beforehand.
Limitations
Like the majority of existing studies on percutaneous interventions for trigeminal neuralgia, this is a retrospective analysis with all the inherent known limitations. Potentially interesting factors that determined initial pain relief and recurrence-free survival in other studies could not be collected, such as the shape of the balloon, which was suggested to represent a key predictor of outcome in PBC [2, 22]. Furthermore, due to our selection criteria (Fig. 1) for one or the other treatment, there is an unavoidable selection bias and thus an unequal distribution in the treatment groups.
Conclusions
Both percutaneous interventions led to a favorable and comparable initial pain relief in trigeminal neuralgia. In the long run, however, PBC was associated with a slightly but not significantly longer recurrence-free survival than RFTC. At present, there are no randomized controlled trials to seriously compare the effectiveness of ablative interventions. Based on the data currently available to us, both treatment modalities harbor distinct advantages and should be understood as concomitant rather than conflicting procedures. An individualized approach in a center able to offer all available modalities is warranted.
Abbreviations
- BNI:
-
Barrow Neurological Institute
- PBC:
-
percutaneous balloon compression
- RFTC:
-
radiofrequency thermocoagulation
References
Abdennebi B, Guenane L (2014) Technical considerations and outcome assessment in retrogasserian balloon compression for treatment of trigeminal neuralgia. Series of 901 patients. Surg Neurol Int 5:118. https://doi.org/10.4103/2152-7806.137838
Asplund P, Linderoth B, Bergenheim AT (2010) The predictive power of balloon shape and change of sensory functions on outcome of percutaneous balloon compression for trigeminal neuralgia. J Neurosurg 113:498–507. https://doi.org/10.3171/2010.2.Jns091466
Bendtsen L, Zakrzewska JM, Abbott J, Braschinsky M, Di Stefano G, Donnet A, Eide PK, Leal PRL, Maarbjerg S, May A, Nurmikko T, Obermann M, Jensen TS, Cruccu G (2019) European Academy of Neurology guideline on trigeminal neuralgia. Eur J Neurol 26:831–849. https://doi.org/10.1111/ene.13950
Bendtsen L, Zakrzewska JM, Heinskou TB, Hodaie M, Leal PRL, Nurmikko T, Obermann M, Cruccu G, Maarbjerg S (2020) Advances in diagnosis, classification, pathophysiology, and management of trigeminal neuralgia. Lancet Neurol 19:784–796. https://doi.org/10.1016/s1474-4422(20)30233-7
Bergenheim AT, Asplund P, Linderoth B (2013) Percutaneous retrogasserian balloon compression for trigeminal neuralgia: review of critical technical details and outcomes. World Neurosurg 79:359–368. https://doi.org/10.1016/j.wneu.2012.03.014
Broggi G, Franzini A, Lasio G, Giorgi C, Servello D (1990) Long-term results of percutaneous retrogasserian thermorhizotomy for "essential" trigeminal neuralgia: considerations in 1000 consecutive patients. Neurosurgery 26:783–786; discussion 786-787. https://doi.org/10.1097/00006123-199005000-00008
Brown JA, McDaniel MD, Weaver MT (1993) Percutaneous trigeminal nerve compression for treatment of trigeminal neuralgia: results in 50 patients. Neurosurgery 32:570–573. https://doi.org/10.1227/00006123-199304000-00012
Brown JA, Hoeflinger B, Long PB, Gunning WT, Rhoades R, Bennett-Clarke CA, Chiaia NL, Weaver MT (1996) Axon and ganglion cell injury in rabbits after percutaneous trigeminal balloon compression. Neurosurgery 38:993–1003 discussion 1003-1004
Chen JF, Tu PH, Lee ST (2011) Long-term follow-up of patients treated with percutaneous balloon compression for trigeminal neuralgia in Taiwan. World Neurosurg 76:586–591. https://doi.org/10.1016/j.wneu.2011.05.021
Cheng JS, Sanchez-Mejia RO, Limbo M, Ward MM, Barbaro NM (2005) Management of medically refractory trigeminal neuralgia in patients with multiple sclerosis. Neurosurg Focus 18:e13. https://doi.org/10.3171/foc.2005.18.5.14
Cheng JS, Lim DA, Chang EF, Barbaro NM (2014) A review of percutaneous treatments for trigeminal neuralgia. Neurosurgery 10(Suppl 1):25–33; discussion 33. https://doi.org/10.1227/neu.00000000000001687
Corrêa CF, Teixeira MJ (1998) Balloon compression of the Gasserian ganglion for the treatment of trigeminal neuralgia. Stereotact Funct Neurosurg 71:83–89. https://doi.org/10.1159/000029651
Cruccu G (2017) Trigeminal Neuralgia. Continuum (Minneap Minn) 23:396–420. https://doi.org/10.1212/con.0000000000000451
Cruccu G, Finnerup NB, Jensen TS, Scholz J, Sindou M, Svensson P, Treede R-D, Zakrzewska JM, Nurmikko T (2016) Trigeminal neuralgia. Neurology 87:220–228. https://doi.org/10.1212/wnl.0000000000002840
Fraioli B, Esposito V, Guidetti B, Cruccu G, Manfredi M (1989) Treatment of trigeminal neuralgia by thermocoagulation, glycerolization, and percutaneous compression of the gasserian ganglion and/or retrogasserian rootlets: long-term results and therapeutic protocol. Neurosurgery 24:239–245. https://doi.org/10.1227/00006123-198902000-00014
Frank F, Fabrizi AP (1989) Percutaneous surgical treatment of trigeminal neuralgia. Acta Neurochir (Wien) 97:128–130. https://doi.org/10.1007/BF01772823
Fritz H (1914b) Über die intracraniale Injektionsbehandlung der Trigeminus-Neuralgie. Med Klin 1:582
Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd ed (2018). Cephalalgia 38:1-211. https://doi.org/10.1177/0333102417738202
Herta J, Schmied T, Loidl TB, Wang W-t, Marik W, Winter F, Tomschik M, Ferraz-Leite H, Rössler K, Dorfer C (2021) Microvascular decompression in trigeminal neuralgia: predictors of pain relief, complication avoidance, and lessons learned. Acta Neurochir. https://doi.org/10.1007/s00701-021-05028-2
Kanpolat Y, Savas A, Bekar A, Berk C (2001) Percutaneous controlled radiofrequency trigeminal rhizotomy for the treatment of idiopathic trigeminal neuralgia: 25-year experience with 1,600 patients. Neurosurgery 48:524–532; discussion 532-524. https://doi.org/10.1097/00006123-200103000-00013
Kosugi S, Shiotani M, Otsuka Y, Suzuki T, Katori N, Hashiguchi S, Morisaki H (2015) Long-term outcomes of percutaneous radiofrequency thermocoagulation of gasserian ganglion for 2nd- and multiple-division trigeminal neuralgia. Pain Pract 15:223–228. https://doi.org/10.1111/papr.12163
Kouzounias K, Schechtmann G, Lind G, Winter J, Linderoth B (2010) Factors that influence outcome of percutaneous balloon compression in the treatment of trigeminal neuralgia. Neurosurgery 67:925–934; discussion 934. https://doi.org/10.1227/NEU.0b013e3181eb5230
Lichtor T, Mullan JF (1990) A 10-year follow-up review of percutaneous microcompression of the trigeminal ganglion. J Neurosurg 72:49–54. https://doi.org/10.3171/jns.1990.72.1.0049
Mohammad-Mohammadi A, Recinos PF, Lee JH, Elson P, Barnett GH (2013) Surgical outcomes of trigeminal neuralgia in patients with multiple sclerosis. Neurosurgery 73:941–950; discussion 950. https://doi.org/10.1227/neu.0000000000000128
Mullan S, Lichtor T (1983) Percutaneous microcompression of the trigeminal ganglion for trigeminal neuralgia. J Neurosurg 59:1007–1012. https://doi.org/10.3171/jns.1983.59.6.1007
Noorani I, Lodge A, Vajramani G, Sparrow O (2016) Comparing Percutaneous Treatments of Trigeminal Neuralgia: 19 Years of Experience in a Single Centre. Stereotact Funct Neurosurg 94:75–85. https://doi.org/10.1159/000445077
Omeis I, Smith D, Kim S, Murali R (2008) Percutaneous balloon compression for the treatment of recurrent trigeminal neuralgia: long-term outcome in 29 patients. Stereotact Funct Neurosurg 86:259–265. https://doi.org/10.1159/000138770
Papaioannou A, Fraidakis O, Michaloudis D, Balalis C, Askitopoulou H (2005) The impact of the type of anaesthesia on cognitive status and delirium during the first postoperative days in elderly patients. Eur J Anaesthesiol 22:492–499. https://doi.org/10.1017/s0265021505000840
Preul MC, Long PB, Brown JA, Velasco ME, Weaver MT (1990) Autonomic and histopathological effects of percutaneous trigeminal ganglion compression in the rabbit. J Neurosurg 72:933–940. https://doi.org/10.3171/jns.1990.72.6.0933
Rogers CL, Shetter AG, Fiedler JA, Smith KA, Han PP, Speiser BL (2000) Gamma knife radiosurgery for trigeminal neuralgia: the initial experience of The Barrow Neurological Institute. Int J Radiat Oncol Biol Phys 47:1013–1019. https://doi.org/10.1016/s0360-3016(00)00513-7
Skirving DJ, Dan NG (2001) A 20-year review of percutaneous balloon compression of the trigeminal ganglion. J Neurosurg 94:913–917. https://doi.org/10.3171/jns.2001.94.6.0913
Sweet WH, Wepsic JG (1974) Controlled thermocoagulation of trigeminal ganglion and rootlets for differential destruction of pain fibers. 1. Trigeminal neuralgia. J Neurosurg 40:143–156. https://doi.org/10.3171/jns.1974.40.2.0143
Taha JM, Tew JM Jr, Buncher CR (1995) A prospective 15-year follow up of 154 consecutive patients with trigeminal neuralgia treated by percutaneous stereotactic radiofrequency thermal rhizotomy. J Neurosurg 83:989–993. https://doi.org/10.3171/jns.1995.83.6.0989
Tang YZ, Wu BS, Yang LQ, Yue JN, He LL, Li N, Ni JX (2015) The Long-Term Effective Rate of Different Branches of Idiopathic Trigeminal Neuralgia After Single Radiofrequency Thermocoagulation: A Cohort Study. Medicine (Baltimore) 94:e1994. https://doi.org/10.1097/md.0000000000001994
Tatli M, Sindou M (2008) Anatomoradiological landmarks for accuracy of radiofrequency thermorhizotomy in the treatment of trigeminal neuralgia. Neurosurgery 63:ONS129–ONS137; discussion ONS137-128. https://doi.org/10.1227/01.neu.0000335027.00650.83
Tatli M, Satici O, Kanpolat Y, Sindou M (2008) Various surgical modalities for trigeminal neuralgia: literature study of respective long-term outcomes. Acta Neurochir (Wien) 150:243–255. https://doi.org/10.1007/s00701-007-1488-3
Tronnier VM, Rasche D, Hamer J, Kienle AL, Kunze S (2001) Treatment of idiopathic trigeminal neuralgia: comparison of long-term outcome after radiofrequency rhizotomy and microvascular decompression. Neurosurgery 48:1261–1267 discussion 1267-1268
Wang JY, Bender MT, Bettegowda C (2016) Percutaneous Procedures for the Treatment of Trigeminal Neuralgia. Neurosurg Clin N. Am 27:277–295. https://doi.org/10.1016/j.nec.2016.02.005
Yoon KB, Wiles JR, Miles JB, Nurmikko TJ (1999) Long-term outcome of percutaneous thermocoagulation for trigeminal neuralgia. Anaesthesia 54:803–808. https://doi.org/10.1046/j.1365-2044.1999.00905.x
Zakrzewska JM, Wu J, Brathwaite TSL (2018) A Systematic Review of the Management of Trigeminal Neuralgia in Patients with Multiple Sclerosis. World Neurosurg 111:291–306. https://doi.org/10.1016/j.wneu.2017.12.147
Zheng S, Li X, Li R, Yang L, He L, Cao G, Yang Z, Ni J (2019) Factors Associated With Long-term Risk of Recurrence After Percutaneous Radiofrequency Thermocoagulation of the Gasserian Ganglion for Patients With Trigeminal Neuralgia: A Multicenter Retrospective Analysis. Clin J Pain 35:958–966. https://doi.org/10.1097/ajp.0000000000000758
Funding
Open access funding provided by Medical University of Vienna.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
Ethikkommission Medizinische Universität Wien / EK Nr. : 1105/2020.
Informed consent
All patients gave their informed consent before the procedure.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Herta, J., Loidl, T.B., Schmied, T. et al. Retrospective comparison of percutaneous balloon compression and radiofrequency-thermocoagulation in the management of trigeminal neuralgia. Acta Neurochir 165, 1943–1954 (2023). https://doi.org/10.1007/s00701-023-05656-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-023-05656-w