Abstract
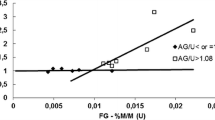
Many analytical methods for glycerol involve chemical techniques or instrumentation that may be unavailable to small biodiesel processors or have potential interactions with other components in the glycerol phase. This work presents an analytical method based on periodate oxidation of glycerol to formaldehyde, which is then quantified using a modification of the Nash test required to remove excess periodate. The method provides a linear calibration over the range of diluted glycerol concentrations from 0.02 to 0.25 mM and is limited at high concentrations by the formation of precipitate during the Nash test. There are no observed interferences from the formic acid coproduct of the glycerol oxidation or from potential contaminants in the crude glycerol, which includes methanol and sodium. Several disaccharides and starch, both as individual solutes or in glycerol solutions, gave no response under the conditions of the described method.







Similar content being viewed by others
References
Hu S, Luo X, Wan C, Li Y (2012) Characterization of crude glycerol from biodiesel plants. J Agric Food Chem 60:5915–5921
Reddy SR, Titu D, Chadha A (2010) A novel method for monitoring the transesterification reaction of oil in biodiesel production by estimation of glycerol. J Am Oil Chem Soc 87:747–754
Plank C, Lorbeer E (1995) Simultaneous determination of glycerol, and mono-, di-, and triglycerides in vegetable oil methyl esters by capillary gas chromatography. J Chromatogr A 697:461–468
AOCS Official Method Ea 6-51 (2009) Glycerol, sodium periodate oxidation method
Pisarello ML, Dalla Costa BO, Veizaga NS, Querini CA (2010) Volumetric method for free and total glycerin determination in biodiesel. Ind Eng Chem Res 49:8935–8941
AOCS Official Method Ea 6-94 (2009) Determination of crude glycerin, titrimetric method
AOCS Official Method Ca 14-56 (2009) Total, free, and combined glycerol iodometric-periodic acid method
Bok SH, Demain AL (1977) An improved colorimetric assay for polyols. Anal Biochem 81:18–20
Fagnani E, Melios CB, Pezza L, Pezza HR (2003) Chromotropic acid–formaldehyde reaction in strongly acidic media. The role of dissolved oxygen and replacement of concentrated sulphuric acid. Talanta 60:171–176
Taylor KA (1997) A colorimetric formaldehyde assay. Appl Biochem Biotechnol 68:81–93
Kleeberg U, Klinger W (1982) Sensitive formaldehyde determination with Nash’s reagent and a tryptophan reaction. J Pharmacol Methods 8:19–31
Quesenberry MS, Lee YC (1996) A rapid formaldehyde assay using purpald reagent: application under periodation conditions. Anal Biochem 234:50–55
Nash T (1953) The colorimetric estimation of formaldehyde by means of the Hantzsch reaction. Biochem J 55:416–421
Bondioli P, Della Bella L (2005) An alternative spectrophotometric method for the determination of free glycerol in biodiesel. Eur J Lipid Sci Technol 107:153–157
Hughes G, Nevell TP (1948) The mechanism of the oxidation of glucose by periodate. Trans Faraday Soc 44:941–948
Acknowledgments
Financial support for this work was provided by the Department of Chemical Engineering, Villanova University. This work was also supported by a donation of analytical equipment made by Air Products and Chemicals, Inc. The authors thank Dr. Charles Coe for his helpful discussions regarding this manuscript.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Bompelly, R., Skaf, D.W. Optimization of a Colorimetric Test Method for Quantifying Glycerol in Aqueous Solution. J Am Oil Chem Soc 91, 1605–1610 (2014). https://doi.org/10.1007/s11746-014-2502-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11746-014-2502-6