Abstract
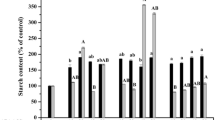
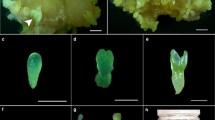
Low efficiency of somatic embryo maturation, germination, and conversion to plantlets is a major problem in many species including J. regia L. Germination efficiency of somatic embryos is very low in walnut. In this study, effects of two carbohydrate sources, sucrose and maltose (each at 3% and 6%), and two kinds of PEG (4000 and 6000) (each at four levels of 1.5%, 3%, 5% and 7.5%) on maturation and germination of walnut embryos were tested. The number of somatic embryos increased conspicuously on medium containing PEG. Furthermore, higher levels of PEG-4000 (7.5%) could remarkably enhance the morphogenesis of somatic embryos and the number of embryos produced. PEG-4000 stimulated somatic embryo maturation of walnut. This stimulatory effect was dependent on the carbohydrate source used. Sucrose in combination with PEG-4000 produced 50% of cotyledonary and normal somatic embryos. Different concentrations of PEG were effective on the number of embryos with a shoot meristem. PEG-4000 7.5% and sucrose 3.0% produced the highest rate (50.0%) of normal shooting embryos. However, PEG (4000, 6000) and maltose caused an unfavorable effect and increased the frequency of abnormal shaped somatic embryos.
Similar content being viewed by others
Abbreviations
- ABA:
-
Abscisic acid
- DKW:
-
Driver & Kuniyuki walnut medium
- PEG:
-
Polyethylene glycol
- SE:
-
Somatic embryo
- CE:
-
Cotyledonary embryo
References
Anandarajah K, McKersie B.D. 1990. Enhanced vigour of dry somatic embryos of Medicago sativa L. with increased sucrose. J. Plant Sci.71: 261–266
Attree SM, Moore D, Sawhney VK, Fowke LC. 1991. Enhanced maturation and desiccation tolerance of white spruce (Picea glauca (Moench.) Voss) somatic embryos: effects of a non-plasmolysing water stress and abscisic acid. J. Ann. Bot. 68: 519–525
Attree SM, Pomeroy MK, Fowke LC. 1995. Development of white spruce (Picea glauca (Moench.) Voss) somatic embryos during culture with abscisic acid and osmoticum and their tolerance to drying and frozen storage. J. Exp. Bot. 46: 433–439
Blanc G, Michaux-Ferrière N, Teisson C, Lardet L, Carron MP. 1999. Effects of carbohydrate addition on the induction of somatic embryogenesis in Hevea brasiliensis. J. Plant Cell Tiss. Org. Cult. 59: 103–112
Capuana M, Debergh PC. 1997. Improvement of the maturation and germination of horse chestnut somatic embryos. J. Plant Cell Tiss. Org. Cult. 48: 23–29
Deng MD, Cornu D. 1992. Maturation and germination of walnut somatic embryos. J. Plant Cell Tiss. Org. Cult. 28: 195–202
Driver JA, Kuniyuki AH. 1984. In vitro propagation of Paradox walnut rootstock. J. Hort. Sci. 19: 507–509
Find J. 1997. Changes in endogenous ABA levels in developing somatic embryos of Norway spruce [Picea abies (L.) Karst] in relation to maturation medium, desiccation and germination. J. Plant Sci. 128: 75–83
Fuentes SRL, Calheiros MBP, Manetti-Filho J, Vieira LGE. 2000. The effects of silver nitrate and different carbohydrate sources on somatic embryogenesis in Coffea canephora. J. Plant Cell Tiss. Org. Cult. 60: 5–13
Garin E, Isabel N, Plourde A. 1998. Screening of large numbers of seed families of Pinus strobus L. for somatic embryogenesis from immature and mature zygotic embryos. J. Plant Cell Rep.18: 37–43
Kauk K. 1995. Somatic embryogenesis in eastern white pine (Pinus strobus L.). In SM Jain, PK Gupta, RJ Newton, eds, Somatic embryogenesis in woody plants, Vol 3. Gymnosperms. Dordrecht: Kluwer Academic Publishers; 257–268
Leal I, Misra S, Attree SM, Fowke LC. 1995. Effect of abscisic acid, osmoticum, and desiccation on 11S storage protein gene expression in somatic embryos of white spruce. J. Plant Sci. 106: 121–128
Lee BC, Shim SY, Lee SK. 1988. Mass propagation and germination of somatic embryos in juglans regia L. (English walnut). Res. Rep. Inst. For. Genet Korea.24: 99–106
Lecouteux CG, Lai FM, McKersie BD. 1993. Maturation of alfalfa (Medicago sativa L.) somatic embryos by abscisic acid, sucrose and chilling stress. J. Plant. Sci. 94: 207–213
Li XY, Huang FH, Grur EE. 1997. Polyethylene glycol-promoted development of somatic embryos in loblolly pine (Pinus taeda L.). J. In Vitro Cell. Dev. Biol. Plant. 33: 184–189
Li XY, Huang FH, Gbur EE. 1998. Effect of basal medium, growth regulators and phytagel concentration on initiation of embryogenic cultures from immature zygotic embryos of loblolly pine (Pinus taeda L.). J. Plant Cell Rep. 17: 298–301
Linossier L, Veisseire P, Cailloux F, Coudret A. 1997. Effects of abscisic acid and high concentration of PEG on Hevea brasiliensis somatic embryos development. J. Plant. Sci.124: 183–191
Mathur G, Von Arnold S, Nadgauda R. 2000. Studies on somatic embryogenesis from immature zygotic embryos of chir pine (Pinus roxburghii Sarg.). J. Curr. Sci. 79: 999–1004
McGranahan G, Leslie C, Uratsu S, Dandekar A. 1990. Improved efficiency of the walnut somatic embryo gene transfer system. J. Plant Cell Rep. 8: 512–516
Miguel C, Goncalves S, Tereso S, Marum L, Oliveira MM. 2004. Somatic embryogenesis from 20 open-pollinated families of Portuguese plus trees of maritime pine. J. Plant Cell Tiss. Org. Cult. 76: 121–130
Norgaard JV. 1997. Somatic embryo maturation and plant regeneration in Abies nordmanniana LK. J. Plant Sci. 124: 211–221
Pullman GS, Webb DT. 1994. An embryo staging system for comparison of zygotic and somatic embryo development. Proceedings of the TAPPI R&D Division Biological Sciences Symposium, Oct 3-6, 1994, Minneapolis, MN. TAPPI Press, Atlanta, GA, pp. 31–34
Ramarosandratana A, Harvengt L, Bouvet A, Calvayrac R, Pâques M. 2001. Influence of the embryonal-suspensor mass (ESM) sampling on development and proliferation of maritime pine somatic embryos. J. Plant Sci. 160: 473–479
Salajova T, Salaj J, Kormutak A. 1999. Initiation of embryogenic tissues and plantlet regeneration from somatic embryos of Pinus nigra Arn. J. Plant Sci. 145: 33–35
Svobodova H, Albrechtova J, Kumstyrova L, Lipavska H, Vagner M, Vondrakova Z. 1999. Somatic embryogenesis in Norway spruce: anatomical study of embryo development and influence of polyethylene glycol on maturation process. J. Plant Physiol. Biochem. 37: 209–221
Stasolla C, Van Zyl L, Egertsdotter U, Craig D, Liu WB, Sederoff RR. 2003. The effect of polyethylene glycol on gene expression of developing white spruce somatic embryos. Plant Physiol. 131: 49–60
Tang H, Ren Z, Reustle G, Krczal G, Germaine E. 2001. Optimizing secondary somatic embryo production in English walnut (Juglans regia L.). Acta Hort. 544: 187–194
Tremblay L, Tremblay FM. 1991. Carbohydrate requirements for the development of black spruce (Picea mariana (Mill.) B.S.P.) and red spruce (P. rubens Sarg.) somatic embryos. J. Plant Cell Tiss. Org. Cult. 27: 95–103
Tulecke W, McGranahan G. 1985. Somatic embryogenesis and plant regeneration from cotyledons of walnut, Juglans regia L. J. Plant Sci. 40: 57–63
Vahdati K, Bayat Sh, Ebrahimzadeh H, Jariteh M, Mirmasoumi M. 2008. Effect of exogenous ABA on somatic embryo maturation and germination in Persian walnut (Juglans regia L.). J. Plant Cell Tiss. Org. Cult.93: 163–171
Vahdati K, Jariteh M, Niknamm V, Mirmasoumi M, Ebrahimzadeh H. 2006. Somatic embryogenesis and embryo maturation in Persian walnut. Acta Hort. 705: 199–205
Walker DR, Parrott WA. 2001. Effect of polyethylene glycol and sugar alcohols on soybean somatic embryo germination and conversion. J. Plant Cell Tiss. Org. Cult. 64: 55–62
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jalali, M.A., Sirmandi, H.B. & Hatamzadeh, A. Effects of carbohydrate source and polyethylene glycol on maturation and germination of somatic embryos in walnut (Juglans regia L.). J. Crop Sci. Biotechnol. 20, 29–35 (2017). https://doi.org/10.1007/s12892-016-0089-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12892-016-0089-1