Abstract
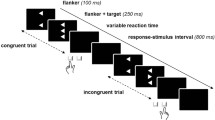
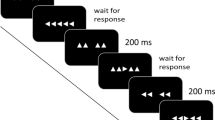
The role of dopaminergic system in decision-making is well documented, and evidence suggests that it could play a significant role in response selection processes. The N-40 is a fronto-central event-related potential, generated by the supplementary motor areas (SMAs) and a physiological index of response selection processes. The aim of the present study was to determine whether infraclinical effects of dopamine depletion on response selection processes could be evidenced via alterations of the N-40. We obtained a dopamine depletion in healthy volunteers with the acute phenylalanine and tyrosine depletion (APTD) method which consists in decreasing the availability of dopamine precursors. Subjects realized a Simon task in the APTD condition and in the control condition. When the stimulus was presented on the same side as the required response, the stimulus-response association was congruent and when the stimulus was presented on the opposite side of the required response, the stimulus-response association was incongruent. The N-40 was smaller for congruent associations than for incongruent associations. Moreover, the N-40 was sensitive to the level of dopaminergic activity with a decrease in APTD condition compared to control condition. This modulation of the N-40 by dopaminergic level could not be explained by a global decrease of cerebral electrogenesis, since negativities and positivities indexing the recruitment of the primary motor cortex (anatomically adjacent to the SMA) were unaffected by APTD. The specific sensitivity of N-40 to ATPD supports the model of Keeler et al. (Neuroscience 282:156–175, 2014) according to which the dopaminergic system is involved in response selection.



Similar content being viewed by others
References
Akkal D, Dum RP, Strick PL (2007) Supplementary motor area and presupplementary motor area: targets of basal ganglia and cerebellar output. J Neurosci 27(40):10659–10673. https://doi.org/10.1523/JNEUROSCI.3134-07.2007
Alexander GE, Crutcher MD (1990) Functional architecture of basal ganglia circuits: neural substrates of parallel processing. Trends Neurosci 13(7):266–271. https://doi.org/10.1016/0166-2236(90)90107-L
Amengual JL, Marco-Pallarés J, Grau C, Münte TF, Rodríguez-Fornells A (2014) Linking motor-related brain potentials and velocity profiles in multi-joint arm reaching movements. Front Hum Neurosci 8:271
Arezzo J, Vaughan HG Jr (1980) Intracortical sources and surface topography of the motor potential and somatosensory evoked potential in the monkey. Prog Brain Res 54:77–83. https://doi.org/10.1016/S0079-6123(08)61610-6
Bonini F, Burle B, Liégeois-Chauvel C, Régis J, Chauvel P, Vidal F (2014) Action monitoring and medial frontal cortex: leading role of supplementary motor area. Science 343(6173):888–891. https://doi.org/10.1126/science.1247412
Brown VJ, Robbins TW (1991) Simple and choice reaction time performance following unilateral striatal dopamine depletion in the rat. Brain 114(1):513–525. https://doi.org/10.1093/brain/114.1.513
Carbonnell L, Ramdani C, Meckler C, Burle B, Hasbroucq T, Vidal F (2013) The N-40: an electrophysiological marker of response selection. Biol Psychol 93(1):231–236. https://doi.org/10.1016/j.biopsycho.2013.02.011
Carvalhaes C, de Barros JA (2015) The surface Laplacian technique in EEG: theory and methods. Int J Psychophysiol 97(3):174–188. https://doi.org/10.1016/j.ijpsycho.2015.04.023
Coull JT, Cheng RK, Meck WH (2011) Neuroanatomical and neurochemical substrates of timing. Neuropsychopharmacology 36(1):3–25. https://doi.org/10.1038/npp.2010.113
Coull JT, Hwang HJ, Leyton M, Dagher A (2012) Dopamine precursor depletion impairs timing in healthy volunteers by attenuating activity in putamen and supplementary motor area. J Neurosci 32(47):16704–16715. https://doi.org/10.1523/JNEUROSCI.1258-12.2012
Deecke L, Grözinger B, Kornhuber HH (1976) Voluntary finger movement in man: cerebral potentials and theory. Biol Cybern 23(2):99–119. https://doi.org/10.1007/BF00336013
Falkenstein M, Hohnsbein J, Hoormann J, Blanke L (1991) Effects of crossmodal divided attention on late ERP components. II. Error processing in choice reaction tasks. Electroencephalogr Clin Neurophysiol 78(6):447–455. https://doi.org/10.1016/0013-4694(91)90062-9
Gehring WJ, Goss B, Coles MGH, Meyer DE, Donchin E (1993) A neural system for error detection and compensation. Psychol Sci 4(6):385–390. https://doi.org/10.1111/j.1467-9280.1993.tb00586.x
Giard MH, Besle J, Aguera PE, Gomot M, Bertrand O (2014) Scalp current density mapping in the analysis of mismatch negativity paradigms. Brain Topogr 27(4):428–437. https://doi.org/10.1007/s10548-013-0324-8
Gratton G, Coles M, Donchin E (1983) A new method for off-line removal of ocular artifact. Electroencephalogr Clin Neurophysiol 55(4):468–484. https://doi.org/10.1016/0013-4694(83)90135-9
Grillner S, Hellgren J, Menard A, Saitoh K, Wikström MA (2005) Mechanisms for selection of basic motor programs—roles for the striatum and pallidum. Trends Neurosci 28(7):364–370. https://doi.org/10.1016/j.tins.2005.05.004
Grillner S, Robertson B, Stephenson-Jones M (2013) The evolutionary origin of the vertebrate basal ganglia and its role in action selection. J Physiol 591(22):5425–5431. https://doi.org/10.1113/jphysiol.2012.246660
Hommel B, Proctor RW, Vu KP (2004) A feature-integration account of sequential effects in the Simon task. Psychol Res 68(1):1–17. https://doi.org/10.1007/s00426-003-0132-y
Kayser J, Tenke CE (2015) On the benefits of using surface Laplacian (current source density) methodology in electrophysiology. Int J Psychophysiol 97(3):171–173. https://doi.org/10.1016/j.ijpsycho.2015.06.001
Keeler JF, Pretsell DO, Robbins TW (2014) Functional implications of dopamine D1 vs. D2 receptors: a ‘prepare and select’ model of the striatal direct vs. indirect pathways. Neuroscience 282:156–175. https://doi.org/10.1016/j.neuroscience.2014.07.021
Kornblum S, Stevens GT, Whipple A, Requin J (1999) The effects of irrelevant stimuli: the time course of S-S and S-R consistency effects with Stroop-like stimuli, Simon-like tasks, and their factorial combinations. J Exp Psychol Hum Percept Perform 25:688–714
Larson MJ, Clayson PE, Primosch M, Leyton M, Steffensen SC (2015) The effects of acute dopamine precursor depletion on the cognitive control functions of performance monitoring and conflict processing: an event-related potential (ERP) study. PLoS One 10(10):e0140770. https://doi.org/10.1371/journal.pone.0140770
Leyton M, Young SN, Pihl RO, Etezadi S, Lauze C, Blier P, Baker GB, Benkelfat C (2000) Effects on mood of acute phenylalanine/tyrosine depletion in healthy women. Neuropsychopharmacology 22(1):52–63
Leyton M, Dagher A, Boileau I, Casey K, Baker GB, Diksic M, Gunn R, Young SN, Benkelfat C (2004) Decreasing amphetamine-induced dopamine release by acute phenylalanine/tyrosine depletion: a PET/[11C] raclopride study in healthy men. Neuropsychopharmacology 29(2):427–432. https://doi.org/10.1038/sj.npp.1300328
Luppino G, Matelli M, Camarda RM, Gallese V, Rizzolatti G (1991) Multiple representations of body movements in mesial area 6 and the adjacent cingulate cortex: an intracortical microstimulation study in the macaque monkey. J Comp Neurol 311(4):463–482. https://doi.org/10.1002/cne.903110403
Matsuzaka Y, Aizawa H, Tanji J (1992) A motor area rostral to the supplementary motor area (presupplementary motor area) in the monkey: neuronal activity during a learned motor task. J Neurophysiol 68(3):653–662. https://doi.org/10.1152/jn.1992.68.3.653
McNeely HE, West R, Christensen BK, Alain C (2003) Neurophysiological evidence for disturbances of conflict processing in patients with schizophrenia. J Abnorm Psychol 112(4):679–688. https://doi.org/10.1037/0021-843X.112.4.679
McTavish SF, Cowen PJ, Sharp T (1999) Effect of a tyrosine-free amino acid mixture on regional brain catecholamine synthesis and release. Psychopharmacology 141(2):182–188. https://doi.org/10.1007/s002130050823
Meckler C, Allain S, Carbonnell L, Hasbroucq T, Burle B, Vidal F (2010) Motor inhibition and response expectancy: a Laplacian ERP study. Biol Psychol 85(3):386–392. https://doi.org/10.1016/j.biopsycho.2010.08.011
Montague PR, Dayan P, Sejnowski TJ (1996) A framework for mesencephalic dopamine systems based on predictive Hebbian learning. J Neurosci 16(5):1936–1947
Montague PR, Hyman SE, Cohen JD (2004) Computational roles for dopamine in behavioural control. Nature 431(7010):760–767. https://doi.org/10.1038/nature03015
Montgomery AJ, McTavish SF, Cowen PJ, Grasby PM (2003) Reduction of brain dopamine concentration with dietary tyrosine plus phenylalanine depletion: an [11C] raclopride PET study. Am J Psychiatry 160(10):1887–1889
Mostofsky SH, Simmonds DJ (2008) Response inhibition and response selection: two sides of the same coin. J Cogn Neurosci 20(5):751–761. https://doi.org/10.1162/jocn.2008.20500
Nagano-Saito A, Leyton M, Monchi O, Goldberg YK, He Y, Dagher A (2008) Dopamine depletion impairs frontostriatal functional connectivity during a set-shifting task. J Neurosci 28(14):3697–3706. https://doi.org/10.1523/JNEUROSCI.3921-07.2008
Nagano-Saito A, Cisek P, Perna AS, Shirdel FZ, Benkelfat C, Leyton M, Dagher A (2012) From anticipation to action, the role of dopamine in perceptual decision making: an fMRI-tyrosine depletion study. J Neurophysiology 108(2):501–512. https://doi.org/10.1152/jn.00592.2011
Nuñez PL (1989) Estimation of large scale neocortical source activity with EEG surface Laplacians. Brain Topogr 2(1–2):141–154
Perrin F, Bertrand O, Pernier J (1987) Scalp current density mapping: value and estimation from potential data. IEEE Trans Biomed Eng 34(4):283–288
Picard N, Strick PL (1996) Motor areas of the medial wall: a review of their location and functional activation. Cereb Cortex 6(3):342–353. https://doi.org/10.1093/cercor/6.3.342
Picard N, Strick PL (2001) Imaging the premotor areas. Curr Opin Neurobiol 11(6):663–672. https://doi.org/10.1016/S0959-4388(01)00266-5
Proctor RW, Reeve TG (Eds.) (1990) Stimulus-response compatibility: an integrated perspective. Amsterdam: North-Holland
Ramdani C, Carbonnell L, Vidal F, Béranger C, Dagher A, Hasbroucq T (2015) Dopamine precursors depletion impairs impulse control in healthy volunteers. Psychopharmacology 232(2):477–487. https://doi.org/10.1007/s00213-014-3686-z
Rogers RD (2011) The roles of dopamine and serotonin in decision making: evidence from pharmacological experiments in humans. Neuropsychopharmacology 36(1):114–132. https://doi.org/10.1038/npp.2010.165
Sejnowski T, Churchland P (1990) Brain and cognition. In: Posner M (ed) Foundations of cognitive sciences. MIT Press, Cambridge
Simon JR (1990) The effects of an irrelevant directional cue on human information processing. In: Reeve RWPTG (ed) Stimulus-response compatibility. North-Holland, Elsevier Science Publishers, pp 31–86
Staude GH (2001) Precise onset detection of human motor responses using a whitening filter and the log-likelihood-ratio test. IEEE Trans Biomed Eng 48(11):1292–1305. https://doi.org/10.1109/10.959325
Sternberg S (2001) Separate modifiability, mental modules, and the use of pure and composite measures to reveal them. Acta Psychol (Amst) 106(1–2):147–246. https://doi.org/10.1016/S0001-6918(00)00045-7
Sternberg S (2011) Modular processes in mind and brain. Cognitive neuropsychology 28(3–4):156–208. https://doi.org/10.1080/02643294.2011.557231
Van Boxtel GJM, Geraats LHD, Van den Bref-Lessen MMC, Brunia CHM (1993) Detection of EMG onset in ERP research. Psychophysiology 30(4):405–412. https://doi.org/10.1111/j.1469-8986.1993.tb02062.x
Van de Laar MC, van den Wildenberg WP, van Boxtel GJ, Huizenga HM, van der Molen MW (2012) Lifespan changes in motor activation and inhibition during choice reactions: a Laplacian ERP study. Biol Psychol 89(2):323–334. https://doi.org/10.1016/j.biopsycho.2011.11.005
Vidal F, Burle B, Grapperon J, Hasbroucq T (2011) An ERP study of cognitive architecture and the insertion of mental processes: Donders revisited. Psychophysiology 48(9):1242–1251. https://doi.org/10.1111/j.1469-8986.2011.01186.x
West R, Alain C (2000) Effects of task context and fluctuations of attention on neural activity supporting performance of the Stroop task. Brain Res 873(1):102–111. https://doi.org/10.1016/S0006-8993(00)02530-0
Yoon JH, Larson P, Grandelis A, La C, Cui E, Carter CS, Minzenberg MJ (2015) Delay period activity of the substantia nigra during proactive control of response selection as determined by a novel fMRI localization method. J Cogn Neurosci 27(6):1238–1248. https://doi.org/10.1162/jocn_a_00775
Acknowledgements
We thank Dominique Reybaud, Bruno Schmid, and the pharmacy personnel of the hospital Sainte Anne for their helpful technical contribution.
Funding
The authors also gratefully acknowledge the financial support from the Institut de Recherches Biomédicales des Armées, France.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ramdani, C., Vidal, F., Dagher, A. et al. Dopamine and response selection: an Acute Phenylalanine/Tyrosine Depletion study. Psychopharmacology 235, 1307–1316 (2018). https://doi.org/10.1007/s00213-018-4846-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-018-4846-3