Abstract
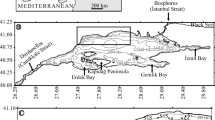
Foraminiferal data were obtained from 66 samples of box cores on the southeastern Brazilian upper margin (between 23.8°–25.9°S and 42.8°–46.13°W) to evaluate the benthic foraminiferal fauna distribution and its relation to some selected abiotic parameters. We focused on areas with different primary production regimes on the southern Brazilian margin, which is generally considered as an oligotrophic region. The total density (D), richness (R), mean diversity (\( \overline{{H^{\prime } }} \)), average living depth (ALD X ) and percentages of specimens of different microhabitats (epifauna, shallow infauna, intermediate infauna and deep infauna) were analyzed. The dominant species identified were Uvigerina spp., Globocassidulina subglobosa, Bulimina marginata, Adercotryma wrighti, Islandiella norcrossi, Rhizammina spp. and Brizalina sp.. We also established a set of mathematical functions for analyzing the vertical foraminiferal distribution patterns, providing a quantitative tool that allows correlating the microfaunal density distributions with abiotic factors. In general, the cores that fit with pure exponential decaying functions were related to the oligotrophic conditions prevalent on the Brazilian margin and to the flow of the Brazilian Current (BC). Different foraminiferal responses were identified in cores located in higher productivity zones, such as the northern and the southern region of the study area, where high percentages of infauna were encountered in these cores, and the functions used to fit these profiles differ appreciably from a pure exponential function, as a response of the significant living fauna in deeper layers of the sediment. One of the main factors supporting the different foraminiferal assemblage responses may be related to the differences in primary productivity of the water column and, consequently, in the estimated carbon flux to the sea floor. Nevertheless, also bottom water velocities, substrate type and water depth need to be considered.




Similar content being viewed by others

References
Altenbach AV, Pflaumann U, Schiebel R, Thies A, Timm S, Trauth M (1999) Scaling percentages and distributional patterns of benthic foraminifera with flux rates of organic carbon. J Foram Res 29(3):173–185
Alve AV, Bernhard JM (1995) Vertical migratory response of benthic foraminifera to controlled oxygen concentrations in an experimental mesocosm. Mar Ecol Prog Ser 116:137–151
Barbosa VP (1998) Foraminíferos Bentoˆnicos Quaterná rios do Talude Continental da Bacia de Santos: Sistema′ tica, Paleobatimetria e Paleoecologia. MSc thesis. Instituto de Geocieˆncias, Universidade Federal do Rio Grande do Sul, 427 pp
Barmawidjaja DM, Jorissen FJ, Puskaric S, Van der Zwaan GF (1992) Microhabitat selection by benthic foraminifera in the northern Adriatic Sea. J Foram Res 22:297–317
Berger WH, Wefer G (1990) Export production: seasonality and intermittency, and paleoceanographic implications. Palaeogeogr Palaeoclimatol Palaeoecol (Global and Planetary Ghange Section) 89:245–254
Berner RA, Raiswell R (1984) C/S method for distinguishing freshwater from marine sedimentary rocks. Geology 12:365–368
Bernhard JM (1986) Characteristic assemblages and morphologies of benthic anoxic, organic-rich deposits; Jurassic through Holocene. J Foram Res 16(3):207–215
Betzer PR, Showers WJ, Laws EA, Winn CD, Di Tullio GR, Kroopnick PP (1984) Primary productivity and particles fluxes on a transect of the equator at 153°W in the Pacific Ocean. Deep-Sea Res 31:1–11
Boltovskoy E, Giussani G, Watanabe S, Wright R (1980) Atlas of benthic shelf foraminífera of the southwest Atlantic. Dr. W. Junk by Publisher, The Hague, 147 p, 17 maps, 36pls
Brandini FP (2006) Hidrografia e produção biológica na região sudeste-sul do Brasil no contexto do programa Revizee. In: Rossi-Wongtschowski CLDB, Madureira LS-P (eds) O Ambiente oceanográfico da plataforma continental e do talude na região sudeste-sul do Brasil. EDUSP, São Paulo, pp 459–466
Bray JR, Curtis JT (1957) Na ordination of the upland forest communities in southern Wisconsin. Ecol Monogr 27:325–349
Burone L, Pires-Vanin AMS (2006) Foraminiferal assemblages in the Ubatuba Bay, Southeastern Brazilian coast. Sci Mar 70(2):203–217
Burone L, Venturini N, Sprechmann P, Valente P, Muníz P (2006) Foraminiferal responses to polluted sediments in the Montevideo coastal zone, Uruguay. Mar Pollut Bull 52:61–73
Burone L, Valente P, Pires-Vanin AMS, Sousa SHM, Mahiques MM, Braga E (2007) Benthic foraminiferal variability on a monthly scale in a subtropical bay moderately affected by urban sewage. Sci Mar 71(4):775–792
Buzas MA (1993) Colonization rate of foraminífera in the Indian River, Florida. J Foram Res 23:156–161
Campos EJD, Velhote D, Silveira ICA (2000) Shelf break upwelling driven by Brazil Current cyclonic meanders. Geophys Res Lett 27(6):751–754
Carbonel CAAH, Valentin JL (1999) Numerical modeling of phytoplankton bloom in the upwelling ecosystem of Cabo Frio (Brazil). Ecol Model 116:135–148
Carr ME, Friedrichs MAM, Schmeltz M, Aité MN, Antoine D, Arrigo KR, Asanuma I, Aumont O, Barber R, Behrenfeld M, Bidigare R, Buitenhuis E, Campbell J, Ciotti A, Dierssen H, Dowell M, Dunne J, Esaias W, Gentili B, Groom S, Hoepffner N, Hishisaka J, Kameda T, Le Quéré C, Lohrenz S, Marra J, Mélin F, Moore K, Morel A, Reddy T, Ryan J, Scardi M, Smyth T, Turpie K, Tilstone G, Waters K, Yamanaka Y (2005) A comparison of global estimates of marine primary production from ocean color. Deep-Sea Res II 53:741–770
Castro Filho BM, Miranda LB (1988) Physical oceanography of the Western Atlantic Continental Shelf located between 4°N and 34°S. In: Robinson AR, Brink KH (eds) The sea, vol 11. Wiley, New York, pp 209–251
Castro Filho BM, Miranda LB, Miyao SY (1987) Hydrographic conditions on the continental shelf offshore off Ubatuba: seasonal and middle scale variations. Bol Inst Oceanogr São Paulo 35:135–151
Corliss BH (1985) Microhabitats of benthic foraminifera within deep-sea sediments. Nature 314(4):435–438
Corliss BH (1991) Morphology and microhabitat preferences of benthic foraminífera from the northwest Atlantic Ocean. Mar Micropaleontol 17:195–236
Corliss BH, Emerson S (1990) Distribution of Rose Bengal stained deep-sea benthic foraminifera from the Nova Scotia continental margin and Gulf of Maine. Deep-Sea Res 37:381–400
Demaison GJ, Moore GT (1980) Anoxic environments and oil source bed genesis. Org Geochem 2:9–31
Duchemin G, Fontanier C, Jorissen FJ, Barras Ch, Griveaud C (2007) Living small-sized (63–150 μm) foraminifera from mid-shelf to mid-slope environments in the Bay of Biscay. J Foram Res 37(1):12–32
Fontanier C, Jorissen FJ, Licari L, Alexandre A, Anschutz P, Carbonel P (2002) Live benthic foraminiferal faunas from the Bay of Biscay: faunal density, composition, and microhabitats. Deep-Sea Res I 49(4):751–785
Gaeta SA, Brandini FP (2006) Produção primaria do Fitoplâncton na região entre o Cabo de Santo Tomé (RJ) r o Chuí (RS). In: Rossi-Wongtschowski CLDB, Madureira LS-P (eds) O Ambiente oceanográfico da plataforma continental e do talude na região sudeste-sul do Brasil. EDUSP, São Paulo, pp 219–264
Gaeta SA, Brino OL, Susini-Ribeiro SM (1994) Distributions of nitrate, chlorophyll and primary productivity in the southwestern region of the South Atlantic during Summer”. In: Abstracts of Southwestern Atlantic Physical Oceanography Workshop. USP/FAPESP, IOUSP, pp 57–60
Gooday A (1986) Meiofaunal foraminiferans from the bathyal Porcupine Seabight (northeast Atlantic): size structure, standing stock, taxonomic composition, species diversity and vertical distribution in the sediment. Deep-Sea Res 33(10):1345–1373
Gooday AJ (1988) A response by benthic foraminifera to the deposition of phytodetritus in the deep sea. Nature 332:70–73
Gooday AJ (1993) Deep-sea benthic foraminiferal species which exploit phytodetritus: Characteristic features and controls on distribution. Mar Microplaeontol 22:187–205
Gooday AJ, Rathburn AE (1999) Temporal variability in living deep-sea foraminifera: a review. Earth Sci Rev 46:187–212
Jones RW (1994) The challenger foramifera. Oxford Science Publications. The Natural History Museum. Oxford University Press, London 149 pp
Jorissen FJ (1999) Benthic foraminiferal microhabitats below the sediment-water interface. In: Sen Gupta BK (ed) Modern foraminifera. Kluwer, Dordrecht, pp 161–179
Jorissen FJ, Stigter HC, de Widmark JGV (1995) A conceptual model explaining benthic foraminiferal habitats. Mar Micropaleontol 26:3–15
Jorissen FJ, Fontanier C, Thomas E (2007) Paleoceanographical proxies based on deep-sea benthic foraminiferal assemblage characteristics. In: Hillaire-Marcel C, de Vernal A (eds) Proxies in late cenozoic paleoceanography. Elsevier, Amsterdam, pp 263–325
Kaiho K (1991) Global changes of paleogene aerobic/anaerobic benthic foraminifera and deep-sea circulation. Palaeogeogr Palaeoclimatol Palaecol 83:65–85
Kaiho K (1994) Benthic foraminiferal dissolved-oxygen index and dissolved-oxigen levels in the modern ocean. Geology 22:719–722
Kitazato H, Shirayama Y, Nakatsuka T, Fujiwara S, Shimanaga M, Kato Y, Okada Y, Kanda J, Yamaoka A, Masukawa T, Suzuki K (2000) Seasonal phytodetritus deposition and responses of bathyal benthic foraminiferal assemblages in Sagami Bay, Japan: preliminary results from ‘Project Sagami 1996–1999'. Mar Micropaleontol 40(3):135–149
Kruskal JB, Wish M (1978) Multidimensional scaling. California Sage, Beverly Hills
Linke P, Lutze GF (1993) Microhabitat preferences of benthic foraminifera—a static concept or a dynamic adaptation to optimize food acquisition? Mar Micropaleontol 20:215–234
Loeblich AR Jr, Tappan H (1988) Foraminiferal genera and their classification—PLATES, vol. 2. Van Nostrand Reinhold, New York, p 970
Loubere P (1991) Deep-sea benthic foraminiferal assemblage response to a surface ocean productivity gradient: a test. Paleoceanography 6:193–204
Loubere P, Fariduddin M (1999a) Quantitative estimation of global patterns of surface ocean biological productivity and its seasonal variation on time scales from centuries to millennia. Global Biogeochem Cycles 13:115–133
Loubere P, Fariduddin M (1999b) Benthic Foraminifera and the flux of organic carbon to the seabed. In: Sen Gupta BG (ed) Modern foraminifera. Kluwer, Netherlands, pp 182–199
Lutze GF, Coulbourn WT (1984) Recent benthic foraminifera from the continental margin off northwest Africa: community, structure and distribution. Mar Micropaleontol 8:361–401
Lutze GF, Thiel H (1989) Epibenthic foraminifera from elevated microhabitats: Cibicidoides wuellerstorfi and Planulina ariminensis. J Foram Res 19:153–158
Mackensen A, Douglas RG (1989) Down-core distribution of live and dead deep -water benthic foraminifera in box cores from the Weddell Sea and the California continental borderland. Deep-Sea Res 36(6):879–900
Mackensen A, Schmiedl G, Harloff J, Giese M (1995) Deep-sea foraminifera in the South Atlantic Ocean: ecology and assemblage generation. Micropaleontology 41(4):342–358
Mahiques MM, Mishima Y, Rodrigues M (1999) Characteristics of the sedimentary organic matter on the inner and middle continental shelf between Guanabara Bay and São Francisco do Sul, south-eastern Brazilian margin. Cont Shelf Res 19:775–798
Mahiques MM, Silveira ICA, Sousa SHM, Rodrigues M (2002) Post-LGM sedimentation on the outer shelf/upper slope of the northernmost part of the São Paulo Bight, south-eastern Brazil. Mar Geol 181:387–400
Mahiques MM, Tessler MG, Ciotti AM, Silveira ICA, Sousa SHM, Figueira RCL, Tassinari CCG, Furtado VV, Passos RF (2004) Hydrodinamically-driven patterns of recent sedimentation in the shelf and upper slope off southeast Brazil. Cont Shelf Res 24:1685–1697
Mahiques MM, Bícego MC, Silveira ICA, Sousa SHM, Lourenço RA, Fukumoto MM (2005) Modern sedimentation in the Cabo Frio upwelling system, Southeastern Brazilian shelf. An Academ Brasil Ciên 77:535–548
Mahiques MM, Tassinari CCG, Marcolini S, Violante R, Figueira RCL, Silveira ICA, Burone L, Sousa SHM (2008) Nd and Pb isotope signatures on the Southeastern South American Upper Margin: implications for sediment transport and source rocks. Mar Geol 250:51–56
Mahiques MM, Wainer IEKC, Burone L, Sousa SHM, Silveira ICA, Bícego MC, Alves DP, Hammer O (2009) A high-resolution Holocene record on the Southern Brazilian shelf: paleoenvironmental implications. Quat Inter 206:52–61
Martins V, Dubert J, Jouanneau J-M, Weber O, da Silva EF, Patinha C, Alverinho Dias JM, Rocha F (2007) A multiproxy approach of the Holocene evolution of shelf-slope circulation on the NW Iberian Continental Shelf. Mar Geol 239:1–18
Matsuura Y (1986) Contribuição ao estudo da estrutura oceanográfica da região sudeste entre Cabo Frio (RJ) e Cabo de Santa Marta Grande (SC). Ciencia e Cultura 38(8):1439–1450
Moodley L, Van der Zwaan GJ, Rutten GMW (1998) Subsurface activity of benthic foraminifera in relation to porewater oxygen content: laboratory experiments. Mar Micropaleontol 34:91–106
Murray JW (1991) Ecology and paleoecology of benthic foraminifera. Longman, Harlow
Murray JW (2003) Foraminiferal assemblage formation in depositional sinks on the continental shelf margin west of Scotland. J Foram Res 33:101–121
Murray JW (2006) Ecology and applications of benthic foraminifera. Cambridge University Press, Cambridge
Nagai RH, Sousa SHM, Burone L, Mahiques MM (2009) Paleoproductivity changes during the Holocene in the inner shelf of Cabo Frio, southeastern Brazilian continental margin: Benthic foraminífera and sedimentological proxies. Quat Int 206:62–71
Ohga T, Kitazato H (1997) Seasonal changes in bathyal foraminiferal assemblages in response to the flux of organic matter (Sagami Bay, Japan): Terra Nova 9(1):33–37
Pielou EC (1975) Ecological diversity. Jhon Wiley, New York, p 165
Piola AR, Campos EJD, Möller O, Charo M, Martínez C (2000) Subtropical shelf front off eastern South America. J Geophys Res 105(3):6565–6578
Raiswell R, Berner RA (1985) Pyrite formation in euxinic and semi-euxinic sediments. Am J Sci 285:710–724
Rodrigues RR, Lorenzzetti JA (2001) A numerical study of the effects of bottom topography and coastline geometry on the Southeast Brazilian coastal upwelling. Cont Shelf Res 21:371–394
Schmiedl G, de Bovée F, Buscail R, Charrière B, Hemleben C, Medernach L, Picon P (2000) Trophic control of benthic foraminiferal abundance and microhabitat in the bathyal Gulf of Lions, western Mediterranean Sea. Mar Micropaleontol 40(3):167–218
Schönfeld J (1997) The impact of the Mediterranean Outflow Water (MOW) on benthic foraminiferal assemblages and surface sediments at the southern Portuguese continental margin. Mar Micropaleontol 29:211–236
Schönfeld J (2001) Benthic foraminifera and pore-water oxygen profiles: a re-assessment of species boundary conditions at the western Iberian margin. J Foraminifer Res 31(2):86–107
Schönfeld J (2002a) A new benthic foraminiferal proxy for near-bottom current velocities in the Gulf of Cadiz, northeastern Atlantic Ocean. Deep-Sea Res I 49:1853–1875
Schönfeld J (2002b) Recent benthic foraminiferal assemblages in deep high-energy environments from the Gulf of Cadiz (Spain). Micropaleontology 44:141–162
Silveira ICA, Schmidt ACK, Campos EJD, Godoi SS, Ikeda Y (2000) A corrente do Brasil ao Largo da Costa Leste Brasileira. Rev Bras Oceanogr 48(2):171–183
Sjoerdsma PG, van der Zwaan GJ (1992) Simulating the effect of changing organic flux and oxygen content on the distribution of benthic foraminifera. Mar Micropaleontol 19:163–180
Sousa SHM, Passos RF, Fukomoto M, Silveira ICA, Figueira RCL, Koutsoukos EAM, Mahiques MM, Rezende CE (2006) Mid-lower bathyal benthic foraminifera of the Campos Basin, Southeastern Brazilian margin: Biotopes and controlling ecological factors. Mar Micropaleontol 61:40–57
Stein R (1991) Accumulation of organic carbon in marine sediments. Results from the Deep Sea Drilling Project/Ocean Drilling Program. Lecture Notes in: Earth Sciences, vol. 34. Springer, Berlin 217 p
Suess E (1980) Particulate organic carbon flux in the oceans-surface productivity and oxygen utilization. Nature 288:260–263
Suguio K (1973) Introdução à sedimentologia. Edgar Blücher/Editora da Universidade de São Paulo, São Paulo, p 317
Sumida PYG, Yoshinaga MY, Ciotti AM, Gaeta A (2005) Benthic response to upwelling events off SE Brazilian coast. Mar Ecol Prog Ser 291:35–42
Tissot B, Deroo G, Herbin JP (1979) Organic matter in cretaceous sediments of the North Atlantic: contribution to sedimentology and paleoceanography. In: Talwani M et al. (eds) Deep Drilling results in the Atlantic Ocean: continental margin and paleonvironment, Amer. Geophys. Union, M. Ewing Ser. vol 3. pp 362–374
Walton WR (1952) Techniques for recognition of living foraminifera. Contrib Cushman Found J Foram Res 3:56–60
Wieking G, Kröncke I (2005) Is benthic trophic structure affected by food quality? The Dogger Bank example. Mar Biol 146:387–400
Zembruscki SG (1979) Geomorphology of the southern Brazilian continental margin and adjacent oceanic basins. In: Chaves HAF (ed) Geomorphology of the Brazilian Continental Margin and Adjacent Oceanic Basins (Final report). REMAC Project Series, no. 7, Rio de Janeiro, Petrobrás, pp 129–177
Acknowledgments
This work was supported by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, Brazilian agency) via a post-doctoral fellowship (Proc. No. 06/59947-3) provided to L.B and via grant No. 03/10740-0. We are grateful to Joachim Schönfeld for the helpful comments and suggestions. The authors also wish to thank Mr. Daniel P. Alves and Mr. Edilson Faria for their help in the laboratory with the sedimentological analyses. We also thank Mrs. Fabiana Barbosa for the calculation of the primary productivity values.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by U.-G. Berninger.
Appendix
Appendix
1Hormosinella sp. (×65), 2Reophax sp. (×80), 3Reophax sp. (×65), 4Adercotryma wrighti (×700), 5Labrospira sp. (×100), 6Melonis sp. (×220), 7Lenticulina vitrea (×37), 8Hoeglundina elegans (×150), 9Pullenia quinqueloba (×230), 10Globocassidulina subglobosa (×800), 11Textularia candeiana (×100), 12Siphotextularia sp. (×190)
Rights and permissions
About this article
Cite this article
Burone, L., de Mello e Sousa, S.H., de Mahiques, M.M. et al. Benthic foraminiferal distribution on the southeastern Brazilian shelf and upper slope. Mar Biol 158, 159–179 (2011). https://doi.org/10.1007/s00227-010-1549-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-010-1549-7