Abstract
Objective
To evaluate the diagnostic agreement between quantitative cultures of samples obtained with endotracheal aspiration (ETA) and plugged telescoping catheter (PTC).
Design
Prospective study.
Setting
Medical ICU.
Patients
Hundred thirty-eight episodes of suspected ventilator-associated pneumonia studied in 100 consecutive patients.
Interventions
For each suspected episode of ventilator-associated pneumonia, ETA and PTC were performed consecutively. The agreement between microbiological results obtained from the two techniques was evaluated (kappa statistic test). Pneumonia was diagnosed on a positive culture result of telescoping catheter with the threshold set at 103 cfu/ml or more. The cut-off points evaluated for ETA ranged from 102 to 106 cfu/ml.
Results
Micro-organisms retrieved by aspiration and telescoping catheter were similar and bacterial counts obtained by the two procedures were well correlated (r=0.71 p<0.001). There was good agreement between positive and negative ETA and PTC specimens (kappa: 0.78) with a diagnostic threshold for ETA of 104 cfu/ml. The sensitivity and specificity of ETA for the diagnosis of PTC-confirmed pneumonia were 92% and 85%, respectively. Kappa decreased to 0.48 when the diagnostic threshold was increased to 106 cfu/ml.
Conclusions
Quantitative cultures of ETA and PTC tallied for both micro-organisms and counts. The simpler ETA appears adequate for determining the presence of pathogenic organisms in significant concentration in the lower respiratory tract.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The accurate diagnosis of ventilator-associated pneumonia (VAP) remains a challenge for clinicians in the ICU setting. A diagnosis of VAP is usually based on a combination of clinical, radiological and microbiological criteria. Quantitative cultures of samples obtained by bronchoscopic techniques such as protected specimen brush sampling (PSB) and bronchoalveolar lavage (BAL) are currently viewed as the tests offering the best combination of sensitivity and specificity [1, 2]. However, routine use of these techniques is limited by the requirement of fiberoptic bronchoscopy, which may not be available on a 24-h basis in many intensive care units, and its interference with intrathoracic pressure, minute ventilation and blood oxygenation [3, 4].
Blind sampling of tracheobronchial specimens with plugged telescoping catheter (PTC) has been suggested as an alternative to more invasive tests, to increase the availability of protected specimens with quantitative culture and reduce the costs of the procedure [5, 6]. The accuracy of these diagnostic techniques accrue from autopsy studies showing that pneumonia often spreads to several pulmonary lobes and particularly to posterior portions of the lower lobes [7, 8]. Yet, among all the techniques used to retrieve bronchial secretions processed by quantitative procedures, tracheal aspiration remains the simplest method in mechanically ventilated patients. When only qualitative results are considered, endotracheal aspiration (ETA) has a good sensitivity but a low specificity [9, 10]. However, its specificity is improved when the tracheal aspirate culture is processed quantitatively. Marquette et al. [11] reported a fair correlation between PSB and ETA at a diagnostic threshold of 106 cfu/ml for the latter; in this study, ETA had a sensitivity of 82% and a specificity of 83%. Similar results were reported with a cut-off of 105 cfu/ml for ETA [12].
The purpose of this prospective study was to evaluate the diagnostic agreement of quantitative culture of samples obtained with ETA and PTC in ventilator-associated pneumonia.
Patients and methods
Patients
Over a 30–month period (from July 1997 to December 1999) a total of 138 clinically suspected VAP episodes were studied in 100 consecutive patients who had been intubated and mechanically ventilated for more than 48 h prior to the onset of clinical manifestations of VAP. Clinical suspicion of bacterial pneumonia was based on the occurrence of new pulmonary infiltrates on chest radiographs and at least two of the following criteria: fever (≥38°5c) or hypothermia (<36.5°c), the presence of purulent tracheobronchial secretions, leukocytosis (>10.109/l) or leukopenia (<3.109/l). The following information was obtained from the patients’ medical records: age, gender, main cause of ICU admission; the disease severity evaluated at admission and at the onset of VAP was assessed by the SAPS II scoring system [13]. In addition, for each episode studied, the following information was recorded: temperature, blood leukocyte count, PaO2/FIO2, radiological manifestation, the duration of prior mechanical ventilation and time between clinical suspicion of VAP and ICU admission.
Specimen collection
Endotracheal aspiration was always performed before PTC. The two procedures were undertaken consecutively and within a 15-min period of time.
Endotracheal aspirates were obtained by sterile suction using a mucus collector (Mucus extractor, ref 534.16, Vygon, Ecouen, France). PTC sampling procedures were conducted as described by Pham et al. [5]. After careful endotracheal suctioning, the PTC (Combicath, 5828.20 Plastimed lab, Saint Leu, France) was advanced blindly into the bronchus until it could not be advanced further. Then, it was retracted a few centimetres. The inner catheter was then advanced 2 or 3 cm beyond the tip of the outer catheter, extruding the plug. Three brief aspirations were then applied to the inner catheter with a 10-ml syringe connected to its proximal port. The catheter and the entire unit were removed from the patient. The distal portion of the outer catheter was wiped with a sterile pad. The inner catheter was then advanced and 1 ml of sterile saline was flushed through its proximal port and collected in a sterile vial. The distal segment of the inner catheter was transected with sterile scissors and collected in the same vial [5].
Bacteriological processing
The samples were transported to the microbiology laboratory within 15 min. Specimens were processed as soon as they were obtained. The samples were centrifuged and then aliquots of 0.01 and 0.1 ml were evaluated for growth 24 and 48 h later. The results were expressed as colony-forming units per millilitre (cfu/ml) of the original 1 ml dilution.
Data analysis
Cultures of potentially pathogenic micro-organisms were classified according to positive or negative. A previously established quantitative threshold (≥103 cfu/ml) for positive culture results of PTC was used and considered as the reference test for diagnosing nosocomial pneumonia. The cut-off points analysed to establish a positive result for ETA were 102, 103, 104, 105 and 106 cfu/ml.
The agreement between the microbiological results obtained from the two techniques was assessed using the kappa statistic test according to the method proposed by Landis and Koch [14, 15]: K less than 0.00, poor agreement; K =0.00–0.2, slight agreement; K =0.21–0.4, fair agreement; K =0.41–0.6, moderate agreement; K =0.61–0.81, substantial agreement; K =0.8–1, almost perfect. Sensitivity, specificity and predictive values were calculated according to standard formulae, taking into account the individual micro-organism species. Graphic representations of diagnostic parameters were performed, plotting true-positive against false-positive percentages to obtain a receiver operator characteristic (ROC) curve. The other results are expressed as means ± standard deviation or as percentages of total values.
Results
During the study period, a total of 138 episodes of VAP were suspected in 100 patients mechanically ventilated for more than 48 h (66 men and 34 women). The patients had a mean age of 56±19 years and mean SAPS II on admission of 36±13. Patients were admitted to the intensive care unit and required ventilator assistance because of respiratory failure (51), cerebrovascular disease (12), infectious disease (9), heart failure (18), drug overdose (2) or miscellaneous conditions (8).
The patients had received mechanical ventilation for 9.5±7 days prior to the suspicion of VAP. One hundred and four evaluations (75%) were made while the patients were receiving systemic antibiotics that had not been changed during the 72 h prior to sampling; the other patients were not on antibiotics. None of these antibiotic courses was a part of a selective digestive decontamination, which is not currently used in our ICU. No sampling procedure caused a significant complication. The characteristics of the patients are shown in Table 1.
The micro-organisms associated with pneumonia (identified by PTC sampling) are shown in Table 2. In 22 episodes, two or more micro-organisms were recovered and the diagnosis of polymicrobial pneumonia was made (Table 3). Gram-negative bacilli were the predominant organisms isolated (70%), represented mainly by Pseudomonas and Acinetobacter (38%) or Enterobacteriaceae (32%).
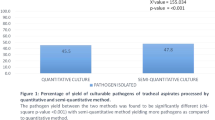
Thirty-four paired cultures were sterile. Eleven ETA yielded one or more micro-organisms while the PTC samples were sterile (Table 3). There was a significant correlation between the bacterial counts obtained by the two procedures (r=0.71, p<0.001; Fig. 1). Agreement between quantitative cultures of ETA and PTC was calculated over a range of cut-off values (from 102 to 106 cfu/ml) for ETA (Table 4). Using 104 cfu/ml as a diagnostic threshold for ETA, there was 92% and 85% agreement between positive and negative ETA and PTC specimens, respectively (kappa: 0.78). Increasing the diagnostic threshold for ETA to 105 cfu/ml or 106 cfu/ml was not associated with better agreement and the kappa statistic was lowered from 0.78 to 0.48.
The diagnostic value of ETA according to different thresholds of quantitative cultures is shown in Table 5. Depending on the threshold value of ETA, only three to eight episodes could be considered positive by ETA while negative by PTC. The best sensitivity/specificity ratio (92/85) was obtained using 104 cfu/ml. With the same threshold, the negative predictive value was 88%. The ROC curve for ETA is shown in Fig. 2.
Discussion
In this study designed to evaluate the diagnostic yield of two non-bronchoscopic sampling techniques in VAP, we observed good agreement between ETA and PTC in terms of micro-organisms retrieved. A threshold of 104 cfu/ml for ETA resulted in a 92% agreement between the two techniques with a sensitivity and specificity of 92% and 85%, respectively, for the diagnosis of VAP. Increasing the threshold to 105 cfu/ml or 106 cfu/ml for ETA resulted in a reduction in the agreement of these techniques.
The presence of prior antimicrobial treatment in up to 75% patients might be regarded as a limitation to accurate diagnosis of VAP, because it may lead to a high number of false-negative results. However, Timsit et al. have shown that the diagnostic yield of some of the procedures used to diagnose VAP (PSB and BAL) is not altered by the administration of long courses of antibiotics (>72 h) to treat earlier septic episodes [16]. Moreover, respiratory secretions usually carry a substantial number of bacteria when a super-infection occurs with a resistant micro-organism to antibiotics currently administered [17]. In addition, since ETA usually preceded PTC in our study, any modification of low respiratory tract flora would have played the same role with both samples.
The clinical diagnosis of VAP is difficult because clinical, biological and radiological signs are neither sensitive nor specific, especially in patients with pre-existing lung disease [18]. In addition, colonisation of the airways by potentially pathogenic micro-organisms is widespread in patients receiving mechanical ventilation. Hence, various techniques have been developed to obtain uncontaminated secretions from the lower airways. These methods include bronchoscopic or blind PSB, bronchoscopic or blind BAL and blind single-sheathed plugged telescoping catheter (PTC). Obviously, in the setting of left-sided pneumonia, bronchoscopic-guided collection of bronchial specimens should minimise the risk of false-negative results. Quantitative cultures of samples obtained by invasive bronchoscopic techniques (PSB and BAL) are currently regarded as the mainstay for the diagnosis of pneumonia. Moreover, Fagon et al. [19] have shown that the combination of quantitative culture and rapid direct examination of bronchoscopic PSB samples or BAL samples in an invasive strategy of management of suspected VAP reduced both mortality and antibiotic use through an early antibiotic cessation in almost 50% of cases. However, this study did not use quantitative cultures of ETA specimens in the control arm. In addition, the routine use of bronchoscopy is limited by its reduced availability in many intensive care units.
Plugged telescoping catheter with quantitative cultures, a technique that is widely used in France, is actually a safer procedure than the other invasive techniques using bronchoscopy [5]. In spite of its technical pitfalls (mainly the blind positioning of the distal tip), PTC has been established as a reliable and accurate procedure for the diagnosis of VAP [5, 20]. It has been validated in mechanically ventilated patients with bacterial infections, where both sensitivity and specificity of quantitative cultures of PTC have ranged between 60 and 100% [5, 21, 22]. The conventional technique of respiratory tract secretion sampling by ETA, however, remains the easiest and most widely used means to obtain bronchial secretions in mechanically ventilated patients. Despite the lack of specificity of non-quantitative cultures of ETA, it has recently gained renewed attention [23, 24, 25, 26]. Indeed, recent studies suggest that ETA associated with quantitative cultures might have an acceptable overall diagnostic accuracy, similar to that of more invasive techniques [11, 12, 26].
In a study by Marquette et al. [11] the operating characteristics of ETA associated with quantitative cultures compared favourably with those of PSB culture, with a slightly higher sensitivity and a lower specificity. Similarly, El Ebiary et al. [12] found, in 54 patients with suspected VAP, that this technique was relatively sensitive (70%) and specific (72%) for the diagnosis of pneumonia. In a study of 57 consecutive episodes of suspected lung infection, Jourdain et al. reported that the area under the ROC curve of ETA was 0.76 and the threshold of 106 cfu/ml appeared to be the most accurate, with a sensitivity of 68% and a specificity of 84% [27]. In addition, studies using histology as a “gold standard” found that microbial species that grew at a level higher than 107 cfu/ml in ETA samples were also found in lung tissue. Torres et al. [2] observed that the lowest rate of false-positive results was obtained using thresholds of 106 for ETA, 105 for PSB and 106 for BAL [2]. The authors reported a specificity of 85% for ETA at 106 cfu/ml, 85% for PSB at 104 cfu/ml and 78% for BAL at 106 cfu/ml.
More recently, two prospective studies using post mortem histological examination of the lung as the “gold standard” assessed the accuracy of cultures obtained using ETA, PSB and BAL [20, 28]. In the first one Marquette et al. [20] found similar sensitivity and specificity of quantitative cultures of samples retrieved by ETA, PSB and BAL. When the diagnostic threshold of ETA was reduced from 106 to 105 cfu/ml, the sensitivity increased and specificity decreased. Papazian et al. [28] reported an area under the ROC curve significantly greater for ETA than for PSB. The authors concluded that, given that the non-invasive ETA was more sensitive, it should be preferred to PSB for the diagnosis of VAP.
To the best of our knowledge, only one study has previously compared the accuracy of ETA and PTC in the diagnosis of VAP [29]. Forty-six bronchial samplings were obtained from 31 patients with suspected VAP. In this study, the authors observed a lower agreement (76%) than that observed in our study. The sensitivity (89.5%) and the specificity (66.7%) of ETA were also below the levels we recorded. We cannot readily account for these differences, particularly since the design of the study by Fangio et al. [29] systematically excluded VAP that developed in the left lung, which would tend to increase the sensitivity of the sampling method under evaluation.
In conclusion, our study shows that quantitative cultures of ETA and PTC were in agreement for both micro-organisms and bacterial counts. Thus, the simpler ETA appears adequate for routine diagnosis of VAP, provided quantitative cultures are obtained.
References
Pingleton SK, Fagon JY, Leeper Jr KV (1992)Patient selection for clinical investigation of ventilator-associated pneumonia: criteria for evaluating diagnosis techniques. Chest 102:5535–5565
Torres A, Martos A, Puig de la Ballacasa J, Ferrer M, el-Ebiary M, Gonzalez J, Gene A, Rodriguez-Roisin R (1993) Specificity of endotracheal aspiration, protected specimen brush, and bronchoalveolar lavage in mechanically ventilated patients. Am Rev Respir Dis 147:952–957
Trouillet JL, Guiget M, Gibert C, Fagon JY, Dreyfuss D, Blanchet F, Chastre J (1990) Fiberoptic bronchoscopy in ventilated patients. Evaluation of cardiopulmonary risk under midazolam sedation. Chest 97:927–933
Montravers P, Gauzit R, Dombert MC, Blanchet F, Desmonts JM (1993) Cardio-pulmonary effects of bronchoalveolar lavage in critically ill patients. Chest 104:1541–1547
Pham LH, Brun-Buisson C, Legrand P, Rauss A, Verra F, Brochard L, Lemaire F (1991) Diagnosis of nosocomial pneumonia in mechanically ventilated patients. Comparison of a plugged telescoping catheter with the protected specimen brush. Am Rev Respir Dis 143:1055–1061
Girault C, Saigne L, Jusserand D, et al. (1997) Evaluation technique du prélèvement bronchique distal protégé à l’aveugle par cathéter en réanimation. Réan Urg 6:667–674
Johanson WG Jr, Seidenfeld JJ, Gomez P, de los Santos R, Coalson JJ (1988) Bacteriologic diagnosis of nosocomial pneumonia following prolonged mechanical ventilation. Am Rev Respir Dis 137:259–264
Fabregas N, Torres A, El-Ebiary M, Ramirez J, Hernandez C, Gonzalez J, Puig de la Bellacasa J, de Anta J, Rodriguez-Roisin R (1996) Histopathologic and microbiologic aspects of ventilator associated pneumonia. Anesthesiology 84:760–771
Higuchi JG, Coalson JJ, Johanson WG (1982) Bacteriologic diagnosis of nosocomial pneumonia in primates. Am Rev Respir Dis 125:53–57
Lambert RS, Vereen LE, George RB (1989) Comparison of tracheal aspirates and protected brush catheter specimens for identifying pathogenic bacteria in mechanically ventilated patients. Am J Med Sci 297:377–382
Marquette CH, Georges H, Wallet F, Ramon P, Saulnier F, Neviere R, Mathieu D, Rime A, Tonnel AB (1993) Diagnostic efficiency of endotracheal aspirates with quantitative bacterial cultures in intubated patients with suspected pneumonia. Comparison with the protected specimen brush. Am Rev Respir Dis 148:138–144
El-Ebiary M, Torres A, Gonzales J, de la Bellacasa JP, Garcia C, Jimenez de Anta MT, Ferrer M, Rodriguez-Roisin R (1993) Accuracy of quantitative cultures of endotracheal aspirates for the diagnosis of ventilator associated pneumonia. Am Rev Respir Dis 148:1552–1557
Le Gall JR, Lemshow S, Saulnier F (1993) A new simplified acute physiology score (SAPS II) based on a European/North America multicenter study. JAMA 270:2957–2963
Fleiss JL (1981) Statistical methods for rates and proportions, 2nd edn. Wiley, New York, pp 212–236
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–174
Timsit JF, Misset B, Renaud B, Goldstein FW, Carlet J (1995) Effect of previous antimicrobial therapy on the accuracy of the main procedures used to diagnose nosocomial pneumonia in patients who are using mechanical ventilation. Chest 108:1036–1040
Chastre J, Fagon JY (2002) Ventilator-associated pneumonia. State of the art. Am J Respir Crit Care Med 165:867–903
Villers D (1990) Critères cliniques, radiologiques et biologiques des pneumopathies nosocomiales. Réan Soins Intens Med Urg 97:1208–1219
Fagon JY, Chastre J, Wolff M, Gervais C, Parer-Aubas S, Stéphan F, Similowski T, Mercat A, Diehl JL, Sollet JP, Tenaillon A (2000) Ann Intern Med 132:621–630
Marquette C, Chopin M, Wallet F (1995) Diagnostic tests for pneumonia in ventilated patients. Prospective evaluation of diagnostic accuracy using histology as a diagnostic gold standard. Am J Respir Crit Care Med 151:1878–1888
Jorda R, Parras F, Ibanez J, Reina J, Bergada J, Raurich JM (1993) Diagnosis of nosocomial pneumonia in mechanically ventilated patients by the blind protected telescoping catheter. Intensive Care Med 19:377–382
Torres A, Puig de la Bellacasa J, Xaubert A, Gonzalez J, Rodriguez-Roisin R, Jimenez de Anta, MT, Agusti Vidal A (1989) Diagnostic value of quantitative cultures of bronchoalveolar lavage and telescoping plugged catheters in mechanically ventilated patients. Am Rev Respir Dis 140:306–310
Berger R, Arango L (1985) Etiologic diagnosis of bacterial nosocomial pneumonia in seriously ill patients. Crit Care Med 13:833–836
Hill JD, Ratliff JL, Parrot JC, Lamy M, Fallat RJ, Koeniger E, Yaeger EM, Whitener G (1976) Pulmonary pathology in acute respiratory insufficiency: lung biopsy as a diagnostic tool. J Thorac Cardiovasc Surg 71:64–71
Lambert RS, Vereen LE, George RB (1989) Comparison of tracheal aspirates and protected brush catheter specimens for identifying pathogenic bacteria in mechanically ventilated patients. Am J Med Sci 377:377–382
Salata RA, Liederman MM, Shlaes DM, Jacobs MR Eckstein E, Tweardy D, Toossi Z, Chmielewski R, Marino J, King CH (1987) Diagnosis of nosocomial pneumonia in intubated, intensive care unit patients. Am Rev Respir Dis 135:426–462
Jourdain B, Novara A, Joly-Guillon M, Dombert M (1995) The diagnosis of quantitative cultures of endotracheal aspirates in the diagnosis of nosocomial pneumonia. Am J Respir Crit Care Med 152:241–246
Papazian L, Thomas P, Garbe L, Guignon I, Thirion X, Charrel J, Bollet C, Fuentes P, Gouin F (1995) Bronchoscopic or blind sampling techniques for the diagnosis of ventilator-associated pneumonia. Am J Respir Crit Care Med 152:1981–1991
Fangio P, Rouquette Vincenti, Rousseau JM, Soullié B, Brinquin L (2002) Diagnosis of ventilator-associated pneumonia: a prospective comparison of the telescoping plugged catheter with the endotracheal aspirate. Ann Fr Anesth Reanim 21:184–192
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Elatrous, S., Boukef, R., Ouanes Besbes, L. et al. Diagnosis of ventilator-associated pneumonia: agreement between quantitative cultures of endotracheal aspiration and plugged telescoping catheter. Intensive Care Med 30, 853–858 (2004). https://doi.org/10.1007/s00134-004-2270-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-004-2270-0