Abstract
Objective/purpose
The aim of the study was to assess the options of treatment and their related outcomes for chondral injuries in the hip based on the available evidence whilst highlighting new and innovative techniques.
Methods
A systematic review of the literature from PubMed (Medline), EMBASE, Google Scholar, British Nursing Index (BNI), Cumulative Index to Nursing and Allied Health Literature (CINAHL) and Allied and Complementary Medicine Database (AMED) was undertaken from their inception to March 2017 using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. Clinical outcome studies, prospective/retrospective case series and case reports that described the outcome of cartilage repair technique for the chondral injury in the hip were included. Studies on total hip replacement, animal studies, basic studies, trial protocols and review articles were excluded.
Results
The systematic review found 21 relevant papers with 596 hips. Over 80% of the included studies were published in or after 2010. Most studies were case series or case reports (18 studies, 85.7%). Arthroscopy was used in 11 studies (52.4%). The minimum follow-up period was six months. Mean age of the participants was 37.2 years; 93.5% of patients had cartilage injuries of the acetabulum and 6.5% of them had injuries of the femoral head. Amongst the 11 techniques described in the systematic review, autologous matrix-induced chondrogenesis, osteochondral autograft transplantation and microfracture were the three frequently reported techniques.
Conclusion
Over ten different techniques are available for cartilage repair in the hip, and most of them have good short- to medium-term outcomes. However, there are no robust comparative studies to assess superiority of one technique over another, and further research is required in this arena.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Isolated chondral and osteochondral defects within the hip joint often present a technical challenge for the hip surgeon. Common causes of cartilage damage in the hip include femoroacetabular impingement (FAI), developmental dysplasia, osteonecrosis, osteochondritis dissecans, loose bodies, slipped capital femoral epiphysis, and trauma [1,2,3,4,5]. Amongst them, FAI has increasingly gained recognition as a major cause of chondral injury and subsequent development of arthritis in the hip joint [6,7,8,9,10]. In CAM FAI, the abnormal contact between the aspherical femoral head-neck junction and the acetabular rim results in a large amount of shear stress being transmitted to the labro-chondral junction. Over a period of time, labral detachment and acetabular chondral damage ensues [2, 11, 12]. On the other hand, the pincer FAI, in which a deep or retroverted acetabulum makes contact with a normal-shaped femoral neck, has a recognised pattern of damage to the labrum, femoral head cartilage and a postero-medial acetabular countercoup lesion [13]. Furthermore, in imaging and surgical techniques like hip arthroscopy have led to increased recognition of chondral lesions. The incidence of chondral lesions at hip arthroscopy for FAI has been reported to be up to 67.3% of the patients in one series [14].
There is relatively little information about articular cartilage restoration in the hip when compared with what is known about cartilage restoration in the knee. Currently, most cartilage repair methods for the hip are based on basic science and strategies that were developed for the knee. Awareness of young adult hip disease has been increasing in recent years, and thus, the field of hip preservation continues to develop; several new innovative techniques have been performed and described in the literature. They include microfracture, autologous chondrocyte implantation (ACI), matrix-associated chondrocyte implantation (MACI), autologous matrix-induced chondrogenesis (AMIC), osteochondral autograft/allograft transplantation, implantation of artificial plug, sticking down of chondral flaps with fibrin adhesive and an intra-articular injection of bone marrow mesenchymal stem cells (BM-MSCs).
Currently, there is a gap in information particularly regarding systematic reviews in the literature that provide hip surgeons with evidence-based recommendations, therefore, on treating cartilage injuries in the hip. The aim of this study was to provide the reader with options of treatment and their related outcomes for chondral injuries in the hip based on the available evidence whilst highlighting new and innovative techniques involved in chondral repair.
Methods
Search strategy
Two reviewers (NN and CG) searched the online databases (PubMed (Medline), EMBASE, Google Scholar, British Nursing Index (BNI), Cumulative Index to Nursing and Allied Health Literature (CINAHL) and Allied and Complementary Medicine Database (AMED) for literature describing the outcome of cartilage repair techniques for the chondral injury in the hip. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were used for designing this study. A detailed search strategy is described in the Appendix.
Study screening/data abstraction
The inclusion and exclusion criteria are shown in Table 1. Both the reviewers independently abstracted the relevant study data from the final pool of included articles and recorded this data on a spreadsheet designed a priori. Participant-specific demographics extracted from each study included the number of hips, gender distribution, mean age with range (years), length of follow-up, location of the cartilage injury (acetabulum or femoral head), surgical approach (open dislocation, arthroscopy or injection), cartilage restoration technique used in the study, pre-operative condition of the damaged cartilage, final outcome and specific comments (if any).
Statistics
The abstracted evidence was collected and analysed using Microsoft Excel 2013 spread sheet. Statistical analysis in this study focused on descriptive statistics.
Results
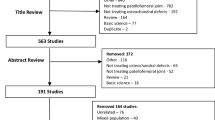
Flowchart of the literature search is shown in Fig. 1. The oldest study included in this review was published in 2003, and over 80% of the included studies (17 out of 21 studies) were published in or after 2010. Study demographics are shown in Table 2. A total of 11 techniques were found from the systematic review: AMIC (5 studies), osteochondral autograft transplantation (mosaicplasty) (5 studies), microfracture (4 studies), artificial plug (TruFit®) (2 studies), fibrin adhesive (2 studies), ACI (2 studies), debridement (1 study), MACI (1 study), osteochondral allograft transplantation (1 study), direct cartilage suture repair (1 study) and intra-articular BM-MSC injection (1 study). Three studies described two techniques and compared them to each other (microfracture and AMIC, 1 study; MACI and AMIC, 1 study; ACI and debridement, 1 study).
Details of the 21 studies included are shown in Table 3.
Discussion
Our objective was to discuss the outcomes of the current strategies for restoration of focal chondral injuries in the hip. This study reviews all the cases of cartilage repair for the chondral injuries in the hip (596 cases) reported in the English literature and describes the outcomes of 11 techniques (including debridement).
Cartilage injuries in the hip have been previously shown to result in poor long-term outcomes, including pain and early secondary degenerative change followed by the subsequent development of osteoarthritis [15, 16]. The current trend is to focused on treating isolated cartilage damage and underlying morphological pathology in younger patients in order to prevent progression to end-stage degeneration. Although a number of procedures for the management of chondral lesions in other large joints (e.g. knee) have been reported, there currently remains little information available for appropriate management of these lesions in the hip [17]. All the techniques found in the systematic review are described and discussed below.
Debridement
Debridement of a cartilage flap from a chondral injury may allow symptoms to resolve and permit a return to activity or sports [6, 18]. Arthroscopy is essential for the diagnosis of an unstable flap if pre-operative imaging is unclear, and arthroscopic debridement is often the definitive therapy. Fontana et al.. [19] carried out a controlled retrospective study of 30 patients (15 ACI, 15 arthroscopic debridements) affected by a post-traumatic hip chondropathy (Outerbridge classification grades 3–4, measuring 2 cm2 in area or more). The post-operative Harris Hip Scores (HHS) in the ACI group were significantly better than those in the debridement group.
Microfracture
Microfracture involves the use of an arthroscopic awl or drill to perforate exposed subchondral bone to create multiple holes and provide an entry portal for marrow-derived cells. The rationale of the technique is to recruit mesenchymal stem cells into the cartilage defect to create fibrocartilage. Following microfracture, a marrow clot forms and provides the ideal environment for mesenchymal stem cells to differentiate into stable repair tissue [20]. The advantages of this technique are that it is technically straightforward, can be performed arthroscopically, without donor site morbidity, and has a low cost. The disadvantage compared with other cartilage repair techniques is that it produces less type II cartilage and has different biomechanical properties than hyaline cartilage, which may raise questions of its resilience and longevity [20]. Also, the concentration of mesenchymal cells in the bone marrow is relatively low and their chondrogenic potential declines with age [21]. Philippon et al. [22] reported the outcome of microfracture in nine patients with a full-thickness chondral defect of the acetabulum. The average percent fill of the acetabular chondral lesions at second-look arthroscopy was 91%, and eight of the nine patients had grade 1/2 repair product at second look. Karthikeyan et al. [23] described the outcome of 20 patients who underwent arthroscopic surgery for FAI with a localised full-thickness acetabular chondral defect which were treated by microfracture. At an average follow-up of 17 months, 19 of the 20 patients had a mean fill of 96% with macroscopically good quality repair tissue. Zaltz and Leunig [24] reviewed ten patients with symptomatic FAI diagnosed with parafoveal chondral defects confirmed at the time of surgical dislocation. Seven of the ten patients were treated by microfracture (other 3 were treated by AMIC), and all the patients were able to return to their pre-operative level of function with the exception of one patient who had a problem in the contralateral hip. At the last follow-up, there was no obvious asymmetric joint space narrowing visible on an AP pelvis view in any of the patients. Fontana et al. [25] compared the outcome of 77 patients who had a microfracture and 70 patient who had AMIC for cartilage injuries in the hip. Although the outcome in both groups significantly improved at six months and one year post-operatively, the outcome in the microfracture group slowly deteriorated four years after surgery, whilst that in the AMIC group remained stable.
Autologous chondrocyte implantation
ACI includes the harvest of chondrocytes with growth and expansion at an off-site facility, followed by reimplantation of the cells into the affected area. ACI is indicated for symptomatic, well-contained defects that are between 2 and 10 cm2 and with less than 6–8 mm of bone loss [26]. Most surgeons who perform ACI regularly are now using a synthetic collagen membrane to cover the implanted chondrocytes [19, 27]. Ellender and Minas [27] presented a clinical case and described ACI for a femoral head chondral defect of 10 cm2 in a 19-year-old female college student who had progression of disease after prior mosaicplasty. Two years after ACI, she remained free of pain. Her post-operative contrast-enhanced MRI demonstrated repair tissue fill and radiographs showed a normal joint space without any sign of change.
Matrix-associated chondrocyte implantation
MACI is a second-generation ACI technique that utilises absorbable scaffolds to support the implanted chondrocytes during healing. Theoretically, it should restore hyaline cartilage at the defect. Unfortunately, same as ACI, it is a two-stage procedure where chondrocytes are harvested from the patient, cultured and then returned to the patient via open surgical dislocation of the hip which is a technically demanding surgical approach. Mancini and Fontana [28] assessed and compared the clinical outcomes of arthroscopic MACI and AMIC for the treatment of acetabular chondral defects between 2 and 4 cm2 consequent to FAI. In both groups, significant improvement in modified HHS (mHHS) was measured over baseline levels at six months post-operation. It continued to improve up to three years post-operation and remained stable until five years follow-up. There was no statistically significant difference between the two groups.
Autologous matrix-induced chondrogenesis
AMIC is a novel single-step procedure in which the microfracture technique has been enhanced by the use of a collagen matrix. The Chondro-Gide matrix is placed in the defect and a porcine collagen I/III matrix is sewn over the lesion to stabilise the fragile blood clot that arises from the microfracture to provide a stable infrastructure for the formation of repair tissue [29]. No cells have to be harvested, cultured and re-implanted in AMIC. Therefore, there is no harvest site morbidity, and the operation can be performed as a single procedure. Moreover, AMIC does not require complex cell expansion techniques. Other than comparative studies with microfracture [24, 25] or MACI [28] described above, Leunig et al. [30] reported six patients with AMIC using surgical dislocation of the hip. No complications occurred, and good post-operative outcome scores were reported. Fontana [31] treated 201 patients with AMIC arthroscopically for Outerbridge grade III/IV chondral lesions of the acetabulum. Modified HHS improved significantly at six months post-operatively in comparison with pre-operative levels, reaching the highest level of improvement at the three year follow-up.
Osteochondral autograft transplantation (mosaicplasty)
Mosaicplasty involves transplanting healthy, mature cartilage from a non-weight-bearing part of the hip or knee to an articular defect. The transplanted cartilage integrates with the adjacent host cartilage via fibrocartilage [32]. The inferior aspect of the femoral head, the femoral head-neck junction and the periphery of the femoral trochlea of the knee can be the potential donor sites. Mosaicplasty offers many potential advantages, including the ability to transfer new mature hyaline cartilage into the defect in a single-stage procedure and the absence of potential disease transmission, which can occur in allograft transplantation. On the contrary, owing to the autologous nature of this technique, it is limited by donor site morbidity, graft availability and the potential for dead space between the grafts [32]. Hart et al. [33] first reported the case of an osteochondral defect of the femoral head and subsequent treatment using mosaicplasty with open surgical dislocation of the hip. At six months following surgery, the patient had a full range of painless movement of the hip with no further complaints of pain related to activities. Emre et al. [34] also presented a case where the defect of the femoral head was treated with surgical dislocation of the hip and mosaicplasty. The patient was symptom-free with nearly complete incorporation of the graft radiologically at three years after the operation. Nam et al. [35] reported two cases of a chondral defect on the femoral head after a traumatic hip dislocation, treated with mosaicplasty from the ipsilateral knee, and the inferior femoral head, respectively. At 1 and five years of follow-up, MRI showed good autograft incorporation with the maintenance of articular surface conformity. Krych et al. [36] reported two cases of post-traumatic osteochondral defects of the femoral head. Both the patients were treated with mosaicplasty from the ipsilateral knee to the femoral head, with successful clinical and radiological results at a mean follow-up of 4.3 years. Girard et al. [37] treated 10 patients for femoral cartilage defects by mosaicplasty of the femoral head through a trochanteric flip osteotomy with surgical dislocation of the hip. At the mean follow-up of 29.2 months, clinical score and range of motion improved significantly. All radiological investigations at the latest follow-up showed that the grafts were well-incorporated at the site of mosaicplasty with intact cartilage over them and smooth interfaces between articulating bony surfaces.
Osteochondral allograft transplantation
Mosaicplasty has been shown to be a useful procedure, but there can be donor site morbidity and there is a limit to the size of the treatable defect. Allograft transplantation can also be a successful solution for the treatment of cartilage defects. It offers not only the potential advantages of transferring immediate functional hyaline cartilage but also the ability to resurface a large area without associated donor site morbidity. Potential allograft donor sources for defects within the acetabular side of the hip were a cadaveric acetabulum or medial tibial plateau. Cartilage is relatively immunoprivileged and avascular; thus, the host immune reaction is considered to be limited [38]. Allograft bone becomes necrotic and is reabsorbed via creeping substitution during the healing process. This provides a scaffold and supports the articular surface as part of gradual incorporation [39]. In the systematic review, Krych et al. [40] reported their experience in two patients who underwent osteochondral allograft transplantation for the acetabular cartilage defects. MRI at 18 months in both cases demonstrated incorporation of the graft into the host acetabulum. Hip Outcome Scores (HOS) were 100 points each in both patients two years post-operatively.
Direct cartilage suture repair
Delamination is a full-thickness cartilage separation from the underlying subchondral bone, which forms an unstable flap at risk for complete detachment [41]. Our review found a case report that presented direct cartilage repair as a possible technique to treat large delaminated full-thickness acetabular cartilage repairs. Sekiya et al. [42] described a case of a 17-year-old boy presented with bilateral hip pain because of bilateral CAM-type FAI and a 1-cm delaminated unstable cartilage flap in the anterior-superior acetabulum. Arthroscopic microfracture underneath the flap of anterior-superior acetabular cartilage and an absorbable monofilament suture repair of the cartilage was conducted. At two years post-operatively, the patient reported 95% of normal function for both hips. Overall, the patient was satisfied with the outcome including a score of 96 on the mHHS, 93 on the HOS Activities of Daily Living subscale and 81 on the HOS Sports subscale at the final follow-up.
Fibrin adhesive
The earliest stage in the formation of an articular cartilage flap is delamination of the overlying articular cartilage from the underlying subchondral bone [43]. Particularly, if the articular cartilage itself may contain a significant number of viable chondrocytes, debriding such an area of chondral instability seems an unnecessary surgical procedure. Fibrin adhesive is a biological substance, which has already been used in general surgery, ophthalmology, neurosurgery, otolaryngology and orthopaedics, thanks to its adhesive properties [44,45,46,47,48]. This procedure involved creating an incision at the periphery of the acetabular labrum and passing an awl underneath to create microfracture. Fibrin glue was inserted between subchondral bone and delaminated cartilage, and the cartilage was pressed down until the adhesive had set. Tzaveas and Villar [49] analysed the efficacy of using fibrin adhesive for arthroscopic repair of chondral delamination lesions with intact gross cartilage structure in 19 patients. Mean mHHS was improved significantly after surgery, and in all five patients who underwent revision arthroscopy at a later date, the chondral repair appeared intact. Stafford et al. [50] reported the results of 43 patients with FAI who have undergone fibrin adhesive technique for re-attachment of delaminated chondral flaps. Both mHHS for pain and function improved significantly after the operation. In three patients who required further arthroscopic interventions for persistent symptoms created by iliopsoas irritation, the previously repaired articular cartilage was found in a good condition.
Intra-articular BM-MSC injection
Adult MSCs were originally believed to only differentiate into tissue-specific cells. However, these cells were recently proven to have the ability to differentiate into a different tissue in response to specific signals released by the site of injury, including cartilage injury [51, 52]. Adding to animal studies, several authors reported on intra-articular injection of MSCs into the knee for the treatment of cartilage defects and showed good results with regard to pain and clinical outcomes [53,54,55,56]. Injected MSCs were incorporated into the articular cartilage of the injected joint. They integrate into the surface of the cartilage and also the interior of the cartilage [52]. Mardones et al. [57] first reported the outcome of intra-articular BM-MSC injection for the cartilage injury in the hip. Three intra-articular injections of 20 × 106 BM-MSCs were conducted from four to six weeks post-operatively in 29 hips that received hip arthroscopy for FAI and focal cartilage injuries. Clinical outcome scores and VAS improved significantly after surgery, and no major complications had been reported at the time of the last follow-up.
Artificial plug
The systematic review found two articles that used an artificial plug, and both of them utilise the TruFit cartilage/bone (CB) plug (Smith & Nephew). It is a resorbable polymer scaffold that can be inserted into osteochondral defects, which acts as a scaffold that provides structural support. Also, native marrow elements can migrate into the plug to promote bone in-growth as well as articular cartilage regeneration. Field et al. [58] described the use of TruFit for the treatment of acetabular cystic cartilage lesions in four patients. Patients underwent hip arthroscopy followed by the antegrade insertion of a plug through the ilium until the surface of the plug coincided with the articular surface. At ten months follow-up, patients reported increased function and improvement in Non-Arthritic Hip Score (NAHS). CT and MRI showed incorporation and continued healing of the plug six months post-operatively. Vundelinckx et al. [59] reported a case of a 34-year-old employee (gender was not described) who underwent TruFit for an osteochondral injury of the femoral head. MRI at six months showed the TruFit was placed in situ whilst there was an irregularity on the border of the articular cartilage surface. They mentioned it was very difficult to interpret early MRI images of ingrowth of TruFit plugs, as described by authors of past radiographic studies [60].
Of the 21 studies found in the systematic review, only 3 studies are level IIIb (retrospective comparative study) and the rest were level IV (case series/report). Two studies described superiority of one cartilage repair method over another [19, 25], and one study showed there was no difference in clinical outcome between two methods [28]. Fontana’s study [19] was limited by the reduced number of patients and the lack of an objective method for the evaluation of the results. Other limitations are the criteria for patient inclusion and selection bias in the randomisation process. Fontana’s study [25] and Mancini’s study [28] were also limited by the lack of randomisation, and clinical outcomes were only assessed using the mHHS.
The strengths of this systematic review include the pursuit of knowledge in an important novel area of investigation and a rigorous methodological approach. Regarding the methodological approach, a broad-based and comprehensive literature search of multiple databases with multiple reviewers allowed for a very inclusive approach to capture the vast majority of existing literature. Nonetheless, there are limitations which include the inclusion of English only studies and the overall low level of evidence available in the included studies on this topic (mostly level IIIb and IV studies). Retrospective designs are prone to data inaccuracy as well as missing information, which subject them to selection and detection bias. Without a doubt, this diminishes the accuracy of the data collected and, therefore, limits the quality of a systematic review, whilst this current level of evidence reflects the novel and emerging nature of cartilage repair strategies in the hip joint. Additionally, our results include a wide spectrum of pathologies and methods of treatment, which also made drawing conclusions and giving specific guidelines difficult. Furthermore, pre-operative condition and post-operative rehabilitation protocol were different in each study, which made comparison among studies difficult as well. Future studies should address comparative effectiveness of the various treatment options, and long-term registry-based studies that report patient reported outcomes and radiographic outcomes will help inform treatment decisions.
Conclusion
Although there are many different cartilage restoration techniques available, current best evidence does not support any one surgical technique as a superior method for treating cartilage injuries in the hip. Unfortunately there remains a paucity of randomised trials with long-term follow-up, which makes it difficult to perform a meaningful assessment of the outcome of each procedure. Of the 21 studies found in the systematic review, AMIC, mosaicplasty and microfracture were relatively well-reported, though they were only described in very limited case series. Also, only two studies described superiority of one cartilage repair method over another—one showed superiority of AMIC over microfracture [25] and another showed superiority of ACI over debridement [19], and one study showed that there was no statistically significant difference between MACI and AMIC in terms of post-operative mHHS [28]. To make any specific recommendations for orthopaedic surgeons with regards to treatment decisions, adequately powered long-term large-scale high-quality randomised-control trials focusing on two or three specific methods of treatment need to be conducted in the future.
Contribution of authors
VK takes responsibility for the integrity of the work as a whole, from inception to the finished manuscript. NN, CG, AD, OA and VK were responsible for the conception and design; NN, CG and VK for the collection, assembly, analysis and interpretation of data; NN, OA and VK for drafting; and NN, CG, AD, OA and VK for the final approval of the manuscript and for the critical revision for important intellectual contents.
References
Crawford K, Philippon MJ, Sekiya JK, Rodkey WG, Steadman JR (2006) Microfracture of the hip in athletes. Clin Sports Med 25:327–335
Yen YM, Kocher MS (2010) Chondral lesions of the hip: microfracture and chondroplasty. Sports Med Arthrosc Rev 18:83–89
Beaule PE, Bleeker H, Singh A, Dobransky J (2017) Defining modes of failure after joint-preserving surgery of the hip. Bone Joint J 99-B:303–309
Fayad TE, Khan MA, Haddad FS (2013) Femoroacetabular impingement: an arthroscopic solution. Bone Joint J 95-B:26–30
Leunig M, Ganz R (2014) The evolution and concepts of joint-preserving surgery of the hip. Bone Joint J 96-B:5–18
Bedi A, Kelly BT, Khanduja V (2013) Arthroscopic hip preservation surgery: current concepts and perspective. Bone Joint J 95-B:10–19
Imam S, Khanduja V (2011) Current concepts in the diagnosis and management of femoroacetabular impingement. Int Orthop 35:1427–1435
Khanduja V, Villar RN (2006) Arthroscopic surgery of the hip: current concepts and recent advances. J Bone Joint Surg Br 88:1557–1566
Khanduja V, Villar RN (2007) The arthroscopic management of femoroacetabular impingement. Knee Surg Sports Traumatol Arthrosc 15:1035–1040
Anderson LA, Peters CL, Park BB, Stoddard GJ, Erickson JA, Crim JR (2009) Acetabular cartilage delamination in femoroacetabular impingement. Risk factors and magnetic resonance imaging diagnosis. J Bone Joint Surg Am 91:305–313
Bedi A, Lynch EB, Sibilsky Enselman ER, Davis ME, Dewolf PD, Makki TA, Kelly BT, Larson CM, Henning PT, Mendias CL (2013) Elevation in circulating biomarkers of cartilage damage and inflammation in athletes with femoroacetabular impingement. Am J Sports Med 41:2585–2590
Johnston TL, Schenker ML, Briggs KK, Philippon MJ (2008) Relationship between offset angle alpha and hip chondral injury in femoroacetabular impingement. Arthroscopy 24:669–675
Ellis HB, Briggs KK, Philippon MJ (2011) Innovation in hip arthroscopy: is hip arthritis preventable in the athlete? Br J Sports Med 45:253–258
Nepple JJ, Carlisle JC, Nunley RM, Clohisy JC (2011) Clinical and radiographic predictors of intra-articular hip disease in arthroscopy. Am J Sports Med 39:296–303
Beck M, Kalhor M, Leunig M, Ganz R (2005) Hip morphology influences the pattern of damage to the acetabular cartilage: femoroacetabular impingement as a cause of early osteoarthritis of the hip. J Bone Joint Surg Br 87:1012–1018
Ganz R, Parvizi J, Beck M, Leunig M, Notzli H, Siebenrock KA (2003) Femoroacetabular impingement: a cause for osteoarthritis of the hip. Clin Orthop Relat Res 417:112–120
McCulloch PC, Kang RW, Sobhy MH, Hayden JK, Cole BJ (2007) Prospective evaluation of prolonged fresh osteochondral allograft transplantation of the femoral condyle: minimum 2-year follow-up. Am J Sports Med 35:411–420
Byrd JW (2001) Lateral impact injury. A source of occult hip pathology. Clin Sports Med 20:801–815
Fontana A, Bistolfi A, Crova M, Rosso F, Massazza G (2012) Arthroscopic treatment of hip chondral defects: autologous chondrocyte transplantation versus simple debridement—a pilot study. Arthroscopy 28:322–329
Frisbie DD, Oxford JT, Southwood L, Trotter GW, Rodkey WG, Steadman JR, Goodnight JL, McIlwraith CW (2003) Early events in cartilage repair after subchondral bone microfracture. Clin Orthop Relat Res 407:215–227
Payne KA, Didiano DM, Chu CR (2010) Donor sex and age influence the chondrogenic potential of human femoral bone marrow stem cells. Osteoarthr Cartil 18:705–713
Philippon MJ, Schenker ML, Briggs KK, Maxwell RB (2008) Can microfracture produce repair tissue in acetabular chondral defects? Arthroscopy 24:46–50
Karthikeyan S, Roberts S, Griffin D (2012) Microfracture for acetabular chondral defects in patients with femoroacetabular impingement: results at second-look arthroscopic surgery. Am J Sports Med 40:2725–2730
Zaltz I, Leunig M (2012) Parafoveal chondral defects associated with femoroacetabular impingement. Clin Orthop Relat Res 470:3383–3389
Fontana A, de Girolamo L (2015) Sustained five-year benefit of autologous matrix-induced chondrogenesis for femoral acetabular impingement-induced chondral lesions compared with microfracture treatment. Bone Joint J 97-B:628–635
Pestka JM, Bode G, Salzmann G, Sudkamp NP, Niemeyer P (2012) Clinical outcome of autologous chondrocyte implantation for failed microfracture treatment of full-thickness cartilage defects of the knee joint. Am J Sports Med 40:325–331
Ellender P, Minas T (2008) Autologous chondrocyte implantation in the hip: case report and technique. Oper Tech Sports Med 16:201–206
Mancini D, Fontana A (2014) Five-year results of arthroscopic techniques for the treatment of acetabular chondral lesions in femoroacetabular impingement. Int Orthop 38:2057–2064
Erggelet C, Endres M, Neumann K, Morawietz L, Ringe J, Haberstroh K, Sittinger M, Kaps C (2009) Formation of cartilage repair tissue in articular cartilage defects pretreated with microfracture and covered with cell-free polymer-based implants. J Orthop Res 27:1353–1360
Leunig M, Tibor LM, Naal FD, Ganz R, Steinwachs MR (2012) Surgical technique: second-generation bone marrow stimulation via surgical dislocation to treat hip cartilage lesions. Clin Orthop Relat Res 470:3421–3431
Fontana A (2016) Autologous membrane induced chondrogenesis (AMIC) for the treatment of acetabular chondral defect. Muscles Ligaments Tendons J 6:367–371
Lane JG, Healey RM, Chen AC, Sah RL, Amiel D (2010) Can osteochondral grafting be augmented with microfracture in an extended-size lesion of articular cartilage? Am J Sports Med 38:1316–1323
Hart R, Janecek M, Visna P, Bucek P, Kocis J (2003) Mosaicplasty for the treatment of femoral head defect after incorrect resorbable screw insertion. Arthroscopy 19:E1-5
Emre TY, Cift H, Seyhan B, Ceyhan E, Uzun M (2012) Mosaicplasty for the treatment of the osteochondral lesion in the femoral head. Bull NYU Hosp Jt Dis 70:288–290
Nam D, Shindle MK, Buly RL, Kelly BT, Lorich DG (2010) Traumatic osteochondral injury of the femoral head treated by mosaicplasty: a report of two cases. HSS J 6:228–234
Krych AJ, Lorich DG, Kelly BT (2012) Osteochondral autograft transfer for a posttraumatic osteochondral defect of the femoral head. Am J Orthop (Belle Mead NJ) 41:472–476
Girard J, Roumazeille T, Sakr M, Migaud H (2011) Osteochondral mosaicplasty of the femoral head. Hip Int 21:542–548
Bugbee WD, Convery FR (1999) Osteochondral allograft transplantation. Clin Sports Med 18:67–75
Williams SK, Amiel D, Ball ST, Allen RT, Tontz WL Jr, Emmerson BC, Badlani NM, Emery SC, Haghighi P, Bugbee WD (2007) Analysis of cartilage tissue on a cellular level in fresh osteochondral allograft retrievals. Am J Sports Med 35:2022–2032
Krych AJ, Lorich DG, Kelly BT (2011) Treatment of focal osteochondral defects of the acetabulum with osteochondral allograft transplantation. Orthopedics 34:e307–e311
Beaule PE, Zaragoza EJ (2003) Surgical images: musculoskeletal acetabular cartilage delamination demonstrated by magnetic resonance arthrography: inverted “Oreo” cookie sign. Can J Surg 46:463–464
Sekiya JK, Martin RL, Lesniak BP (2009) Arthroscopic repair of delaminated acetabular articular cartilage in femoroacetabular impingement. Orthopedics 32(9)
Beck M, Leunig M, Parvizi J, Boutier V, Wyss D, Ganz R (2004) Anterior femoroacetabular impingement: part II. Midterm results of surgical treatment. Clin Orthop Relat Res 418:67–73
Kennedy JG, Saunders RL (1984) Use of cryoprecipitate coagulum to control tumor-bed bleeding. Case report J Neurosurg 60:1099–1101
Lagoutte FM, Gauthier L, Comte PR (1989) A fibrin sealant for perforated and preperforated corneal ulcers. Br J Ophthalmol 73:757–761
Matthew TL, Spotnitz WD, Kron IL, Daniel TM, Tribble CG, Nolan SP (1990) Four years’ experience with fibrin sealant in thoracic and cardiovascular surgery. Ann Thorac Surg 50:40–44
Shah MA, Ebert AM, Sanders WE (2002) Fibrin glue fixation of a digital osteochondral fracture: case report and review of the literature. J Hand Surg Am 27:464–469
Silberstein LE, Williams LJ, Hughlett MA, Magee DA, Weisman RA (1988) An autologous fibrinogen-based adhesive for use in otologic surgery. Transfusion 28:319–321
Tzaveas AP, Villar RN (2010) Arthroscopic repair of acetabular chondral delamination with fibrin adhesive. Hip Int 20:115–119
Stafford GH, Bunn JR, Villar RN (2011) Arthroscopic repair of delaminated acetabular articular cartilage using fibrin adhesive. Results at one to three years. Hip Int 21:744–750
Chen A, Siow B, Blamire AM, Lako M, Clowry GJ (2010) Transplantation of magnetically labeled mesenchymal stem cells in a model of perinatal brain injury. Stem Cell Res 5:255–266
Mokbel AN, El Tookhy OS, Shamaa AA, Rashed LA, Sabry D, El Sayed AM (2011) Homing and reparative effect of intra-articular injection of autologus mesenchymal stem cells in osteoarthritic animal model. BMC Musculoskelet Disord 12:259
Bornes TD, Adesida AB, Jomha NM (2014) Mesenchymal stem cells in the treatment of traumatic articular cartilage defects: a comprehensive review. Arthritis Res Ther 16:432
Davatchi F, Abdollahi BS, Mohyeddin M, Shahram F, Nikbin B (2011) Mesenchymal stem cell therapy for knee osteoarthritis. Preliminary report of four patients. Int J Rheum Dis 14:211–215
Emadedin M, Aghdami N, Taghiyar L, Fazeli R, Moghadasali R, Jahangir S, Farjad R, Baghaban Eslaminejad M (2012) Intra-articular injection of autologous mesenchymal stem cells in six patients with knee osteoarthritis. Arch Iran Med 15:422–428
Lee KB, Hui JH, Song IC, Ardany L, Lee EH (2007) Injectable mesenchymal stem cell therapy for large cartilage defects—a porcine model. Stem Cells 25:2964–2971
Mardones R, Via AG, Jofre C, Minguell J, Rodriguez C, Tomic A, Salineros M (2016) Cell therapy for cartilage defects of the hip. Muscles Ligaments Tendons J 6:361–366
Field RE, Rajakulendran K, Strambi F (2011) Arthroscopic grafting of chondral defects and subchondral cysts of the acetabulum. Hip Int 21:479–486
Vundelinckx B, De Mulder K, De Schepper J (2012) Osteochondral defect in femoral head: Trufit implantation under fluoroscopic and arthroscopic control. Acta Orthop Belg 78:796–799
Shindle MK, Foo LF, Kelly BT, Khanna AJ, Domb BG, Farber A, Wanich T, Potter HG (2006) Magnetic resonance imaging of cartilage in the athlete: current techniques and spectrum of disease. J Bone Joint Surg Am 88(Suppl 4):27–46
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants.
Appendix
Appendix
Search strategy
Two reviewers (NN and CG) searched the online databases (PubMed (Medline), EMBASE, Google Scholar, BNI, CINAHL and AMED) for literature describing the outcome of cartilage repair techniques for the chondral injury in the hip. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were used for designing this study. Database search was conducted on 1st March 2017 and retrieved articles from database inception to the search date. The research question and individual study eligibility criteria were established a priori. We used medical subject headings including the following key search terms: hip, cartilage, chondral, repair, regeneration, restoration, refixation, implantation, chondroplasty and chondrogenic. Terms were connected by the Boolean operators ‘AND’ and ‘OR’. Levels I, II, III, IV and V evidence (according to the Oxford Centre for Evidence-Based Medicine) English-language studies were eligible for inclusion in the systematic review. The search also included the yet to be printed search results. Results were pooled, and duplicate searches were excluded by having two reviewers (NN and CG) independently review all the titles and abstracts. Both of the reviewers had been trained in a field of clinical research and had enough experience at the stage of abstract screening and manuscript review. Any discrepancies at the title and abstract stage were resolved by automatic inclusion to ensure thoroughness. The remaining search results were divided equally between two reviewers (NN and CG) and reviewed in duplicate applying the inclusion and exclusion criteria. Any discrepancies at the full-text stage were resolved by consensus between the two reviewers. If a consensus could not be reached, a third more senior reviewer (VK) was consulted to resolve the discrepancy. Also, for quality control, VK reviewed a 25% random sample of excluded studies and all included title and abstracts.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Nakano, N., Gohal, C., Duong, A. et al. Outcomes of cartilage repair techniques for chondral injury in the hip—a systematic review. International Orthopaedics (SICOT) 42, 2309–2322 (2018). https://doi.org/10.1007/s00264-018-3862-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-018-3862-6